Survey of Chemical Substances in Consumer Products, No. 88, 2007
A survey and health assessment of cosmetic products for children
Contents
2 Definition of cosmetic products for children
- 4.1 Survey of cosmetic products for children
- 4.2 Database of cosmetic products for children
- 4.3 Results of the survey
- 4.3.1 Product name
- 4.3.2 Importers/producers
- 4.3.3 How are the products procured?
- 4.3.4 Product type
- 4.3.5 Marketing of the products – directly towards children
- 4.3.6 Chemical substances contained in the products
- 4.3.7 Constituents with hazard classification
- 4.3.8 Constituents in the guiding list for self-classification of the Danish Environmental Protection Agency
- 4.3.9 Constituents with restrictions in relation to the statutory order
- 4.3.10 Specification of number of constituents
- 4.3.11 Content of perfume
- 4.3.12 Content of preservatives
- 4.3.13 No content of triclosan
- 4.3.14 Content of colouring agents
- 4.3.15 Design of packaging and CE labelling
- 4.3.16 Plastic type of the packaging
- 4.3.17 Bar code
- 4.3.18 Batch number
- 5.1 Selection of products for analysis
- 5.2 Analyses for selected constituents
- 5.3 Analyses of selected packaging materials
- 8.1 Risk assessment based on the analyse results
- 8.2 The analyse results for packaging
- 8.3 General health assessment of the cosmetic products for children
- 8.4 Recommendations on purchase and apply of cosmetic products for children
Appendix A – Extractions from the database of cosmetic products for children
- 1 Overview of the content of the database
- 2 All chemical substances found in the mapped products
- 3 Chemical substances in the mapped products distributed on the type of products
- 4 Constituents with restrictions
Appendix B – The 26 fragrances mandatory to declare
Preface
This project ”A survey and health assessment of cosmetic products for children” is carried out for the Danish Environmental Protection Agency by FORCE Technology. The conducted analyses are partly carried out by Eurofins (analyses of constituents) and partly by FORCE Technology (analyses of packaging material).
The purpose was to map the Danish market of cosmetic products for children and based on this to select a number of products for chemical analysis partly of constituents and partly of constituents in the packaging material. Furthermore, the purpose was to select a number of substances of which a health assessment is carried out.
The project is completed in a period of eight months (April to November) in 2006. The study itself of the cosmetic products for children is completed in the months April to June 2006.
As it can be seen from the study new regulations became effective in March 2005 regarding declaration of 26 fragrances mandatory to declare. According to the Cosmetics Statutory Order these fragrances (being listed in Appendix B to this report) have to be stated in the the declaration of content when the concentration is higher than 0.001% in products which are not to be cleansed after use and 0.01% in products which are to be cleansed after use.
The results of the analysis show that a few products do not comply with this new regulation in the Cosmetics Statutory Order regarding separate declaration of the 26 fragrances. According to the Cosmetics Statutory Order the new regulations apply to products being produced after this date (March 2005). It is possible that the analyzed products are produced before March 2005 and thus meet the rules of the Cosmetics Statutory Order.
Summary and conclusions
Background and purpose
A number of the cosmetic products, especially within the ranges of shampoo, body shampoo and soap, are marketed as products for children. The packaging materials of the products are often very colourful and may be designed as for instance cartoon figures. This means that in some cases the products may also be used as toys.
Based on the assumption that this market is increasing the Danish Environmental Protection Agency started this survey in the spring of 2006 with the purpose to map cosmetic products for children marketed on the Danish market.
The purpose of the survey was to get an overview of the chemical substances that the products contain and the constituents which may be problematic for children’s health. The focus of the project was on allergenic fragrances and preservatives.
Finally, this project has had the purpose to test the packaging material for the cosmetic products for children for a content of possible problematic substances. Focus has been on packaging materials designed as special figures and selected packaging materials are tested for a content of heavy metals and phthalates among other things.
The survey
The project is conducted by FORCE Technology, Department of Sustainability and Management, who has been in charge of the survey, the selection of products for analysis as well as the health assessment. The analyses of the constituents of the products were conducted by Eurofins, while the analyses of the constituents of the packing materials were conducted by FORCE Technology, Department of Chemical Analysis.
The survey of the cosmetic products for children was completed in the period from April to June 2006 by shopping in all kinds of shops selling cosmetic products for children. Shops as chemists, toy stores, 10-kroners shops, 1000 Things shops, convenience stores, supermarkets, bookstores, children’s clothes stores, hairdressers, department stores and some other shops were visited – mainly in the northern part of Zealand and in the Copenhagen area. Furthermore, products were ordered via the Internet and declarations of content being available via the Internet have also been used in the survey.
Main conclusions
The project shows that cosmetic products for children in the age of 3-14 years represent a large market. The volume of the yearly sale of cosmetic products for children is not investigated but the number of different products shows that cosmetic products for children represent a significant market. In total 208 different products were identified in the survey.
The survey is not complete as especially up to the Christmas trade many bath products for children designed as different cartoon figures are found, but the survey is estimated to include the majority of the products which are daily found on the Danish market.
The most important conclusions from the survey are:
- In total 461 different constituents were found in the 208 mapped products. An average product contained 16 different constituents.
- The most frequently used constituents in the cosmetic products for children were water and perfume.
- A large part of the cosmetic products for children contained perfume – in total 74%. 34% of all products had a content of one or more of the 26 fragrances which must be declared separately in the declaration of content.
- More than the half (63%) of the products contained preservatives. 12 of the 23 applied preservatives are considered to be allergenic or potentially allergenic.
- More than the half (57%) of the products contained colouring agents.
- None of the mapped products contained triclosan.
- About a quarter (56 products or 27%) of the products was designed as a figure. For another quarter (24%) of the products the packing material was decorated with pictures of special cartoon figures but without a special design of the packaging material. The remaining products had either a quite neutral packaging or a colourful but neutral design of the packaging.
- Of the 56 products where the packaging was designed as a figure the Danish Safety Technology Authority has indicatively stated that 19 products were comprised by the legislation on toys.
- The analyses of selected packaging materials showed that all the tested bath products designed as a figure and made in soft plastic are made of PVC. These packaging materials contain between 26 and 31% phthalates.
The most significantly health impacts to which children can be exposed when using the tested cosmetic products for children are assessed to be the risk of allergy due to the added fragrances and preservatives. Furthermore, in some cases the packaging can contain phthalates of which some are on the EU list of endocrine-disrupting substances. With the new phthalate legislation which became effective as per 16 April 2007 it is now banned to apply these phthalates in toys for children up to 14 years and in child care articles in concentrations above 0.1%. This ban covers the all 10 products, where the packaging material are analyzed for content of phthalates, since they are either comprised by the legislation on toys or are defined as child care articles.
Project results
In total 208 different cosmetic products for children were identified in the survey, distributed on the following product types (listed after falling number of mapped products):
- Body shampoo/bath gel
- Bath confetti/caviar/fizzle salt
- Shampoo
- Bobble bath
- Tooth paste
- Body lotion/cream
- Soap - solid
- Eau de toilette - perfume
- Bath oil
- Others (for instance body spray products)
- Hair dye (rinsing colour)
- Balsam
- Hairstyling products
- Deodorant
- Massage oil
- Soap - liquid
A large part of the products contained allergenic substances
A large part of the mapped products contained allergenic substances in the form of fragrances, preservatives and/or colouring agents. 74% of all products contained perfume but if the products with no declaration of content available as well as the non-perfumed tooth paste products are left out 86% of the remaining products had a content of perfume. About one-third of the products contained one or more of the 26 fragrances which must be declared separately.
More than the half (63%) of the products contained preservatives. A number of preservatives is considered to be allergenic. Twelve of the 23 applied preservatives are considered to be allergenic or potentially allergenic. Among the six most frequently applied preservatives five were parabens of which propyl paraben, butyl paraben and isobutyl paraben are a subject for a debate about endocrine disrupting effects.
Correspondingly, more than the half (57%) of the products contained colouring agents. Few colouring agents being considered to be allergenic were found in the cosmetic products for children and these colouring agents were only found in the hair dye products (rinsing colours) – seven products in total. The EU Commission has banned 22 substances in hair dyes per 1 December 2006. None of these colouring agents were found in the mapped products for children.
Analysis of the products
The project focus was on the allergenic properties of the cosmetic products for children. Therefore, 17 products were selected for analysis of their quantitative content of the 26 fragrances mandatory to declare as the EU considers them to be allergenic. Additionally, 11 products have been analyzed for their quantitative content of the allergenic preservative Kathon[1] which was found in 15 products.
The analyses showed a total content of the 26 fragrances of up to 7,800 mg/kg (0.78%) – found in an eau de toilette product. The analyses also showed that six products did not comply with the legislation for the declaration of content as described in the Cosmetics Statutory Order. One of the products had no declared content of “perfume” in the product even if the analysis showed fragrances in the product. For the five other products, a declaration of a few of the 26 fragrances was missing as these fragrances according to the analyses are found in a concentration in the products so they have to be declared separately as described in the Cosmetics Statutory Order.
According to the Cosmetics Statutory Order the new legislation applies for products being produced after March 2005. It is possible that the analyzed products are produced before March 2005 and therefore still keep the Cosmetics Statutory Order.
The analyses of the 11 products for Kathon showed that none of the products exceeded the permissible content of 15 mg/kg. However, in three of the products a content of Kathon was identified even if the products according to the declaration of content had no content of Kathon.
Analysis of the packaging material of the products
A number of products (14) were examined for a content of chlorine in the packaging material as an indication that the material was made of PVC. Based on the plastic type code on the packaging the survey showed that eight other products had packaging of PVC. In total 10 packaging materials were examined for the content of phthalates. The phthalate types being identified were primarily DEHP and DINP. The total phthalate content in the examined packaging materials was 31% as a maximum.
All the examined products with a content of phthalates were designed as a cartoon figure, an animal or similar. DEHP and DINP were banned as per 16 April 2007 in toys for children up to 14 years in a concentration above 0.1% - for DINP only in toys which children might put into the mouth. The Danish Safety Technology Authority has indicatively stated that two of the analyzed products are toys while the other analyzed products are child care articles. Some of these products will thus be in discrepancy with the new phthalate legislation. All the analyzed products were legal at the time of analysis (November 2006).
Ten products were analyzed for their content of heavy metals in the packaging material. All the measured total values of heavy metals in the packaging materials are significantly below the set threshold values indicating the amount of metals which may migrate out of the toys when in use. The measured total values are not cause for concern in relation to the existing threshold limits for migration of substances from toy materials. For the same reason no migration analyses of selected products were conducted.
Health assesment/risk assesment
On the basis of the results of the quantitative analyses of cosmetic products for children a risk assessment of the analyzed products for their content of Kathon and benzyl alcohol is conducted based on the acute toxicity of the substances.
The risk assessment showed that the margin of safety (MoS) is far from being exceeded in most cases. In general, there is no health risk by using the analyzed products in relation to the content of Kathon and benzyl alcohol respectively.
1 Introduction
A number of cosmetic products, especially shampoo, soap, eau de toilette and hair dyes, are marketed as products for children. The Danish Environmental Protection Agency had the assumption that the market for this product group is increasing. The packaging of the products is often very colourful and can be designed as for instance cartoon figures. This causes that the products can also be used as toys in some cases.
The products are regulated by the Cosmetics Statutory Order (Danish: Kosmetikbekendtgørelsen) and in some cases also the regulations for toys. It is the Danish Safety Technology Authority who as a competent authority assesses which products coming under the regulations for toys. In this project there is a distinction between products being assessed to be designed as for instance figures and therefore can be used as toys, and products which according to the indicative statement of the Danish Safety Technology Authority are assessed to be covered by the regulations for toys.
The purpose of the project was to
- Map whether cosmetic products for children contain substances which can be problematic for children’s health. Focus is especially on perfume substances, preservatives and colouring agents.
- Map whether the product packaging causes that the products can be used as toys.
- Investigate whether cosmetic products for children meet the labelling requirements for cosmetics (i.e. whether the declaration of contents and producer is stated) and possibly also for toys (CE marking).
- Assess possible health consequences of the found constituents in cosmetic products for children.
- Assess possible health consequences of the found constituents in the packaging of products which can be used as toys.
2 Definition of cosmetic products for children
The project deals with cosmetic products for children. In this connection two things ought to be defined: What are children? And what are cosmetic products for children?
According to the ILO (International Labour Organisation) convention – C13 Minimum Age Convention (ILO, 1973) children are defined as being below 15 years when it comes to age limits for child labour. “Young workers” are defined as being older than a child, i.e. 15 years and above, but below 18 years.
In this project the same definition of children is used, i.e. children are up to 14 years. This corresponds to the limit being used in the Toys Statutory Order (Danish: Legetøjsbekendtgørelsen). Here toys are defined as products which clearly are designed or intended for play purposes for children below 14 years” (Stat. Ord. 1116, 2003).
However, there is also a need for a low limit as the project by definition from the Danish Environmental Protection Agency does not cover products for babies. Babies or infants are often defined as children less than 1 year or children who cannot walk yet. However, the purpose of this project is to survey cosmetic products aimed at children or attractive for children, i.e. it is essential to include an aspect regarding when children can express a wish to get something. It is expected that this ability is not particularly labelled until the age of about 3 years. Therefore, the age of 3 years is used as the low limit in this project.
In other words, cosmetic products for children are in this connection defined as products focusing on children at the age of 3-14 years.
Insofar as products for quite small children are not included in the survey the aspect that small children can suck the toys is neither included.
In this project, cosmetic products for children are defined as:
- Products directed to children. I.e. they are labelled with ”Kids”, ”Children’s” (Danish: “Børne-“) or ”Junior”.
- Products which by virtue of their appearance are directed at children (for instance in the form of a car or princess figure). In other words, they have a special design or a special appearance appealing to children.
The cosmetic products do not include:
- Decorative cosmetics, i.e. make-up, nail varnish, Shrovetide make-up, as this area has earlier been covered by the survey projects of the Danish EPA.
- Lip care products if even they are especially directed to children as this area has earlier been covered by the survey projects of the Danish EPA (No. 55 “Survey of lip care products with scent, taste etc.”).
- Suntan lotion. This is a wish from the Danish EPA as suntan lotion also includes other problems, aside from allergenic substances.
- Products, especially directed to babies and infants. I.e. among others products with the label inscription “baby”.
Table 2.1 is an overview of the types of cosmetic products for children.
Table 2.1: Types of cosmetic products for children
| Products for children | |
| Products labelled especially for children with ”Kids” or similar | Hair shampoo Balsam/balsam spray Body shampoo/bath gel Body lotion/cream Eau de toilette/perfume Deodorant Massage oil[2] Soap (solid soap – small soap figures or liquid) Hair dyes (rinsing colours) Bath oil Bath confetti/bath caviar/fizzle salt Hairstyling products Bobble bath Toothpaste |
| Products with special design so it can be confused with toys | Hair shampoo Balsam Body shampoo/bath gel Body lotion Bobble bath Eau de toilette/perfume Can all be designed as dragoon, cars, Star Wars, Disney, Barbie etc. However, especially bobble bath is designed as all sorts of figures. |
3 Legislation
In the following there is a short description of the legislation which is relevant for cosmetic products for children. It is the Cosmetics Statutory Order (Danish: Kosmetikbekendtgørelsen) which is relevant for all cosmetic products for children and the Toys Statutory Order (Danish: Legetøjsbekendtgørelsen) which is only relevant for the cosmetic products for children being assessed as toys on basis of the indicative statement of the Danish Safety Technology Authority.
3.1 The Cosmetics Statutory Order
Cosmetic products for children are like other cosmetic products regulated in “Cosmetics Statutory Order” – Statutory Order no. 422 dated 4 May 2006 (Stat. Ord. 422, 2006). The statutory order implements the European provisions in the cosmetics area and includes a number of provisions about apply of chemical substances in cosmetics as well as about labelling of the products.
According to § 10 of the Cosmetics Statutory Order cosmetic products that are marketed in the EU must not be harmful to human health when they are used under normal conditions or under conditions which reasonably can be foreseen. The Cosmetics Statutory Order states among other things a number of restrictions of application of chemical substances that form a part of cosmetic products.
The one who markets the cosmetics product has the responsibility that the regulations are kept according to the statutory order.
3.1.1 Labelling of cosmetic products for children
Cosmetic products (among these also products for children) have to be labelled with the following information on the actual product (Stat. Ord. 422, 2006):
- Company name and address of the manufacturer who within the EU is responsible for the marketing (§ 19).
- Content in weight or volume (if more than 5 g or more than 5 ml (§ 20)).
- Durability date (if the shelf life is below 30 months (§ 21)).
When specifying the durability a symbol with an open cream box is used stating the durability in months and years.
- Safety regulations for application (§ 22).
- The number of the production batch or the reference specification so that date and place of production can be identified (§ 23).
- The function of the agent (unless it appears from its presentation (§ 24).
- Declaration of contents, i.e. a list of the constituents of the product arranged in order after descending weight at that time the constituents are added to the cosmetic agent (§ 25).
3.1.2 Declaration of contents
The following special circumstances have their effects on the declaration of contents of cosmetic products (Stat. Ord. 422 § 25, 2006):
- Impurities are not considered to be constituents.
- Scented or aromatic compounds shall only be stated by the term “perfume” or “aroma” respectively. According to appendix 3 of the statutory order 26 fragrances which the EU regards as allergenic have to be stated in the declaration of contents irrespective of their function in the products when the concentration is higher than 0.001% in products which are not to be cleansed and 0.01% in products which are to be cleansed. This regulation with special statement of the 26 fragrances mandatory to declare became effective in 2005 and applies for all cosmetics being produced after 10 March 2005.
- Constituents in a concentration less than 1% can be mentioned in any order after the constituents with a concentration higher than 1%.
- Hair dyes can be mentioned with a colour index number (or indication from appendix 4 on colouring agents) in any order after the other constituents.
- The constituents are stated by their usual name according to the common nomenclature (INCI name) for cosmetics ingredients.
- Regarding small cosmetic products or packaging of such a form that in practice it is impossible to state the declaration of content clearly enough the ingredients must be stated on an enclosed notice, label, strip or card which is fastened to the cosmetics product. If it is impossible to fasten a message or similar to products (because for instance they are too small) the declaration of content must be stated in a notice close to the products.
INCI is an abbreviation of ”International Nomenclature Cosmetic Ingredients” and is a common nomenclature for apply in declarations of contents for cosmetic products in the EU. An INCI name can cover several different chemical units. The INCI list is indicative, this means it is not a list of approved constituents in cosmetics but a list indicating the constituents that are used (Pharmacos, 2001). If an INCI name for a constituent is not available the chemical name of the substance is to be used and an application for an INCI name for the substance shall be sent (Stat. Ord. 422, 2006).
3.1.3 Restrictions in apply of chemical substances in cosmetic products
The Cosmetics Statutory Order (Stat. Ord. 422, 2006) sets a number of restrictions in apply of cosmetic products. Among others, which substances that are not allowed to be used in cosmetic products, which substances that only are allowed to be used under special conditions (for instance, at a maximum concentration) as well as which substances that only are allowed to be used (positive lists) within a special group (f. ex. hair dyes, preservatives).
Substances which are not allowed in cosmetic products
According to §12 of the Cosmetics Statutory Order “substances which are entered in the appendix 2 (of the statutory order) are not allowed as constituents in cosmetic products”.
Allowed substances with limitations in cosmetic products
According to §13 of the Cosmetics Statutory Order substances listed in appendix 3 may be used in cosmetic products under the limitations and the conditions stated in the appendix.
Allowed hair dyes in cosmetic products
According to §14 of the Cosmetics Statutory Order ”cosmetic products (except the hair dyes exclusively intended to hair dyeing) are only allowed to contain the hair dyes as well as lacquers, salts and pigments from the lists which are entered in the appendix 3 and 4 (of the statutory order) and with those restrictions and conditions stated in these”.
Allowed preservatives in cosmetic products
According to § 15 of the Cosmetics Statutory Order ”cosmetic products are not allowed to contain other preservatives than those entered in the appendix 5 (of the statutory order)”.
Allowed U.V. absorbers in cosmetic products
According to § 16 of the Cosmetics Statutory Order ”cosmetic products are not allowed to contain other U.V. absorbers than those entered in the appendix 6 (of the statutory order)”. However, other U.V. absorbers exclusively used in cosmetic products to protect the products themselves against decomposition of U.V. radiation are not included in the appendix 6 (of the statutory order).
3.2 Toys
3.2.1 Safety requirements to toys
For toys the ”Statutory order on safety requirements to toys and products which due to their appearance can be confused with food” applies (Stat. Ord. 1116 dated 12 December 2003). Toys are defined as products ”which clearly are designed or determined for play purposes for children below 14 years”. This means when the cosmetic products for children are designed as a figure (a dragoon, Barbie, Mickey Moapply, a mobile phone or similar) then the statutory order on safety requirements to toys must also be met.
According to the Toys Statutory Order (Stat. Ord. 1116, 2003), a toy is only allowed to be sold if it meets the EU legislation on safety requirements to toys or if it is manufactured in accordance with a customs-approved prototype (and approved by a regulatory body in an EU country). Toys meeting these safety requirements must be assigned with a CE label before it is sold on the market.
The EU legislation on safety requirements includes the standards stated in the appendix 3 of the Toys Statutory Order (Stat. Ord. 1116, 2003). These are the EN71 series on safety requirements to toys as well as the Statutory Order regarding high voltage current for electrical toys. Furthermore, toys are not allowed to contain hazardous substances or compounds as defined in directive 67/548/EØF and 88/379/EØF in quantities which can be harmful to health for the children using it.
3.2.2 Ban on phthalates in toys for children
According to statutory order no. 151 dated 15 March 1999 “Statutory order on ban on phthalates in toys for children in the age 0-3 years as well as certain child care articles” it is prohibited to produce, sell or import products containing more than 0.05% (w/w) phthalates for the following types of products:
- Toys that is clearly designed or intended for play purposes for children in the age 0-3 years.
- Child care articles for children in the age 0-3 years, i.e. products that are intended to or must be expected to be put into the mouth (dummies, bibs, jewelries as well as bathing equipment etc.).
- Products that must be expected to be used as toys by children in the age 0-3 years as a consequence of the product construction and design including choice of subject.
However, this statutory order is replaced by a new ”Statutory order on ban on phthalates in toys and child care articles” which became effective as per April 2007 (Stat. Ord. 786, 2006; Stat. Ord. 1074, 2006). The new regulations include ban on six phthalates in child care articles and in toys for children up to 14 years.
According to the new statutory order it is prohibited to apply, import and sell products containing the phthalates DEHP, DBP and BBP in concentrations above 0.1%. Furthermore, it is prohibited to apply, import and sell the phthalates DINP, DIDP and DNOP in concentrations above 0.1% in products which children might put into the mouth.
Finally, it is prohibited to apply all other kind of phthalates in concentrations above 0.05% in toys for children from 0-3 years as well as in child care articles for children in the age of 0-3 years which is intended or may normally be expected to be put into the mouth by the children.
4 Survey
- 4.1 Survey of cosmetic products for children
- 4.2 Database of cosmetic products for children
- 4.3 Results of the survey
- 4.3.1 Product name
- 4.3.2 Importers/producers
- 4.3.3 How are the products procured?
- 4.3.4 Product type
- 4.3.5 Marketing of the products – directly towards children
- 4.3.6 Chemical substances contained in the products
- 4.3.7 Constituents with hazard classification
- 4.3.8 Constituents in the guiding list for self-classification of the Danish Environmental Protection Agency
- 4.3.9 Constituents with restrictions in relation to the statutory order
- 4.3.10 Specification of number of constituents
- 4.3.11 Content of perfume
- 4.3.12 Content of preservatives
- 4.3.13 No content of triclosan
- 4.3.14 Content of colouring agents
- 4.3.15 Design of packaging and CE labelling
- 4.3.16 Plastic type of the packaging
- 4.3.17 Bar code
- 4.3.18 Batch number
One of the purposes of this project was to map the chemical substances which can be found in cosmetic products for children on the Danish market. First and foremost, such a survey takes place by surveying the cosmetic products that are sold in Denmark. The starting point of the project was to collect in total 200 cosmetic products for children as it was expected that such a number would cover a considerable part of the cosmetic products for children on the Danish market. (Quantity considerations are not included in the evaluation of the market coverage).
In the project a database of the mapped cosmetic products for children was developed for the Danish Environmental Protection Agency. Below is described how information about the products was procured and how the database of the products is built-up and which information is entered. Finally, the results of the survey and various extractions from the database are presented.
4.1 Survey of cosmetic products for children
The survey of cosmetic products for children in Denmark has taken place by use of the following sources:
- Association of Danish Cosmetics, Toiletries, Soap and Detergent Industries (SPT).
- Searches on the Internet
- Systematic purchase of cosmetic products for children in retail shops and via purchase on the Internet.
No complete survey of all cosmetic products for children being marketed in Denmark has taken place but a database with the 208 cosmetic products for children identified in the project has been developed.
Information about the content of chemical substances in the cosmetic products for children is procured in two different ways, either by purchasing the products or by asking for products or declarations of contents directly from the importers/producers. The survey of the products is carried out during a period of approximately 2 months (mainly from mid-April to mid-June 2006 and that is the reason why most of the products have been bought, as this was far the fastest way to get the products.
However, the declaration of contents for a few products has been available via the Internet and in these cases the information has thus been used.
According to SPT only a few of the Danish producers/importers market cosmetic products for children directly. One of these has sent the declarations of contents of their products electronically.
Products from both the retail shops and via the Internet have been bought so that the survey of the cosmetic products for children could cover as many products as possible. A few products can only be ordered by mail order or via the Internet and these products have also been bought. A few products are found at hairdressers. However, emphasis has been on products in the retail shops as it is estimated to be the most important way of shopping.
The survey is undertaken by buying products in the retail shops and at hairdressers at first. Then a search on the Internet after products not found in the retail shops has been completed. The few products bought via the Internet are thus primarily products which are not easily found in the retail shops. Conversely, many of the products in the retail shops can be bought via the Internet.
We evaluate that we have got hold of a significant part of the cosmetic products for children on the Danish market. However, the survey has shown that bath products designed as different figures – for instance Minnie Mouse, Barbie, Star Wars etc. – primarily are a product type being sold up to Christmas. When survey and thus the purchases in the project took place during the months April, May and June it has not been possible to procure all products of this type. It has primarily been the leftovers from the Christmas trade which we have bought. In the light of this we estimate that we have got hold of about 85-95 % of the products on the Danish market.
One of the big importers of bath products for children which are designed as different figures has informed that they re-sell these products to shops in connection with the Christmas trade. One of the big foreign producers of this product type has been contacted as we hoped to receive declarations of contents of their products electronically – but with no result.
Furthermore, the project has shown that the products on the market are frequently replaced; this means that new products are frequently launched on the market with new appearance and form. It was especially seen when a few products being chosen for analysis were impossible to find again. On the other hand a few quite new products had entered the market. However, this case occurs especially for bath products designed as different animals or things.
4.1.1 Purchase of cosmetic products for children in retail shops
As described, purchase of products in the retail shops has been the primary way of finding the products. Search for cosmetic products for children has taken place in different types of shops (convenience stores, toy stores, children’s clothes stores, bookstores, various “10 kroner” stores and hairdressers) – mainly in the area of Copenhagen and Northern part of Zealand. Table 4.1 is an overview of the shops where cosmetic products for children are found as well as where we have looked for cosmetic products for children without any result. No found cosmetic products for children in for instance Kvickly, Super Brugsen or Fakta mean that they only had products on the shelves which already were found elsewhere.
Table 4.1: Shops where we have bought and looked for cosmetic products for children
| Shops where we have looked for products without any result | Shops where we have bought products |
| Bahne Bam Bam Bog & Idé Crabtree & Evelyn (Lyngby) Debenhams Deres Elefant Esthetique Fakta Frisørhapplyt Frisør Nørgaard Fætter BR Gun-Britt Helsemin (Field’s) Inspiration Irma Kejserens Nye Klær’ (Gentofte) Kvickly Netto Nørgaard Olfert og Ofelia Only SuperBrugsen Søstrene Grene The Body Shop Toppen og Bolden Toys’R’Us |
B. O. Bøger Bilka Capella Toy Cha Cha Cha Din Helsam Butik Douglas Føtex Gad Guldklumperne (Allerød) Gyngehesten Hair Construction Hennes & Mauritz Lotus Magasin Matas Stuhr Søs Coiffure Tiger Urtehapplyt Victoria's Zara Ærlige Bent Ærlige Bent / EXPO |
4.1.2 Purchase of cosmetic products for children via the Internet
After purchase of products in the retail shops a search for cosmetic products for children via the Internet has been conducted. Products not already bought were ordered via the Internet. Products were bought on the following home pages:
- www.babynature.dk
- www.duft-natur.dk
- www.jydsklasercenter.dk
- www.konceptkids.dk
- www.kropsprodukter.dk
- www.lavera.dk
- www.nuno.dk
- www.onlinekids.dk
- www.oriflame.dk
- www.satika.dk
4.1.3 Which types of products were bought?
As described in chapter 2 ”Definition of cosmetic products for children”, cosmetic products for children in this project are defined as products aiming at children in the age 3-14 years and they are products directly addressed to children (i.e. they are labelled with “Kids”, “Children’s” or “Junior”) or they are products which in their appearance are directed at children (i.e. they have either a special appearance or a special look appealing to children).
Most of the products are directly marketed to children either via the name or the appearance. But with regard to a number of products it can be discussed whether they are products directly intended for children. However, these products are included in the survey because they appeal to children due to their appearance and because in some cases they have even been found on the same shelf as other cosmetic products for children.
Among others, a number of products in the product category “Bath confetti/bath caviar/fizzle salt” are bought. This category covers product types like bath confetti, bath caviar and bath salt. These three product types are products to be used in the bath tub. The products are put into the bath water and will slowly be dissolved. Typically, they have both a soap function and a scent function and generally this product type is designed as colourful flowers, petals, hearts or similar. These products might primarily appeal to the female gender and they appeal to both women and girls. The products have been included in the survey as we ourselves have seen that they also appeal to girls below 15 years in a buying situation and as a few of this type of products are also found on the same shelf as other products for children.
Furthermore, a number of hair dyes (rinsing colours) has been bought. According to the shop assistant the hair dye series is addressed towards young girls as the products are rinsing colours which are washed out after a couple of hair washes so the girls can make experiments with the hair dyeing. On the packaging drawings of young girls with bare stomachs can be seen. Even if the hair dyes might also appeal to an older audience it is estimated that also girls below 15 years will buy these.
One cosmetics series is bought as there are drawings of Snoopy and strong colours – green or pink – on the packaging. It can be discussed whether a picture of Snoopy only appeals to adults and young persons or whether it also appeals to children (teenagers). Fact is that the cosmetics series was exhibited in the shop together with decorative cosmetics and hair slides etc. which also appeal to older children and the youngest teenagers.
Similarly, a skin care series is bought as these products are for teenagers according to the shop assistant. Thus the products also appeal to somewhat older persons than the target group of this project but also to the older part of the target group of this project.
Finally, a cosmetic series is bought despite that neither name nor design necessarily is directed towards children but because it is sold in a shop exclusively selling toys, children’s clothes and baby articles.
4.2 Database of cosmetic products for children
For the Danish Environmental Protection Agency a database in Microsoft Access 2000 is developed with in total 208 mapped cosmetic products for children. The purpose of the database of the cosmetic products for children is to create an overview of the products and their content of chemical substances in a relatively simple way. Furthermore it is possible in the database to sort the information and to extract information of the entered data criss-cross.
The database is built around a product overview where the following is recorded for each product:
- Content of chemical substances
- Various information about the product (type, place of purchase etc.)
Appendix A (chapter 1) is an overview of the content in the database.
In Table 4.2 is stated the information which is recorded in the database for all of the mapped cosmetic products for children.
Table 4.2: information about the cosmetic products recorded in the database
| Type information | Remarks |
| Product name | |
| Product type | The database operates with the following product types: Shampoo, balsam, body shampoo/bath gel, bobble bath, soap solid or liquid, bath confetti/bath caviar/fizzle salt, tooth paste, eau de toilette/perfume, deodorant, body lotion/cream, hair dyes (rinsing colours), hair styling products, massage oil, bath oil or other things |
| The chemical substances contained in the products including ranking order | The ranking order states the order of the substances in the list of constituents |
| Number of chemical substances (constituents) contained in the product in total | |
| Specification of how the product is procured | That means whether the product is bought or the declaration of contents is sent (or downloaded via the Internet) |
| Specification of where the product is bought or procured | The name of the convenience store chain and in which town |
| Specification whether the product is bought in a shop, at a hair dresser or via the Internet | |
| The name of the producer or importer | |
| When the product is bought or received | Stated by date. For products bought on the Internet the recorded date is the date when the products are ordered. Regarding declarations of contents the recorded date is the date where the declarations either are received or printed from the Internet |
| The bar code on the product | If available |
| Batch number | If available |
| Specifications of packaging material | That means a description of the type of packaging in question – for instance whether it is neutral or designed as a special figure |
| Description of the packaging material | For instance, whether the bottle is designed as Winnie the Pooh sitting in an umbrella. This description makes it much more easier to identify the products |
| Description of plastic type of the packaging via specification of plastic number if such a number is stated on the packaging | If the packaging is not made of plastic this field has no relevance |
| Whether the product is CE marked | |
| Description of the scent of the product, i.e. whether it contains perfume. | If perfume is stated in the declaration of contents it is only recorded as ”Perfume” in the database |
| Description whether the products are marketed directly to children | |
| Possible comments | For instance, whether products are sold in twin pack or notes about clarification of INCI names |
In the following a more detailed description with information about the registrations in the database of each of the above points is presented.
4.2.1 Chemical substances contained in the products
According to the Cosmetics Statutory Order (Stat. Ord. 422, 2006) the chemical substances that form a part of the cosmetic products for children have to be stated on the container with their INCI name that is a common nomenclature for use in declarations of contents of cosmetic products in the EU. An INCI name can cover several different chemical units (INCI, 2006).
Furthermore, the constituents shall be stated in order after descending weight. However, constituents in a concentration of less than 1% may be mentioned in a random order after the constituents whose concentration is higher than 1%. Hair dyes can be mentioned in a random order after the other constituents and scented and aromatic compounds can simply be named “perfume” and “aroma” respectively. Since March 2005 26 fragrances have to be stated in the declaration of contents when the concentration is higher than 0.001% in products which are not to be cleansed and 0.01 in products which are to be cleansed.
For products which with exception of the colours being alike the different colouring agents being used for the series might be stated with “+/-“ or “may contain”. This is seen for some of the mapped products. In these cases all colouring agents for the products are noted in the database even if the product with a particular colour might only contain one of the stated colouring agents. This means of course that the number of constituents as stated in the database for some products is larger than the real number.
Despite these exceptions the order in which the constituents are mentioned on the container is an indication of the quantity of the different constituents in the product.
Therefore, for each product information is entered into the database about the constituents which form a part of the products but also in which order (ranking) the constituents are listed on the product. Thus the ranking is an indication of the relative concentration of the constituents in the products. A low number (high ranking) expresses that the substance is a main constituent in the products whereas a high number (low ranking) indicates that the substance is an additive, for instance a preservative.
It shall be noted that in general it is not examined whether the products in fact contain the chemical substances that are stated on the products. It is possible that there are mistakes on the labels compared with the actual content which we cannot and have not taken into account in this project. The declaration of contents and the actual content were expected to be identical as a general rule.
The latest English edition of the EU INCI list (24.2.2006) containing about 7,600 constituents applied in cosmetic products is entered as a table in the database. In the first place, it makes the keying in of the constituents in the database much easier as in this way it is possible to select the constituents from a list defined in advance. In the second place, it means that the risk of mistakes when keying in is reduced as it is only possible to key in constituents which already are in this INCI list. Finally, it also means that it is possible to control whether all constituents can be found in the INCI list.
During the keying in it turned out that several products contained constituents that are not in the latest INCI list (dated 24.2.2006). In these cases the constituents in question are added to the EU INCI list of the database with the remark that they are added. In total 38 substances are added to the INCI list of the database. There might be some reasons for the fact that a substance is not in the INCI list dated 24.2.2006:
- The substance is not in the latest updating of the INCI list, i.e. application for use of this INCI name is in preparation or under consideration or the EU INCI list has not been updated..
- It is not the INCI name that is stated on the products as required and it has not been possible to find or guess the “correct” INCI name.
- There are spelling mistakes in the declaration of the product where it has not been possible to conclude the “correct” INCI name that should be stated on the packing. (All assumptions about spelling mistakes are noted).
4.2.2 Bar code
For all products with a printed bar code, this bar code has been registered. To print a bar code (EAN No.) on a product is voluntary. The bar code is a tool for registration of goods in connection with stock control and sales. The bar code is stated both by a number (the EAN number) and the very bar code. The two first digits state in which country the product is registered but not necessarily where it is produced. 57 is the code for Denmark. The next five digits are a marketing number for producer, importer or retail trade. Hereafter five digits follow. These are the internal item number of the marketing person and the last one is a control digit. The price of the products is not stated in the EAN number and bar code (the Labelling Guide, 1997)
4.2.3 Batch number
According to the Cosmetics Statutory Order cosmetic products must be labelled with the number of the production series or a reference specification, i.e. a batch number so that date and place of production can be identified (Stat. Ord. 422, 2006). The batch number for all the mapped products is registered in the database.
4.2.4 Specifications of packaging
In the project the packagings of the cosmetic products are divided into the following four categories:
1. Neutral
2. Colourful, no special design
3. Colourful, with pictures of figures, but no special design
4. Colourful, designed as a special figure
The Danish Safety Technology Authority has given an indicative statement of which products from category 4 being under the legislation for toys in order to be able to compare with the products which ought to have a CE labelling.
4.2.5 Description of plastic type of the packaging
For most of the products of plastic it is stated on the packaging which type of plastic the packaging is made of. This plastic type is stated in the database. The following parameters are used:
- 01 - PET
- 02 - HDPE
- 03 - PVC
- 04 - LDPE
- 05 - PP
- 06 - PS
- 07 - Others
- Not stated
- Not relevant
The parameter ”Not relevant” is used for products which for example is made of glass or metal. The parameter “Not stated” means that the packaging is made of plastic but the type is not stated or it has not been possible to read the stated type of plastic.
4.2.6 CE labelling
Some of the cosmetic products for children are designed as various figures, for example Winnie the Pooh, Barbie, Minnie Mouse, a mobile phone or simply a kind of bobble bath designed for instance as a frog. According to the Toys Statutory Order (see section 3.2.1 ”Safety requirements to toys”) products clearly designed or intended for playing purposes for children below 14 years must be CE labelled (Stat. Ord. 1116, 2003).
It is the Danish Safety Technology Authority who is responsible for and assesses whether a product comes under the definition of toys and thus must be CE labelled. In the database it is stated whether the mapped products are CE labelled or not.
4.2.7 Marketing of the products – directly towards children?
This point is added to the database to give a varied description of how the cosmetic products are marketed towards children. The reason is that during the survey it turned out that there is a number of grey area products where it might be discussed whether they are products being directly marketed towards children or they just are products appealing to children due to for instance the design or appearance of the products.
As described in chapter 2 ”Definition of cosmetic products for children”, cosmetic products for children in this project are defined both as products which are directly marketed to children, i.e. as an example they are called “Children’s”, and products which via their design or appearance appeal to children. However, in some cases, the latter is a matter of opinion and therefore this assessment has been made for each product and this is noted in the database.
4.2.8 Possible comments
In the database there is space to note possible comments about the product in question. The noted comments are for instance the following:
- Information about possible spelling mistakes in the INCI names of the declaration of contents as well as a note with an assumption of the INCI names which should have been stated.
- Information about the constituents that could not be found in the present INCI list and therefore is added to the INCI list of the database.
4.3 Results of the survey
In the following the data material which can be extracted from the database of the 208 purchased cosmetic products for children is presented.
4.3.1 Product name
As mentioned, in total 208 cosmetic products for children were bought. These cover all the products of cosmetic products for children which we could find in the survey period from mid-April 2006 to mid-June 2006. Intentionally, all product names are kept out of the report but the information is available at the Danish Environmental Protection Agency.
4.3.2 Importers/producers
The names of the importers and producers are intentionally taken away from the report but the information is available at the Danish Environmental Protection Agency.
4.3.3 How are the products procured?
Table 4.3 shows that the products are first and foremost procured via purchase. The major part of the products are bought (93%) – either via retail shops (81 %), hairdressers (1%) or the Internet (11%).
Table 4.3: Overview of the number of products in the survey
| Bought in retail shops | Bought at hairdressers | Bought via the Internet | Number of products in total | |
| Bought products | 169 | 3 | 22 | 194 (93%) |
| Products where the declaration of contents is sent from importer or procured via the Internet | 14 (7%) |
|||
| Number of products in total | 169 (81%) |
3 (1%) |
22 (11%) |
208 (100%) |
4.3.4 Product type
In Table 4.4 is stated the number of products of the different types which are bought and where they are bought (in shop, at hairdresser or via the Internet).
Table 4.4: Overview of the type of the cosmetic products for children
| Bought in shop | Bought at a hairdresser | Bought via the Internet | Number of products in total | |
| Bath confetti/bath caviar/fizzle salt | 29 | 29 | ||
| Bath oil | 3 | 5 | 8 | |
| Balsam | 2 | 1 | 2 | 5 |
| Body lotion/cream | 11 | 6 | 17 | |
| Body shampoo/bath gel | 42 | 1 | 43 | |
| Deodorant | 1 | 1 | ||
| Eau de toilette - perfume | 8 | 1 | 9 | |
| Hair dyes (rinsing colours) | 7 | 7 | ||
| Hair styling products | 2 | 2 | ||
| Massage oil | 1 | 1 | ||
| Shampoo | 17 | 2 | 7 | 26 |
| Bobble bath | 11 | 10 | 21 | |
| Soap – solid | 10 | 1 | 11 | |
| Soap – liquid | 1 | 1 | ||
| Tooth paste | 17 | 2 | 19 | |
| Others | 7 | 1 | 8 | |
| Number of products in total | 169 | 3 | 36 | 208 |
As it can be seen in Table 4.4 the database contains most products of the type body shampoo/bath gel, bath confetti/bath caviar/fizzle salt, shampoo and tooth paste but this illustrates quite well the reality as they are the most common products in the retail trade. The purchases have been incidental and therefore the database, also at this point, represents the Danish market of cosmetic products for children quite well.
The category ”Others” covers some undefinable product types such as Body Splash (body spray), preventive head lice shampoo or various cleansing products for the skin (primarily for the older children – teenagers). If we have not been able clearly to categorize the product under the other product types, “Others” is used.
4.3.5 Marketing of the products – directly towards children
As earlier described the majority of the mapped products is marketed directly towards children, i.e. they are for instance called something with “Kids” or “Children”, are designed as particular (cartoon) figures, have drawings of particular (cartoon) figures on the packaging, are on an Internet page under “Children’s products” or are sold in shops/Internet shops which exclusively sell things for children. This is the case for 146 of the 208 products in total.
The other 61 products consist of the products in the category “Bath confetti/caviar/fizzle salt” and other products which quite as much appeal to the somewhat older public (older children or adult women). The products are included in the survey because the products also appeal to especially the somewhat older girls because the shop assistant described for a part of the products that the products also were sold to the target group of this project and because some of the products also are found on the shelf together with other cosmetic products for children.
4.3.6 Chemical substances contained in the products
Appendix A contains a number of extractions from the database regarding the chemical substances being identified in the 208 mapped cosmetic products for children:
- Appendix A – chapter 2 is a list of all chemical substances being found in the mapped products. At first the list is stated after descending frequency and thereafter the same list is stated in alphabetical order.
- Appendix A – chapter 3 is a list of all chemical substances being identified for the individual types of products. The lists are only stated after descending frequency and there is only stated a list of the constituents for the most frequent types of producs, i.e.:
- Bath confetti/caviar/fizzle salt
- Balsam
- Bodylotion/cream
- Body shampoo/bath gel
- Shampoo
- Bobble bath
- Tooth paste
In total 461 different substances are found in the 208 cosmetic products for children which are recorded in the database. Furthermore, Appendix A (chapter 2) shows how often the different chemical substances are found and which average ranking they have. The ranking is an indication of the relative concentration of the constituents in the products. A low number (high ranking) expresses that the substance is the main constituent in the product whereas a high number (low ranking) indicates that the substance is an additive, for instance preservatives.
Appendix A (chapter 3) shows the chemical substances in the cosmetic products for children divided on the type of product. I.e. which substances are found in shampoo, tooth paste, hair styling products, perfume etc. At the same time Appendix A (chapter 3) shows which average ranking the substances form a part of for the mentioned product type, i.e. which average relative concentration the substances form a part of.
Table 4.5 below shows the 50 most abundant chemical substances in the mapped products. At the same time the table shows how many of the 208 products the substances form a part of as well as with which average ranking.
Table 4.5: the 50 most abundant constituents in the cosmetic products for children.
As it is seen from the table water and perfume are the two constituents which are most frequently used in the cosmetic products for children. Of the 26 fragrances mandatory to declare (see a detailed description in section 4.3.11 ”Content of perfume”) 13 of these are on the list of the 50 most frequently used substances in cosmetic products for children.
Of other frequently used constituents in the cosmetic products for children parabens (preservatives) can be mentioned. Listed below after declining frequency:
- Methylparaben (in 79 products)
- Propylparaben (in 70 products)
- Butylparaben (in 48 products)
- Ethylparaben (in 46 products)
- Isobutylparaben (in 39 products)
4.3.7 Constituents with hazard classification
Out of the 461 different chemical substances which form a part of the 208 cosmetic products for children 19 of these are classified as hazardous in pure form according to the List of dangerous substances (Stat. Ord. 923, 2005). In practice, this is checked via a data run where CAS numbers as stated on the INCI list are compared with CAS numbers from the List of dangerous substances. The result is seen below.
Table 4.6: Constituents in the cosmetic products for children which in pure form are classified as hazardous.
| INCI Name | Function (as stated in the INCI list) |
CAS no. | Classification | In number of products | Average ranking |
| PHENOXYETHANOL | preservative | 122-99-6 | Xn;R22 Xi;R36 | 50 | 9.2 |
| LIMONENE | 1 | 5989-27-5 | R10 Xi;R38 R43 N;R50/53 | 48 | 14.0 |
| ALCOHOL | solvent | 64-17-5 | F;R11 | 20 | 7.1 |
| BENZYL ALCOHOL | preservative / solvent | 100-51-6 | Xn;R20/22 | 20 | 16.3 |
| BENZYL BENZOATE | solvent | 120-51-4 | Xn;R22 | 17 | 11.3 |
| CITRAL | masking ² | 5392-40-5 | Xi;R38 R43 | 17 | 16.1 |
| SODIUM FLUORIDE | oral care ³/ antiplaque | 7681-49-4 | T;R25 R32 Xi;R36/38 | 11 | 10.1 |
| SODIUM HYDROXIDE | buffering / denaturant | 1310-73-2 | C;R35 | 8 | 13.0 |
| BUTANE | propellant | 106-97-8 | Fx;R124 | 7 | 24.1 |
| DIMETHYL ETHER | propellant / solvent | 115-10-6 | Fx;R12 | 7 | 4.0 |
| ISOBUTANE | propellant | 75-28-5 | Fx;R124 | 7 | 6.3 |
| PROPANE | propellant | 74-98-6 | Fx;R12 | 7 | 19.3 |
| 2-BROMO-2-NITROPROPANE-1,3-DIOL | preservative | 52-51-7 | Xn;R21/22 Xi;R37/38-41 N;R50 | 6 | 12.7 |
| ETHYLHEXYLGLYCERIN | skin conditioning | 70445-33-9 | Xi;R41 R52/53 | 3 | 12.0 |
| AMINOMETHYL PROPANOL | buffering | 124-68-5 | Xi;R36/38 R52/53 | 1 | 10.0 |
| CYCLOMETHICONE | antistatic / emollient / humectant / solvent / viscosity controlling / hair conditioning | 556-67-2 | Rep3;R62 R53 | 1 | 17.0 |
| HC VIOLET NO. 2 | hair dyeing | 104226-19-9 | R43 R52/53 | 1 | 7.0 |
| HEXYLENE GLYCOL | solvent / emulsifying / surfactant / skin conditioning | 107-41-5 | Xi;R36/38 | 1 | 8.0 |
| ZINC OXIDE | bulking / uv absorber / skin protecting | 1314-13-2 | N;R50/53 | 1 | 7.0 |
1 The function of Limonene is a solvent and aroma/fragrance
2 The function of Citral is also aroma/fragrance
3 Sodium fluoride strengthens against caries
4 Propelling gases as butane and isobutene are prohibited to use as propelling gas with Carc1 classification
4.3.8 Constituents in the guiding list for self-classification of the Danish Environmental Protection Agency
Of the in total 461 different chemical substances which form a part of the 208 cosmetic products for children, 38 of them are in the guiding list for self-classification of dangerous substances of the Danish Environmental Protection Agency (Environmental Project 635, 2001). The list is prepared as an offer to importers/producers on classification of chemical substances where no other reliable data are available. The list is prepared on basis of QSAR models. The result is seen below.
In practice, a database run is made where CAS numbers as stated in the INCI list are compared to CAS numbers from the guiding list for self-classification of dangerous substances of the Danish Environmental Protection Agency. The result is seen below.
Table 4.7: Constituents in cosmetic products for children which in pure form are in the guiding list for self-classification of dangerous substances of the Danish Environmental Protection Agency
4.3.9 Constituents with restrictions in relation to the statutory order
A search after which constituents being required restrictions in relation to the Cosmetics Statutory Order is made. The search has been conducted via the used EU INCI list of the identified constituents in the mapped products. 66 of the 461 constituents being found in the 208 mapped products have a restriction in relation to the Cosmetics Statutory Order. In Appendix A (chapter 4) is a list of the 66 constituents with the stated restrictions.
In the table below is shortly described what the individual restriction of the constituents means. Please note that the latest edition of the INCI list from the project start (from 24 February 2006) has been used. This means that among other things the latest changes with colouring agents and partly also the 26 fragrances are not in the INCI list in the database.
Table 4.8: Overview of the restrictions with which selected constituents in the products are marked in the Cosmetics Statutory Order.
| Restriction | Number of substances with the restriction | Related appendices | Explanation |
| III/1, x | 8 | Appendix 3, section 1: List of substances which are allowed to be used under the stated circumstances | X states the number for identification of the substance in the appendix. A maximum per cent of content of the substance is set. |
| IV/1 | 35 | Appendix 4: List of permitted colouring agents | 1 means that the colouring agent is permitted in all cosmetic products |
| VI/1,x | 23 | Appendix 5: List of permitted preservatives | 1,x states the number for identification of the substance in the appendix. A maximum per cent of content of the substances is set as well as possible limitations and requirements |
| VII/1,x | 1 | Appendix 6: List of permitted UV absorbers | 1,x states the number for identification of the substance in the appendix. A maximum per cent of content of the substances is set as well as possible limitations and requirements |
| Sum | 67 |
Please note that one of the substances (benzyl alcohol) has both a III/1,x restriction and a VI/1,x restriction and therefore the sum here is 67 constituents.
It is primarily for colouring agents and preservatives where restrictions in relation to the Cosmetics Statutory Order are found. The restrictions are stated as maximum allowed concentrations and possible limitations, such as “only to be used in products to be washed off”.
Please note that the 26 fragrances to be declared separately also have restriction via appendix 3 section 1 (III/1) but not all of these fragrances appeared from the used INCI list and, if above a certain content, the latest restriction on separated declaration has not been stated in the used INCI list. Only benzyl alcohol is mentioned with restriction but benzyl alcohol is also a preservative. Therefore, fragrances are not a part of the above in general.
4.3.10 Specification of number of constituents
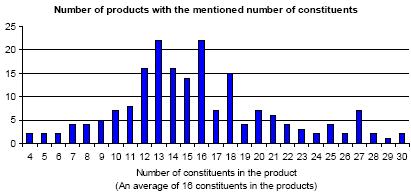
Based on the declaration of content on the products it is stated how many constituents each product contains. The number of constituents varies from 3 to 36 different chemical substances. In Figure 4.1 a distribution of the number of constituents in the mapped products is seen. The products contain 16 constituents (16.2) on average.
For products being alike except for the colour the different colouring agents being used in the series can be marked with “+/-“ or “may contain” in the declaration of content. This is seen for some of the mapped products. In these cases, all colouring agents of the product are noted in the database even if the product with a particular colour might only contain one of the stated colouring agents. This means that the number of constituents as stated in the database for certain products is larger than the real number.
Figure 4.1: Distribution of the number of constituents in the mapped products.
4.3.11 Content of perfume
There is a declared content of perfume in 153 of the 208 products. This means that there is perfume in 74% of the mapped cosmetic products for children. The product type tooth paste (in total 19 products) has no products with a declared content of perfume and furthermore 11 products lack the declaration of content for some reason. If these products are left out there is a declared content of perfume in 86% of the mapped products.
Table 4.9: Overview of products with perfume divided on the type of the cosmetic products for children
| Number of products in total | Number of products with content of perfume Stated in total and % |
Number of products with content of one or more of the 26 fragrances | |||
| Bath confetti/caviar/fizzle salt | 29 | 28 | (97%) | 16 | (55%) |
| Bath oil | 8 | 3 | (38%) | 2 | (25%) |
| Balsam | 5 | 2 | (40%) | 1 | (20%) |
| Body lotion/cream | 17 | 13 | (76%) | 6 | (35%) |
| Body shampoo/bath gel | 43 | 37 | (86%) | 7 | (16%) |
| Deodorant | 1 | 1 | (100%) | 0 | (0%) |
| Eau de toilette - perfume | 9 | 9 | (100%) | 6 | (67%) |
| Hair dyes (rinsing colour) | 7 | 7 | (100%) | 7 | (100%) |
| Hairstyling products | 2 | 2 | (100%) | 0 | (0%) |
| Massage oil | 1 | 1 | (100%) | 1 | (100%) |
| Shampoo | 26 | 17 | (65%) | 4 | (15%) |
| Boblle bath | 21 | 14 | (63%) | 4 | (19%) |
| Soap – solid | 11 | 11 | (100%) | 3 | (27%) |
| Soap – liquid | 1 | 1 | (100%) | 1 | (100%) |
| Tooth paste | 19 | 0 | (0%) | 9 | (47%) |
| Others | 8 | 7 | (88%) | 4 | (50%) |
| Number of products in total | 208 | 153 | (74%) | 71 | (34%) |
Table 4.9 states how many of the products within the individual product type that contain perfume. As seen all deodorants, eau de toilette, hair dyes (rinsing colours), hairstyling products and massage oils (only one product) contain perfume whereas none of the tooth paste products has a declared content of perfume.
4.3.11.1 The 26 fragrances mandatory to declare
According to the Cosmetics Statutory Order (Stat. Ord. §25, 2006) all scented compounds are to be listed via the definition “perfume”, “perfum” or “aroma” only. According to appendix 3 of the statutory order, 26 fragrances must be stated in the declaration of content regardless of their function in the product when the concentration is higher than 0.001% in products which are not to be cleansed and 0.01% in products which are to be cleansed. This order with special definition of the fragrances mandatory to declare became effective in 2005 and applies for all cosmetics which are produced after 10 March 2005.
The 26 fragrances mandatory to declare can be seen in the table below. In the table is also stated which of these are found in the cosmetic products for children and the number of products. The most frequently applied fragrances are in the top of the table. The first 13 substances mandatory to declare are in the list of the 50 most frequently applied constituents in cosmetic products for children as stated in Table 4.5.
22 of the 26 fragrances mandatory to declare are found in the mapped products. The product group with the most frequent occurrence of these fragrances is “Bath confetti/caviar/fizzle salt”. It is seen that in general many of the scented products have not a content of the 26 fragrances. The reason may be that these fragrances are not used much or that the producers do not meet the regulation that these fragrances must be declared separately, but this is unknown. This will be considered when analyzing the products.
In total one or more of the 26 fragrances mandatory to declare are found in 71 different products corresponding to 34% of the mapped products.
Table 4.10: Occurrence of the 26 fragrances mandatory to declare in cosmetic products for children
| The 26 fragrances mandatory to declare (stated with their INCI name) | Number of products in which they occur (out of 208) | Type of product in which the fragrance occurs |
| Limonene | 48 | 6 Bath confetti/caviar/fizzle salt 2 Bath oils 1 Balsam 5 Body lotion/cream 4 Body shampoo/bath gel 3 Eau de toilette – perfume 7 Hair dyes (rinsing colours) 1 Massage oil 2 Shampoo 3 Bobble bath 1 Soap – solid 1 Soap – fliquid 9 Tooth paste 3 Other products (skin care products) |
| Linalool | 45 | 13 Bath confetti/caviar/fizzle salt 2 Bath oils 4 Body lotion/cream 1 Body shampoo/bath gel 5 Eau de toilette – perfume 7 Hair dyes (rinsing colours) 1 Massage oil 1 Shampoo 2 Bobble bath 3 Soap – solid 1 Soap – liquid 1 Tooth paste 4 Other products (skin care products, body splash) |
| Geraniol | 25 | 12 Bath confetti/caviar/fizzle salt 2 Bath oils 2 Body lotion/cream 6 Eau de toilette – perfume 1 Massage oil 1 Soap – solid 1 Soap – liquid |
| Hexyl Cinnamal | 21 | 6 Bath confetti/caviar/fizzle salt 1 Body lotion/cream 4 Body shampoo/bath gel 1 Eau de toilette – perfume 7 Hair dyes (rinsing colours) 2 Soap – solid |
| Citronellol | 22 | 12 Bath confetti/caviar/fizzle salt 1 Balsam 1 Body lotion/cream 4 Eau de toilette - perfume 1 Massage oil 3 Soap – solid |
| Benzyl Salicylate | 20 | 12 Bath confetti/caviar/fizzle salt 1 Balsam 2 Body lotion/cream 3 Eau de toilette – perfume 1 Shampoo 1 Other product (body splash) |
| Benzyl Alcohol | 20 | 6 Bath confetti/caviar/fizzle salt 1 Body lotion/cream 1 Body shampoo/bath gel 1 Eau de toilette - perfume 7 Hair dyes (rinsing colours) 2 Shampoo 1 Tooth paste 1 Other product (body splash) |
| Benzyl benzoate | 19 | 12 Bath confetti/caviar/fizzle salt 1 Balsam 2 Body lotion/cream 1 Body shampoo/bath gel 2 Eau de toilette – perfume 1 Shampoo |
| Amyl Cinnamal | 17 | 6 Bath confetti/caviar/fizzle salt 1 Balsam 1 Body lotion/cream 7 Hair dyes (rinsing colours) 2 Soap – solid |
| Citral | 17 | 8 Bath confetti/caviar/fizzle salt 2 Body lotion/cream 2 Eau de toilette – perfume 1 Massage oil 1 Soap - liquid 3 Other products (skin care products) |
| Butylphenyl Methylpropional | 16 | 10 Bath confetti/caviar/fizzle salt 3 Eau de toilette - perfume 1 Bobble bath 2 Soap – solid |
| Eugenol | 15 | 12 Bath confetti/caviar/fizzle salt 1 Eau de toilette – perfume 2 Tooth paste |
| Cinnamyl Alcohol | 14 | 14 Bath confetti/caviar/fizzle salt |
| Hydroxycitronellal | 13 | 6 Bath confetti/caviar/fizzle salt 1 Balsam 1 Body lotion/cream 2 Eau de toilette – perfume 1 Shampoo 2 Soap – solid |
| a-Isomethyl Ionone | 12 | 6 Bath confetti/caviar/fizzle salt 1 Body lotion/cream 2 Soap – solid 3 Eau de toilette – perfume |
| Hydroxyisohexyl 3-Cyclohexene Carboxaldehyde | 12 | 7 Bath confetti/caviar/fizzle salt 4 Eau de toilette - perfume 1 Soap – liquid |
| Coumarin | 10 | 6 Bath confetti/caviar/fizzle salt 1 Body lotion/cream 3 Eau de toilette – perfume |
| Amylcinnamyl Alcohol | 6 | 6 Bath confetti/caviar/fizzle salt |
| Benzyl Cinnamate | 6 | 6 Bath confetti/caviar/fizzle salt |
| Farnesol | 6 | 6 Bath confetti/caviar/fizzle salt |
| Cinnamal | 2 | 2 Tooth paste |
| Isoeugenol | 1 | 1 Body lotion/cream |
| Anise Alcohol | 0 | |
| Evernia Prunastri (Oakmoss) Extract | 0 | |
| Evernia Furfuracea (Treemoss) Extract | 0 | |
| Methyl 2-Octynoate | 0 |
With regard to tooth paste there is the special condition that 9 of the 19 mapped tooth pastes contain between 1 and 3 of the 26 fragrances that must be declared according to the Cosmetics Statutory Order even if “perfume” is not declared on the products.
From Table 4.11 it can be seen that the limonene is the fragrance which is in all 9 tooth pastes. The explanation for this is probably that limonene is a fragrance from citrus fruits and is added to give the tooth paste a special taste. Correspondingly, linalool comes from oranges and is probably added to give taste. Eugenol and cinnamal come from clove oil and cinnamon oil respectively and have probably been added with the same purpose.
Furthermore, one of the products contains benzyl alcohol which besides being a fragrance is a preservative too. It might be due to this function that the substance is added.
Table 4.11: Content of fragrances in tooth paste that must be declared
| ID no. | Fragrances contained (INCI name) |
| 39 | LIMONENE |
| 54 | LINALOOL LIMONENE |
| 60 | LIMONENE |
| 61 | LIMONENE |
| 63 | CINNAMAL LIMONENE EUGENOL |
| 64 | LIMONENE |
| 120 | EUGENOL LIMONENE CINNAMAL |
| 121 | BENZYL ALCOHOL LIMONENE |
| 180 | LIMONENE |
4.3.12 Content of preservatives
132 of the 208 products contain a preservative (where the function preservative is stated via the INCI list). In total, 23 different preservatives are found in the 208 mapped products. These are listed in the table below after frequency.
Table 4.12: Appliedd preservatives in cosmetic products for children
| INCI Name | Occurrence in number of products | Remarks | Maximum permissible concentration1 |
| METHYLPARABEN | 79 | 0.4% (acid) for one ester 0.8% (acid) for ester mixtures |
|
| PROPYLPARABEN | 70 | Under evaluation for hormone-disrupting properties | 0.4% (acid) for one ester 0.8% (acid) for ester mixtures |
| PHENOXYETHANOL | 50 | 1% | |
| BUTYLPARABEN | 48 | Under evaluation for hormone-disrupting properties | 0.4% (acid) for one ester 0.8% (acid) for ester mixtures |
| ETHYLPARABEN | 46 | 0.4% (acid) for one ester 0.8% (acid) for ester mixtures |
|
| ISOBUTYLPARABEN | 39 | Under evaluation for hormone-disrupting properties | 0.4% (acid) for one ester 0.8% (acid) for ester mixtures |
| BENZYL ALCOHOL | 20 | Allergenic (one of the 26 fragrances) | 1% |
| METHYLCHLORO-ISOTHIAZOLINONE² | 15 | Allergenic (according to self-classification of the Danish EPA) | 0.0015% of a mixture in the ratio 3:1 of methylchloroisothiazolinone and methylisothiazolinone |
| METHYLISOTHIAZOLINONE² | 15 | Allergenic (according to self-classification of the Danish EPA) | 0.0015% of a mixture in the ratio 3:1 of methylchloroisothiazolinone and methylisothiazolinone |
| 5-BROMO-5-NITRO-1,3-DIOXANE | 15 | Allergenic (according to self-classification of the Danish EPA) Separates formaldehyde |
0.1% Only to be used in products which are to be cleansed after apply |
| SODIUM BENZOATE | 13 | 0.5% (like acid) | |
| DMDM HYDANTOIN | 11 | Allergenic (according to self-classification of the Danish EPA) Separates formaldehyde |
0.6% |
| CETRIMONIUM CHLORIDE | 9 | 0.1% | |
| IMIDAZOLIDINYL UREA | 7 | Allergenic (according to self-classification of the Danish EPA) Separates formaldehyde |
0.6% |
| BEHENTRIMONIUM CHLORIDE | 7 | 0.1% | |
| 2-BROMO-2-NITROPROPANE-1,3-DIOL | 6 | Potentially allergenic. Separates formaledehyde | 0.1% |
| DIAZOLIDINYL UREA | 4 | Potentially allergenic. Separates formaldehyde | 0.5% |
| ISOPROPYLPARABEN | 4 | Under evaluation for hormone-disrupting properties. Allergenic (according to self-classification of the Danish EPA) |
0.4% (acid) for one ester 0.8% (acid) for ester mixtures |
| QUATERNIUM-15 | 2 | Potentially allergenic. Separates formaldehyde | 0.2% |
| LAURTRIMONIUM CHLORIDE | 2 | 0.1% | |
| IODOPROPYNYL BUTYLCARBAMATE | 1 | Allergenic | 0.05% Must not be used in products for oral hygiene or for the lips. If the concentration in products which are not to be cleansed of the skin is higher than 0.02%, then “contains” iodine” must be stated |
| POTASSIUM SORBATE | 1 | Allergenic (according to self-classification of the Danish EPA) |
0.6% (like acid) |
| SODIUM METHYLPARABEN | 1 | 0.4% (acid) for one ester 0.8% (acid) for ester mixtures |
1 According to Appendix 5 in the Cosmetics Statutory Order (Stat. Ord. 422, 2006).
² The mixture of methylisothiazolinone and methylchloroisothiazolinone is also called Kathon.
The allergenic preservative MG (methyldibromo glutaronitrile) being mentioned much during the last couple of years is not found in any of the mapped products.
4.3.12.1 Preservatives with application limitations
In general, there are concentration limitations on all the applied preservatives. Furthermore, two of the preservatives have application limitations.
5-bromo-5-nitro-1,3-dioxane must only be applied in products which are to be cleansed of after apply. This preservative is found in totally 15 products and all these products are body shampoo/bath gel or bobble bath; that means products being cleansed of after apply.
Iodopropynyl butylcarbamate must not be used for oral hygiene or for the lips and if the concentration is higher than a certain level for products not being cleansed of after apply it must be stated on the product that it contains iodine. Iodopropynyl butylcarbamate is only in one product which is a balsam; that means a product being cleansed of after apply.
4.3.13 No content of triclosan
None of the 208 mapped products contains triclosan.
4.3.14 Content of colouring agents
119 of the 208 products contain a colouring agent (where the function hair dyeing or cosmetic colorant is stated via the INCI list). In total 45 different colouring agents are found in the 208 mapped products. These are listed in the table below after frequency.
Table 4.13: Applied colouring agents in cosmetic products for children
| INCI Name | Function (as stated in the INCI list) | Occurrence in number of products | Remarks |
| CI 42090 | cosmetic colorant | 62 | |
| CI 17200 | cosmetic colorant | 50 | |
| CI 19140 | cosmetic colorant | 40 | |
| CI 16255 | cosmetic colorant | 16 | |
| CI 16035 | cosmetic colorant | 14 | |
| CI 14700 | cosmetic colorant | 12 | |
| CI 77891 | cosmetic colorant | 10 | |
| CI 73360 | cosmetic colorant | 7 | |
| CI 47005 | cosmetic colorant | 6 | |
| CI 77007 | cosmetic colorant | 6 | |
| HC RED NO. 3 | hair dyeing | 6 | Allergenic (according to self-classification of the Danish EPA) |
| 3-NITRO-p-HYDROXYETHYLAMINOPHENOL | hair dyeing | 5 | |
| CI 12085 | cosmetic colorant | 5 | |
| CI 15985 | cosmetic colorant | 5 | |
| CI 16185 | cosmetic colorant | 5 | |
| CI 59040 | cosmetic colorant | 5 | |
| CI 61570 | cosmetic colorant | 5 | |
| CI 75810 | cosmetic colorant | 5 | |
| 4-AMINO-3-NITROPHENOL | hair dyeing | 4 | |
| CI 45100 | cosmetic colorant | 4 | |
| CI 45380 | cosmetic colorant | 4 | |
| CI 45430 | cosmetic colorant | 4 | |
| BASIC BLUE 99 | hair dyeing | 3 | |
| CI 77491 | cosmetic colorant | 3 | |
| CI 77499 | cosmetic colorant | 3 | |
| HC BLUE NO. 2 | hair dyeing | 3 | Allergenic (according to self-classification of the Danish EPA) |
| HC ORANGE NO. 2 | hair dyeing | 3 | |
| HC YELLOW NO. 9 | hair dyeing | 3 | |
| 2-NITRO-5-GLYCERYL METHYLANILINE | hair dyeing | 2 | |
| BETA VULGARIS EXTRACT | cosmetic colorant | 2 | |
| CI 15510 | cosmetic colorant | 2 | |
| CI 45410 | cosmetic colorant | 2 | |
| CI 74160 | cosmetic colorant | 2 | |
| CI 75120 | cosmetic colorant | 2 | |
| CI 77289 | cosmetic colorant | 2 | |
| CI 77492 | cosmetic colorant | 2 | |
| HYDROXYANTHRAQ UINONEAMINOPROPYL METHYL MORPHOLINIUM METHOSULFATE | hair dyeing | 2 | |
| BASIC YELLOW 57 | hair dyeing | 1 | |
| CI 42051 | cosmetic colorant | 1 | |
| CI 42053 | cosmetic colorant | 1 | |
| CI 45350 | cosmetic colorant | 1 | |
| CI 74260 | cosmetic colorant | 1 | |
| CI 75470 | cosmetic colorant | 1 | |
| HC RED NO. 7 | hair dyeing | 1 | Allergenic (according to self-classification of the Danish EPA) |
| HC VIOLET NO. 2 | hair dyeing | 1 | Allergi-causing (LOFS R43) |
The EU Commission bans 22 substances in hair dyes as per 1 December 2006[3]. None of these 22 colouring agents is found in the mapped cosmetic products for children.
4.3.14.1 Colouring agents with application limitations
According to Appendix 4 in the Cosmetics Statutory Order (List of colouring agents allowed in cosmetic products), all the CI colouring agents in the list are permitted in all cosmetic products (application area 1). However, there are limitations on maximum permissible concentration in the end product for a number of the colouring agents.
According to Appendix 4 in the Cosmetics Statutory Order (List of colouring agents permitted in cosmetic products), CI 12085 must as a maximum occur in a concentration of 3% in the end product.
According to Appendix 4 in the Cosmetics Statutory Order (List of colouring agents permitted in cosmetic products), CI 45350 must as a maximum occur in a concentration of 6% in the end product.
According to Appendix 4 in the Cosmetics Statutory Order (List of colouring agents permitted in cosmetic products), CI 45380 must as a maximum occur in a concentration of 1% and 2% in fluorescein and monobromfluorescein respectively.
According to Appendix 4 in the Cosmetics Statutory Order (List of colouring agents permitted in cosmetic products), CI 45430 must as a maximum occur in a concentration of 1% and 3% in fluorescein and monobromfluorescein respectively.
According to Appendix 4 in the Cosmetics Statutory Order (List of colouring agents permitted in cosmetic products), CI 77289 must be free of chromate ion.
HC Blue No. 2 (CAS no. 33229-34-4) is in Appendix 3 section 2 in the Cosmetics Statutory Order which is a list of temporarily permitted substances in cosmetic products. It is permitted until 31 December 2007[4] in a maximum concentration of 2.8% (w/w).
HC Orange No. 2 (CAS no. 85765-48-6) is also in Appendix 3 section 2 – List of temporarily permitted substances in cosmetic products. It is permitted until 31 December 2007[5] in a maximum concentration of 1.0% (w/w).
Correspondingly, HC Violet No. 2 (CAS-nr. 104226-19-9) is in Appendix 3 section 2 – List of temporarily permitted substances in cosmetic products. It is permitted until 31 December 2007[6] in a maximum concentration of 2.0% (w/w).
4-amino-3-nitrophenol (CAS no. 610-81-1) is in Appendix 3 section 2 – List of temporarily permitted substances in cosmetic products. It is permitted until 31 December 2007[7] in a maximum concentration of 3.0% (w/w).
Finally, 3-nitro-p-hydroxyethylaminophenol (CAS no. 65235-31-6) is in Appendix 3 section 2 – List of temporarily permitted substances in cosmetic products. It is permitted until 31 December 2007[8] in a maximum concentration of 6.0% (w/w).
4.3.15 Design of packaging and CE labelling
Some of the cosmetic products for children are designed as various figures, for instance Winnie the Pooh, Barbie, Minnie Mouse, a mobile phone and some small bobble bath products in soft plastic designed as animals and things. According to the Toys Statutory Order (see section 3.2.1 ”Safety requirements to toys”), products ”which clearly are designed or determined for play purposes for children below 14 years” must be CE labelled (Stat. Ord. 1116, 2003).
Table 4.14 states the distribution of the number of products on the stated packaging types.
Table 4.14: Distribution of the number of products on the stated packaging types
| Packaging type | Number of products | |
| 1 | Neutral | 34 |
| 2 | Colourful, no special design | 68 |
| 3 | Colourful, with pictures of figures, but no special design | 50 |
| 4 | Colourful, designed as a special figure | 56 |
| In total | 208 |
Thus, 27% of the mapped products are designed as a particular figure.
It is a matter of interpretation whether for instance a bobble bath product designed as a heart “clearly” is intended for playing purposes whereas it is more certain that a bobble bath product designed as for instance a cow or a Barbie figure will be seen as a toy from a child’s point of view.
Therefore, products from category 4 have been sent to the Danish Safety Technology Authority who is the competent authority in this area. The Danish Safety Technology Authority has given an indicative statement where 19 of the 56 products in category 4 are assessed to be comprised by the rules for toys.
4.3.16 Plastic type of the packaging
f the in total 208 products in the database 8 products are stated to be of PVC. The figures are three Barbie products, four Disney products and one shampoo with a preventive head lice shampoo. Of all products, 120 products have not stated the plastic type. They are primarily products like tooth paste and bobble bath products designed as various figures (for instance a frog, a cow). Common feature of the majority of these products is that the packaging is made of soft plastic.
4.3.17 Bar code
For all products with a printed bar code, this bar code has been registered. In total 175 of the 208 mapped products has a bar code. To print a bar code (EAN No.) on a product is voluntary. The two first digits state in which country the product is registered but not necessarily where it is produced. 57 is the code for Denmark (27 of the products are registered with the bar code starting with 57).
4.3.18 Batch number
According to the Cosmetics Statutory Order cosmetic products must be labelled with the number of the production series or a reference specification, i.e. a batch number so that date and place of production can be identified (Stat. Ord. 422, 2006). In total, 184 of the 208 mapped products have a batch number. For in total 14 of the mapped products the producer/importer has sent the declaration of content or we have found the declaration of content on the Internet and for these product which we have not had in our hand physically, the batch number is unknown and therefore it has not been entered into the database. The remaining 10 products have no information about declaration of content and other general information including batch number (for eight products), and for two products the batch number itself is missing.
5 Analysis results
- 5.1 Selection of products for analysis
- 5.2 Analyses for selected constituents
- 5.3 Analyses of selected packaging materials
The purpose of this project was to study whether cosmetic products for children contain problematic chemical substances but also to study whether the packaging contains problematic substances.
Based on the survey and the provided database of the constituents of the cosmetic products for children, it was decided in consultancy with the Danish Environmental Protection Agency to focus on the 26 fragrances mandatory to declare and the allergenic preservative Kathon (it is a mixture of the two substances methylchloroisothiazolinone and methylisothiazolinone in the ratio 3:1).
The reason for this is that a very large part of the mapped products has a declared content of perfume – in total 74% and one third of all the products has a declared content of one or more of the 26 fragrances mandatory to declare.
Furthermore, Kathon was chosen as it is assessed to be allergenic (R43 – Might cause allergy via contact with the skin) according to the guiding list for self-classification of hazardous substances of the Danish Environmental Protection Agency (Environmental project 635, 2001). Kathon is not classified as allergenic according to the List of hazardous substances but at the beginning of the eighties when Kathon was introduced as a preservative in cosmetics it gave cause for an epidemic of allergy cases in Europe. Kathon is now applied in small quantities in cosmetics (0.0003 – 0.0015%) and gives nevertheless cause for many allergy cases (Denmark’s National Allergy Research Centre, 2006a).
With regard to the packaging of the products focus has been on the content of heavy metals and phthalates. To identify packaging materials of PVC a quick and cheap analysis for the content of chlorine is carried out. A content of chlorine might be an indication of the packaging being made of PVC and thus possibly containing phthalates. Then the packaging materials of PVC have been selected for a quantitative analysis for the content of phthalates.
In the table below the analysis programme of the project is presented. Analyses for constituents are made by the company Eurofins and analyses of the packaging are made by FORCE Technology, the Department of Chemical Analysis.
Table 5.1: Analysis programme
| Analyses for constituents | Analyses of packaging |
| 17 products are quantitatively analyzed for the 26 fragrances 11 products are quantitatively analyzed for Kathon |
10 packaging materials are quantitatively analyzed for heavy metals 14 packaging materials are quantitatively examined for content of chlorine (PVC) 10 packaging materials are quantitatively analyzed for content of phthalates |
5.1 Selection of products for analysis
5.1.1 Analysis for the 26 fragrances
The 17 products selected for an analysis for the 26 fragrances are stated in the table below. In the selection the emphasis has been on the more common product types, that means body shampoo/bath gel, shampoo and bobble bath but a few other product types have also been selected.
The selected products for the analysis are primarily products which already had a declared content of perfume. However, one product is selected because it smelled of perfume but no content of perfume was declared. Furthermore, some products not having any separate declaration of the 26 fragrances are selected. The purpose of these analyses is to check whether the new regulation regarding separate declaration of these substances is introduced at the producers. Theoretically, there might still be legal products on the shelves in the shops without a separate declaration of the 26 fragrances as the regulation applies to all cosmetics being produced after 10 March 2005. In the project period we ourselves saw that the circumstance is so for some products as products which were initially bought for the survey had changed declaration when we bought the products again for the analyses. Now a content of the 26 fragrances was declared separately.
Furthermore, the products are selected so they cover several producers and represent both retail trade products and products via the Internet; however, with a substantial majority of retail trade products.
Table 5.2: Product selected for analysis for content of the 26 fragrances.
| ID no. | Product type | Design of the product |
| 5 | Body shampoo/bath gel | 4 – Colourful, designed as a special figure |
| 9 | Soap – liquid | 1 – Neutral |
| 21 | Bobble bath | 4 – Colourful, designed as a special figure |
| 22 | Body shampoo/bath gel | 4 – Colourful, designed as a special figure |
| 32 | Body shampoo/bath gel | 4 – Colourful, designed as a special figure |
| 33 | Body shampoo/bath gel | 4 – Colourful, designed as a special figure |
| 34 | Body shampoo/bath gel | 4 – Colourful, designed as a special figure |
| 35 | Body shampoo/bath gel | 4 – Colourful, designed as a special figure |
| 49 | Shampoo | 1 – Neutral |
| 69 | Shampoo | 2 - Colourful, but no special design |
| 82 | Bobble bath | 3 – Colourful, with drawings of figures, but no special design |
| 98 | Bath confetti/caviar/fizzle salt | 1 – Neutral |
| 166 | Body lotion/cream | 3 – Colourful, with drawings of figures, but no special design |
| 170 | Body shampoo/bath gel | 3 – Colourful, with drawings of figures, but no special design |
| 191 | Shampoo | 4 – Colourful, designed as a special figure |
| 206 | Shampoo | 3 – Colourful, with drawings of figures, but no special design |
| 209 | Eau de toilette - perfume | 3 – Colourful, with drawings of figures, but no special design |
5.1.2 Analysis for Kathon
In total, 15 products with a content of Kathon (a combination of methylchloroisothiazolinone and methylisothiazolinone) are found in the survey. Eight of these products were selected for a quantitative analysis for Kathon. These are stated in the table below. Two series of products contain Kathon – one series of five products and one series of two products. From these two series only one product from each series has been selected.
The remaining six products for a Kathon analysis were selected randomly (out of the remaining eight possible products). The majority of the products with a contant of Kathon is non-stay-on products. One of the two stay-on products is selected for analysis (as the two stay-on products are from the same series).
Due to a mistake products without a declared content of Kathon were also analyzed quantitatively for a content of Kathon. It proved that three products had a content of kathon despite the fact that it is not declared on the product. The results from these three products are also presented in the following. All in all, the following 11 products were analyzed for a content of Kathon.
Table 5.3: Selected for analysis for content of Kathon
| ID no. | Product type | Design of the product |
| 5 | Body shampoo/bath gel | 4 – Colourful, designed as a special figure |
| 9 | Soap – liquid | 1 – Neutral |
| 19 | Body shampoo/bath gel | 3 – Colourful, with drawings of figures, but no special design |
| 21 | Bobble bath | 4 – Colourful, designed as a special figure |
| 33 | Body shampoo/bath gel | 4 – Colourful, designed as a special figure |
| 34 | Body shampoo/bath gel | 4 – Colourful, designed as a special figure |
| 35 | Body shampoo/bath gel | 4 – Colourful, designed as a special figure |
| 82 | Bobble bath | 3 – Colourful, with drawings of figures, but no special design |
| 170 | Body shampoo/bath gel | 3 – Colourful, with drawings of figures, but no special design |
| 193 | Body lotion/cream | 4 – Colourful, designed as a special figure |
| 206 | Shampoo | 3 – Colourful, with drawings of figures, but no special design |
5.1.3 Analysis for heavy metals in the packaging
The 10 products selected for analysis of heavy metals in the packaging are stated in the table below. In the selection emphasis was on strongly colourful products as these products most probably contain the largest quantities of heavy metals. Furthermore, the emphasis is on the more common product types, i.e. body shampoo/bath gel, shampoo and bobble bath but the product types body lotion and hairstyling product are also selected due to strongly colourful packaging materials.
Table 5.4: Selected for analysis for content of heavy metals in the packaging
| ID no. | Product type | Design of the product |
| 21 | Bobble bath | 4 – Colourful, designed as a special figure |
| 27 | Body shampoo/bath gel | 4 – Colourful, designed as a special figure |
| 32 | Body shampoo/bath gel | 4 – Colourful, designed as a special figure |
| 40 | Hairstyling product | 3 – Colourful, with drawings of figures, but no special design |
| 74 | Shampoo | 2 – Colourful, no special design |
| 166 | Body lotion/cream | 3 – Colourful, with drawings of figures, but no special design |
| 184 | Body lotion/cream | 2 – Colourful, no special design |
| 193 | Body lotion/cream | 4 – Colourful, designed as a special figure |
| 206 | Shampoo | 3 – Colourful, with drawings of figures, but no special design |
| 207 | Bobble bath | 4 – Colourful, designed as a special figure |
In the selection it was also emphasized that a part of the products had to be designed as a particular figure as these products might be used as toys (for instance in the bath) and especially small children (or small siblings) might be expected to put them into the mouth. Therefore, it is relevant to examine the quantity of heavy metals to be able to assess the impact in such situations.
Furthermore, the products have been selected so they cover several producers and so they represent both the retail trade products and products via the Internet, however, with a substantial majority of retail trade products.
5.1.4 Analysis for chlorine and phthalates in the packaging
The 14 products selected for analysis of chlorine in the packaging are stated in the table below. In the selection emphasis was exclusively on products of soft plastic as these products are probably made of PVC and thus contain chlorine. Furthermore, the emphasis is on the most common product types, i.e. body shampoo/bath gel, shampoo and bobble bath.
In the selection it is also emphasized that a part of the products was designed as a particular figure (more than half) as these products might be used as toys (for instance in the bath) and especially small children (or small siblings) might be expected to put them into the mouth. Therefore, it is relevant to examine for a possible content of PVC and later for a content of phthalates to be able to assess the impact in such situations.
Furthermore, the products have been selected so they cover several producers and so they represent both the retail trade products and products via the Internet, however, with a substantial majority of retail trade products.
Table 5.5: Selected for analysis for content of chlorine in the packaging
| ID no. | Product type | Design og the product |
| 5 | Body shampoo/bath gel | 4 – Colourful, designed as a special figure |
| 18 | Body shampoo/bath gel | 4 – Colourful, designed as a special figure |
| 33 | Body shampoo/bath gel | 4 – Colourful, designed as a special figure |
| 34 | Body shampoo/bath gel | 4 – Colourful, designed as a special figure |
| 35 | Body shampoo/bath gel | 4 – Colourful, designed as a special figure |
| 40 | Hairstyling product | 3 – Colourful, with drawings of figures, but no special design |
| 82 | Bobble bath | 3 – Colourful, with drawings of figures, but no special design |
| 128 | Body shampoo/bath gel | 4 – Colourful, designed as a special figure |
| 193 | Body lotion/cream | 4 – Colourful, designed as a special figure |
| 194 | Body shampoo/bath gel | 4 – Colourful, designed as a special figure |
| 196 | Body shampoo/bath gel | 4 – Colourful, designed as a special figure |
| 206 | Shampoo | 3 – Colourful, with drawings of figures, but no special design |
| 207 | Bobble bath | 4 – Colourful, designed as a special figure |
| 208 | Bobble bath | 4 – Colourful, designed as a special figure |
Based on the results of the survey (which showed that eight of the products had a packaging made of PVC) and of the results of the analyses for the content of chlorine in the packaging (see a description later) 10 products were selected for a quantitative determination of the content of phthalates in the products.
The 10 products for analysis for a content of phthalates were selected so they mainly are products designed as a particular figure as these products might be used as toys (for instance in the bath) and especially small children (or small siblings) might be expected to put them into the mouth. Therefore, it is relevant to examine for a possible content of PVC to be able to assess the impact in such situations.
Furthermore, the products have been selected so they cover several producers and so they represent both the retail trade products and products via the Internet, however, with a substantial majority of retail trade products.
Table 5.6: Selected for analysis for content of phthalates in the packaging
| ID no. | Product type | Design of the product |
| 5 | Body shampoo/bath gel | 4 – Colourful, designed as a special figure |
| 21 | Bobble bath | 4 – Colourful, designed as a special figure |
| 27 | Body shampoo/bath gel | 4 – Colourful, designed as a special figure |
| 32 | Body shampoo/bath gel | 4 – Colourful, designed as a special figure |
| 33 | Body shampoo/bath gel | 4 – Colourful, designed as a special figure |
| 34 | Body shampoo/bath gel | 4 – Colourful, designed as a special figure |
| 82 | Bobble bath | 3 – Colourful, with drawings of figures, but no special design |
| 128 | Body shampoo/bath gel | 4 – Colourful, designed as a special figure |
| 196 | Body shampoo/bath gel | 4 – Colourful, designed as a special figure |
| 207 | Bobble bath | 4 – Colourful, designed as a special figure |
5.2 Analyses for selected constituents
5.2.1 Analysis methods
5.2.1.1 Fragrances
A partial sample of the product is extracted by water and tert-butylmethylether by means of suspending, heating, cooling and standing during about 16 hours. A partial sample of the extract is extracted and analyzed directly via combined gas chromatography and mass spectrometry (GC/MS). The analyses are conducted as real repeat determination.
The detection limit is 1 mg/kg and the analysis error is 10-15% RSD.
For Oak moss extract and Tree moss extract a detection limit cannot be set as they are natural extracts with many components and not exclusively one pure substance. As the content of these natural extracts varies an exact detection limit cannot be calculated. Instead the limit is state as “Not proven”.
5.2.1.2 Chlormethy- a methylisothiazolones (Kathon)
A representative partial sample of about 2.5 g is extracted and diluted in demineralized water. It is filtered via a 0.45µm filter. The filtered solution is analyzed via liquid chromatography with UV detection (HPLC/DAD) with the following chromatographic conditions:
Flow: 0.500 ml/min
Eluentes: 35% methanol and 65% water, isochratic
Wavelength: 212 og 280 nm
Analysis time: 15 min.
The analyses are conducted as real repeat determination.
The analysis error is 10-15%. The detection limit is 2 mg/kg.
Reference: Matissek, R; Zur Analytik mikrobiocider Isothiazolone, Fresenius Z Anal. Chem. (1985) 322: 465-469.
5.2.2 Analysis results
5.2.2.1 Fragrances
17 cosmetics samples were totally analyzed for 26 fragrances in repeat determination. The sum of the proven fragrances varied from 1 mg/kg to 7800 mg/kg corresponding to from 0.0001% (w/w) to 0.78 (w/w).
The results are stated in the table in the unit mg/kg.
As described in chapter 3 ”Legislation”, and according to Appendix 3 of the statutory order, the 26 fragrances mandatory to declare must be stated in the declaration of content, no matter their function in the products, when the concentration is higher than 0.001% (i.e. 10 mg/kg) in products which are not to be cleansed and 0.01% (i.e. 100 mg/kg) in products which are to be cleansed. This regulation with special statement of the 26 fragrances mandatory to declare became effective in 2005 and applies for all cosmetics being produced after 10 March 2005 (Stat. Ord. 422 §25, 2006).
In theory, it is still possible to buy products in the shops which are produced before 10 March 2005 so that a non-declared content of one of the 26 fragrances mandatory to declare is legal. In connection with the survey we have seen that a number of products being bought for the survey did not have any separate declaration of the fragrances mandatory to declare but when buying for the analyses these fragrances were declared separately.
In the tables below three columns for each analyzed product is presented. The first two columns state the results from the repeat determinations. The third column states with a “+” which of the 26 fragrances that is separately declared on the declaration of content of the products. A “+” in the sum row indicates whether there is a content of “perfume” according to the declaration of content – and correspondingly a “-“ in the sum row states that perfume is not a part of the product. At last, “Yes” or “No” states whether the declaration matches the analysis results. So a “No” means divergences in relation to the Cosmetics Statutory Order. Please note, with exception of two (ID no. 166 and 209), that all the analyzed products are not stay-on products; that means that only a content of perfume above 100 mg/kg must be declared according to the Cosmetics Statutory Order.
Table 5.7: Results from the analysis for fragrances. The unit is mg/kg. The two results state the repeat determinations.
| The 26 fragrances (stated with their INCI name) | D.l. | 5 | 9 | 21 | ||||||
| Body shampoo/ bath gel |
Liquid soap | Bobble bath | ||||||||
| A | B | Decl. | A | B | Decl. | A | B | Decl. | ||
| Anise alcohol | 1 | - | - | - | - | - | - | |||
| Amyl cinnama | 1 | - | - | - | - | - | - | |||
| Amylcinnamyl alcohol | 1 | - | - | - | - | - | - | |||
| Benzyl alcohol | 1 | 95 | 91 | - | - | - | - | |||
| Benzyl benzoate | 1 | 20 | 25 | 4 | 6 | - | - | |||
| Benzyl cinnamate | 1 | - | - | - | - | - | - | |||
| Benzyl salicylate | 1 | - | - | - | - | 82 | 78 | |||
| Cinnamyl alcohol | 1 | - | - | - | - | - | - | |||
| Cinnamal | 1 | - | - | - | - | - | - | |||
| Citral | 1 | - | - | 73 | 69 | + | - | - | ||
| Citronellol | 1 | - | - | 50 | 53 | - | - | |||
| Coumarin | 1 | - | - | 21 | 23 | - | - | |||
| Eugenol | 1 | - | - | 39 | 42 | - | - | |||
| Farnesol | 1 | - | - | - | - | - | - | |||
| Geraniol | 1 | - | - | 180 | 180 | + | - | - | ||
| Hexyl cinnamal | 1 | - | - | - | - | 170 | 160 | |||
| Hydroxycitronellal | 1 | - | - | 32 | 33 | - | - | |||
| a-Isomethylionone | 1 | - | - | - | - | - | - | |||
| Butylpheny methylpropional (Lillial) | 1 | - | - | - | - | - | - | |||
| Limonene | 1 | - | - | 2100 | 2200 | + | - | - | ||
| Linalool | 1 | 15 | 12 | 1000 | 1100 | + | - | - | ||
| Hydroxyisohexyl 3-cyclohexer carboxaldehyde (Lyral) | 1 | - | - | 250 | 250 | + | - | - | ||
| Isoeugenol | 1 | - | - | - | - | - | - | |||
| Methyl 2-Octynoate | 1 | - | - | - | - | - | - | |||
| Evernia Prunastri (Oakmoss) Extract | * | * | * | * | * | * | * | |||
| Evernia Furfurancea (Treemoss) Extract | * | * | * | * | * | * | * | |||
| Sum | 130 | 130 | - | 3700 | 4000 | + | 250 | 240 | + | |
| Declaration matches the analysis results | No | Yes | No | |||||||
D.l.: means detection limit
-: means not proved above the detection limit
*: undetectable. A detection limit cannot be fixed
In the row marked “Declaration matches the analysis results”:
+: means that the fragrance is declared separately in the declaration of content on the product
-: marked under ”Sum” means that there is no declared content of perfume in the product. The word “perfume” is not in the declaration of content.
Table 5.7 (cont’d): Results from the analysis for fragrances. The unit is mg/kg. The two results state the repeat determinations.
| The 26 fragrances (stated with their INCI name) | D.l. | 22 | 32 | 33 | ||||||
| Body shampoo/ bath gel | Body shampoo/ bath gel | Body shampoo/ bath gel | ||||||||
| A | B | Decl. | A | B | Decl. | A | B | Decl. | ||
| Anise alcohol | 1 | - | - | - | - | - | - | |||
| Amyl cinnama | 1 | 210 | 230 | - | - | 18 | 17 | |||
| Amylcinnamyl alcohol | 1 | - | - | - | - | - | - | |||
| Benzyl alcohol | 1 | 4 | 6 | - | - | 7 | 7 | |||
| Benzyl benzoate | 1 | - | - | - | - | 9 | 11 | |||
| Benzyl cinnamate | 1 | - | - | - | - | - | - | |||
| Benzyl salicylate | 1 | - | - | - | - | 23 | 21 | |||
| Cinnamyl alcohol | 1 | 37 | 42 | - | - | - | - | |||
| Cinnamal | 1 | 2 | 2 | - | - | - | - | |||
| Citral | 1 | - | - | - | - | - | - | |||
| Citronellol | 1 | 8 | 9 | - | - | 12 | 13 | |||
| Coumarin | 1 | - | - | - | - | - | - | |||
| Eugenol | 1 | 6 | 8 | - | - | - | - | |||
| Farnesol | 1 | - | - | - | - | - | - | |||
| Geraniol | 1 | - | - | - | - | 9 | 8 | |||
| Hexyl cinnamal | 1 | 5 | 7 | - | - | 21 | 22 | + | ||
| Hydroxycitronellal | 1 | - | - | - | - | 19 | 17 | |||
| a-Isomethylionone | 1 | 16 | 18 | - | - | 7 | 8 | |||
| Butylpheny methylpropional (Lillial) | 1 | 47 | 53 | 13 | 12 | 37 | 39 | |||
| Limonene | 1 | 320 | 300 | + | - | - | - | - | + | |
| Linalool | 1 | - | - | 8 | 7 | 31 | 27 | |||
| Hydroxyisohexyl 3-cyclohexer carboxaldehyde (Lyral) | 1 | - | - | - | - | 7 | 7 | |||
| Isoeugenol | 1 | - | - | - | - | - | - | |||
| Methyl 2-Octynoate | 1 | - | - | - | - | - | - | |||
| Evernia Prunastri (Oakmoss) Extract | * | * | * | * | * | * | * | |||
| Evernia Furfurancea (Treemoss) Extract | * | * | * | * | * | * | * | |||
| Sum | 660 | 680 | + | 21 | 19 | + | 200 | 200 | + | |
| Declaration matches the analysis results | No | Yes | Yes | |||||||
D.l.: means detection limit
-: means not proved above the detection limit
*: undetectable. A detection limit cannot be fixed
In the row marked “Declaration matches the analysis results”:
+: means that the fragrance is declared separately in the declaration of content on the product
-: marked under ”Sum” means that there is no declared content of perfume in the product. The word “perfume” is not in the declaration of content.
Table 5.7 (cont’d): Results from the analysis for fragrances. The unit is mg/kg. The two results state the repeat determinations.
| The 26 fragrances (stated with their INCI name) | D.l. | 34 | 35 | 49 | ||||||
| Body shampoo/ bath gel | Body shampoo/ bath gel | Shampoo | ||||||||
| A | B | Decl. | A | B | Decl. | A | B | Decl. | ||
| Anise alcohol | 1 | - | - | - | - | - | - | |||
| Amyl cinnama | 1 | - | - | 5 | 5 | - | - | |||
| Amylcinnamyl alcohol | 1 | - | - | - | - | - | - | |||
| Benzyl alcohol | 1 | 110 | 120 | 21 | 22 | - | - | |||
| Benzyl benzoate | 1 | 27 | 33 | 31 | 35 | - | - | |||
| Benzyl cinnamate | 1 | - | - | - | - | - | - | |||
| Benzyl salicylate | 1 | - | - | 3 | 3 | - | - | |||
| Cinnamyl alcohol | 1 | - | - | - | - | - | - | |||
| Cinnamal | 1 | - | - | - | - | - | - | |||
| Citral | 1 | - | - | - | - | 6 | 8 | |||
| Citronellol | 1 | - | - | 4 | 4 | 5 | 3 | |||
| Coumarin | 1 | - | - | - | - | - | - | |||
| Eugenol | 1 | - | - | - | - | - | - | |||
| Farnesol | 1 | - | - | - | - | - | - | |||
| Geraniol | 1 | - | - | 3 | 3 | 61 | 56 | |||
| Hexyl cinnamal | 1 | - | - | 10 | 11 | + | - | - | ||
| Hydroxycitronellal | 1 | - | - | - | - | - | - | |||
| a-Isomethylionone | 1 | - | - | 3 | 3 | - | - | |||
| Butylpheny methylpropional (Lillial) | 1 | - | - | 14 | 15 | - | - | |||
| Limonene | 1 | - | - | - | - | + | 1100 | 1100 | + | |
| Linalool | 1 | - | - | + | 12 | 11 | 72 | 64 | + | |
| Hydroxyisohexyl 3-cyclohexer carboxaldehyde (Lyral) | 1 | - | - | - | - | - | - | |||
| Isoeugenol | 1 | - | - | - | - | - | - | |||
| Methyl 2-Octynoate | 1 | - | - | - | - | - | - | |||
| Evernia Prunastri (Oakmoss) Extract | * | * | * | * | * | * | * | |||
| Evernia Furfurancea (Treemoss) Extract | * | * | * | * | * | * | * | |||
| Sum | 140 | 150 | + | 110 | 110 | + | 1200 | 1200 | + | |
| Declaration matches the analysis results | No | Yes | Yes | |||||||
D.l.: means detection limit
-: means not proved above the detection limit
*: undetectable. A detection limit cannot be fixed
In the row marked “Declaration matches the analysis results”:
+: means that the fragrance is declared separately in the declaration of content on the product
-: marked under ”Sum” means that there is no declared content of perfume in the product. The word “perfume” is not in the declaration of content.
Table 5.7 (cont’d): Results from the analysis for fragrances. The unit is mg/kg. The two results state the repeat determinations.
| The 26 fragrances (stated with their INCI name) | D.l. | 69 | 82 | 98 | ||||||
| Shampoo | Bobble bath | Bath confetti/caviar/ fizzle salt |
||||||||
| A | B | Decl. | A | B | Decl. | A | B | Decl. | ||
| Anise alcohol | 1 | - | - | - | - | - | - | |||
| Amyl cinnama | 1 | - | - | - | - | - | - | |||
| Amylcinnamyl alcohol | 1 | - | - | - | - | - | - | + | ||
| Benzyl alcohol | 1 | 1 | 1 | - | - | 2 | 2 | |||
| Benzyl benzoate | 1 | - | - | - | - | - | - | + | ||
| Benzyl cinnamate | 1 | - | - | - | - | - | - | + | ||
| Benzyl salicylate | 1 | - | - | - | - | - | - | + | ||
| Cinnamyl alcohol | 1 | - | - | - | - | - | - | + | ||
| Cinnamal | 1 | - | - | - | - | - | - | |||
| Citral | 1 | - | - | - | - | - | - | + | ||
| Citronellol | 1 | - | - | - | - | - | - | + | ||
| Coumarin | 1 | - | - | - | - | - | - | + | ||
| Eugenol | 1 | - | - | - | - | - | - | + | ||
| Farnesol | 1 | - | - | - | - | - | - | + | ||
| Geraniol | 1 | - | - | - | - | - | - | + | ||
| Hexyl cinnamal | 1 | - | - | - | - | - | - | + | ||
| Hydroxycitronellal | 1 | - | - | - | - | - | - | |||
| a-Isomethylionone | 1 | - | - | 10 | 14 | - | - | + | ||
| Butylpheny methylpropional (Lillial) | 1 | - | - | - | - | - | - | + | ||
| Limonene | 1 | - | - | 250 | 230 | + | - | - | + | |
| Linalool | 1 | - | - | - | - | - | - | + | ||
| Hydroxyisohexyl 3-cyclohexer carboxaldehyde (Lyral) | 1 | - | - | - | - | - | - | + | ||
| Isoeugenol | 1 | - | - | - | - | - | - | |||
| Methyl 2-Octynoate | 1 | - | - | - | - | - | - | |||
| Evernia Prunastri (Oakmoss) Extract | * | * | * | * | * | * | * | |||
| Evernia Furfurancea (Treemoss) Extract | * | * | * | * | * | * | * | |||
| Sum | 1 | 1 | + | 260 | 240 | + | 2 | 2 | + | |
| Declaration matches the analysis results | Yes | Yes | No, but legal | |||||||
D.l.: means detection limit
-: means not proved above the detection limit
*: undetectable. A detection limit cannot be fixed
In the row marked “Declaration matches the analysis results”:
+: means that the fragrance is declared separately in the declaration of content on the product
-: marked under ”Sum” means that there is no declared content of perfume in the product. The word “perfume” is not in the declaration of content.
Table 5.7 (cont’d): Results from the analysis for fragrances. The unit is mg/kg. The two results state the repeat determinations.
| The 26 fragrances (stated with their INCI name) | D.l. | 166[9] | 170 | 191 | ||||||
| Body lotion/cream | Body shampoo/ bath gel | Shampoo | ||||||||
| A | B | Decl. | A | B | Decl. | A | B | Decl. | ||
| Anise alcohol | 1 | - | - | - | - | - | - | |||
| Amyl cinnama | 1 | - | - | - | - | - | - | |||
| Amylcinnamyl alcohol | 1 | - | - | - | - | - | - | |||
| Benzyl alcohol | 1 | 780 | 790 | 11 | 10 | 8 | 7 | + | ||
| Benzyl benzoate | 1 | 210 | 200 | + | 40 | 52 | - | - | ||
| Benzyl cinnamate | 1 | - | - | - | - | - | - | |||
| Benzyl salicylate | 1 | 15 | 14 | + | 23 | 24 | - | - | ||
| Cinnamyl alcohol | 1 | - | - | - | - | - | - | |||
| Cinnamal | 1 | - | - | - | - | - | - | |||
| Citral | 1 | - | - | - | - | - | - | |||
| Citronellol | 1 | 10 | 10 | + | - | - | - | - | ||
| Coumarin | 1 | - | - | - | - | - | - | |||
| Eugenol | 1 | - | - | - | - | - | - | |||
| Farnesol | 1 | - | - | - | - | - | - | |||
| Geraniol | 1 | - | - | - | - | 2 | 2 | |||
| Hexyl cinnamal | 1 | 140 | 110 | + | 120 | 120 | + | - | - | |
| Hydroxycitronellal | 1 | - | - | - | - | - | - | |||
| a-Isomethylionone | 1 | 21 | 22 | - | - | - | - | |||
| Butylpheny methylpropional (Lillial) | 1 | - | - | - | - | - | - | |||
| Limonene | 1 | 400 | 350 | + | 6 | 6 | - | - | ||
| Linalool | 1 | 31 | 26 | 9 | 9 | - | - | |||
| Hydroxyisohexyl 3-cyclohexer carboxaldehyde (Lyral) | 1 | - | - | 14 | 16 | - | - | |||
| Isoeugenol | 1 | - | - | - | - | - | - | |||
| Methyl 2-Octynoate | 1 | - | - | - | - | - | - | |||
| Evernia Prunastri (Oakmoss) Extract | * | * | * | * | * | * | * | |||
| Evernia Furfurancea (Treemoss) Extract | * | * | * | * | * | * | * | |||
| Sum | 1600 | 1500 | + | 220 | 240 | + | 10 | 9 | + | |
| Declaration matches the analysis results | No | Yes | Yes | |||||||
D.l.: means detection limit
-: means not proved above the detection limit
*: undetectable. A detection limit cannot be fixed
In the row marked “Declaration matches the analysis results”:
+: means that the fragrance is declared separately in the declaration of content on the product
-: marked under ”Sum” means that there is no declared content of perfume in the product. The word “perfume” is not in the declaration of content.
Table 5.7 (cont’d): Results from the analysis for fragrances. The unit is mg/kg.
The two results state the repeat determinations.
| The 26 fragrances (stated with their INCI name) | D.l. | 206 | 209[10] | ||||
| Shampoo | Eau de toilette | ||||||
| A | B | Decl. | A | B | Decl. | ||
| Anise alcohol | 1 | - | - | - | - | ||
| Amyl cinnama | 1 | - | - | - | - | ||
| Amylcinnamyl alcohol | 1 | - | - | - | - | ||
| Benzyl alcohol | 1 | 8 | 8 | 5 | 5 | ||
| Benzyl benzoate | 1 | - | - | 16 | 21 | + | |
| Benzyl cinnamate | 1 | - | - | - | - | ||
| Benzyl salicylate | 1 | - | - | 17 | 17 | + | |
| Cinnamyl alcohol | 1 | - | - | - | - | ||
| Cinnamal | 1 | - | - | - | - | ||
| Citral | 1 | - | - | 4 | 5 | ||
| Citronellol | 1 | - | - | 290 | 300 | + | |
| Coumarin | 1 | - | - | - | - | ||
| Eugenol | 1 | - | - | - | - | ||
| Farnesol | 1 | - | - | 7 | 7 | ||
| Geraniol | 1 | - | - | 33 | 34 | + | |
| Hexyl cinnamal | 1 | - | - | - | - | ||
| Hydroxycitronellal | 1 | - | - | 2 | 2 | ||
| a-Isomethylionone | 1 | - | - | 480 | 530 | + | |
| Butylpheny methylpropional (Lillial) | 1 | - | - | 3400 | 3400 | + | |
| Limonene | 1 | - | - | 48 | 59 | ||
| Linalool | 1 | - | - | 750 | 730 | + | |
| Hydroxyisohexyl 3-cyclohexer carboxaldehyde (Lyral) | 1 | - | - | 2700 | 2700 | + | |
| Isoeugenol | 1 | - | - | - | - | ||
| Methyl 2-Octynoate | 1 | - | - | - | - | ||
| Evernia Prunastri (Oakmoss) Extract | * | * | * | * | * | ||
| Evernia Furfurancea (Treemoss) Extract | * | * | * | * | * | ||
| Sum | 8 | 8 | + | 7800 | 7800 | + | |
| Declaration matches the analysis results | Yes | No | |||||
D.l.: means detection limit
-: means not proved above the detection limit
*: undetectable. A detection limit cannot be fixed
In the row marked “Declaration matches the analysis results”:
+: means that the fragrance is declared separately in the declaration of content on the product
-: marked under ”Sum” means that there is no declared content of perfume in the product. The word “perfume” is not in the declaration of content.
As it can be seen from the analysis results six of the 17 analyzed products have declaration of content which does not match the analysis results. With regard to these six products there are divergences in relation to the Cosmetics Statutory Order if the products are produced after 10 March 2005. For one of the products (ID no. 5) there is no declared content of perfume in the products – nor in the form of the word “perfume” but three of the fragrances mandatory to declare are identified in the product; however, each in a concentration so the substances have not to be declared separately. It shall also be mentioned that a part of the found concentrations of fragrances are very low. Here it may be the case that the fragrances are a part of the products as an impurity from the production and are not actively added. In such cases the substance is not seen as a constituent and therefore it is not to be declared.
Table 5.8: Summary of the analysis results for fragrances
| Number of products analyzed in total | 17 |
| Number of products where declaration and analysis results are in accordance | 10 |
| Number of products where declaration and analysis results deviate from one another but the declaration is legal as too many fragrances are declared | 1 |
| Number of products where declaration and analysis results deviate from one another – that means illegal products if produced after 10 March 2005 | 6 |
| Measured concentrations of the 26 fragrances in total | Between 1 and 7800 mg/kg |
| Measured concentrations of a single fragrance | Between 1 and 3400 mg/kg |
In a single product 18 of 26 fragrances mandatory to declare are declared but none of these 18 fragrances is identified when analyzing the product.
The table below shows the fragrances occuring in the maximum concentration and above the 100 mg/kg requiring a separate declaration (for non-stay-on products).
Table 5.9: Overview of fragrances measured in the maximum concentration in the analyzed products
| Fragrance (stated with their INCI name) | Maximum measured concentration mg/kg | Occurs in x out of the 208 mapped products[11] | |
| 1 | Butylphenyl methylpropionial (Lillial) | 3400 | 16 |
| 2 | Hydroxy isohexyl 3-cyclohexene carboxaldehyde (Lyral) | 2700 | 12 |
| 3 | D-Limonene (limonene) | 2200 | 48 |
| 4 | Linalool | 1100 | 45 |
| 5 | Benzyl alcohol | 790 | 20 |
| 6 | a-isomethylionone | 480 | 12 |
| 7 | Citronellol | 300 | 22 |
| 8 | Amyl cinnamal | 230 | 17 |
| 9 | Benzyl benzoate | 210 | 19 |
| 10 | Geraniol | 180 | 25 |
| 11 | Hexylcinnamal | 170 | 21 |
5.2.2.2 Chlormethyl and methyliso-thiazolones (Kathon)
Kathon is a mixture of methylisothiazolone and chlormethylthiazolone. In total 11 samples were analyzed and the results are stated in Table 2.
Table 5.10: Results from the analysis for Chlormethyl and methylisothiazolones.
The unit is mg/kg. The two results state the repeat determinations.
| D.l. | 5 | 9 | 19 | ||||
| Body shampoo/ bath gel |
Liquid soap | Body shampoo/ bath gel |
|||||
| A | B | A | B | A | B | ||
| Chlormethyl and methylisothiazolones | 2 | 10 | 12 | 7.5 | 7.7 | ||
| D.l. | 21 | 33 | 34 | ||||
| Bobble bath | Body shampoo/ bath gel |
Body shampoo/ bath gel |
|||||
| A | B | A | B | A | B | ||
| Chlormethyl and methylisothiazolones | 2 | 3.0 | 3.2 | ||||
| D.l. | 35 | 82 | 170 | ||||
| Body shampoo/ bath gel |
Bobble bath | Body shampoo/ bath gel |
|||||
| A | B | A | B | A | B | ||
| Chlormethyl and methylisothiazolones | 2 | 11 | 7.4 | 5.1 | 7.1 | ||
| D.l. | 193 | 206 | |||
| Body lotion/ cream | Shampoo | ||||
| A | B | A | B | ||
| Chlormethyl and methylisothiazolones | 2 | 6.7 | 4.4 | ||
D.l.: means ”detection limit”
-: means ”not proved above the detection limit”
According to the Cosmetics Statutory Order (Stat. Ord. 422 §25, 2006) the maximum allowed concentration of Kathon in cosmetic products is 0.0015% of a mixture in the ration 3:1 of methylchloroisothiazolinone and methylisothiazolinone. This corresponds to 15 mg/kg. Therefore, none of the 11 analyzed products exceeds the allowed value.
However, it is worth noting that three of the analyzed products (ID 21, 82 and 170) have no declared content of Kathon despite an analyzed content of Kathon. Therefore, these products deviate from the Cosmetics Statutory Order.
5.3 Analyses of selected packaging materials
5.3.1 Analysis methods
5.3.1.1 Heavy metal determination by apply of X-ray analysis (XRF)
The XRF analyses are prepared in a X-LAB 2000 instrument (Spectro). By apply of this technique all elements larger than or equal with no. 11, Natrium (Na), are analyzed. Minimum quantity which can be determined depends on matrix and element but for certain elements it is <10ppm. The sample which is to be analyzed is placed directly in the instrument. The analysis is a surface analysis, that means that the analysis is in a maximum depth of about 100µm dependent on the material. By use of the analysis the content of heavy metals is available and also an indication whether the sample contains chlorinated or brominated flame retardants.
Detection limits and uncertainties are stated in Table 5.11.
5.3.1.2 Polymers (incl. PVC)
Beilstein’s test is used to determine whether a polymer is PVC. Beilstein’s test is a quick quantitative method for determination of halogens as a little piece of the sample is burnt on a copper wire in a flame. Green colouring of the flame indicates a content of chlorine. Beilstein’s test is carried out as screening on all polymers.
5.3.1.3 Phthalates
Products can be analyzed for the quantitative content of the different phthalates, for instance where a content of phthalates is identified via the FTIR analysis. The samples are extracted with dichlormethan and the extracts are analyzed with GC-MS. For the GC-MS analyses Varian Saturn 2000 ion-trap GC-MS system is used.
Detection limits and uncertainties are stated in Table 5.11.
Table 5.11: Detection limits and uncertainties for the stated analyses
| Substance | Analysis method | Detection limit | Uncertainty % rel. |
|
| Migration | Quantitative | |||
| Heavy metals | X-ray analysis | 1 mg/kg | 10 | |
| Phthalates | prEN71:11 section.5.7 Extraction with dichlormethan, GCMS |
0.005 mg/l | 0.001W/W% | 10 |
5.3.2 Analysis results
5.3.2.1 Content of chlorine in the packaging
As mentioned, 14 products have been analyzed for content of chlorine in the packaging. The results of Beilstein’s test are stated in the table below. A “+” states a content of chlorine in the packaging. A few products have been via both X-ray analysis (for metals) and Beilstein’s test. For these products the result of the chlorine content via the X-ray analysis is also stated (in %) in Table 5.12.
The results confirm that all small body shampoo/bath gel or bobble bath products being produced in soft plastic and designed as a particular figure are most probably made of PVC as the packing materials contain chlorine. Products in this test which are tested negatively for content of chlorine are all of a different type, primarily produced in soft plastic.
Table 5.12: Results of Beilstein’s test for chlorine in the packaging
| ID no. | Product type | Product description | Part | Content of chlorine | Cl (NB: %) |
| 5 | Body shampoo/bath gel | 4 – Colourful, designed as a special figure. In soft plastic. |
Outside Plate inside |
+ + |
|
| 18 | Body shampoo/bath gel | 4 – Colourful, designed as a special figure. In soft plastic. |
+ | ||
| 33 | Body shampoo/bath gel | 4 – Colourful, designed as a special figure. In soft plastic. |
+ | ||
| 34 | Body shampoo/bath gel | 4 – Colourful, designed as a special figure. In soft plastic. |
+ | ||
| 35 | Body shampoo/bath gel | 4 – Colourful, designed as a special figure. In soft plastic. |
+ | ||
| 40 | Hairstyling product | 3 – Colourful, with drawings of figures, but no special design | Container Cover (stopper) |
- - |
0.01% |
| 82 | Bobble bath | 3 – Colourful, with drawings of figures, but no special design | Container Stopper |
+ - |
|
| 128 | Body shampoo/bath gel | 4 – Colourful, designed as a special figure. In soft plastic. |
+ | ||
| 193 | Body lotion/cream | 4 – Colourful, designed as a special figure | Yellow plastic Green plastic stomach |
- - |
0.05% |
| 194 | Body shampoo/bath gel | 4 – Colourful, designed as a special figure. In soft plastic. |
+ | ||
| 196 | Body shampoo/bath gel | 4 – Colourful, designed as a special figure. In soft plastic. |
+ | ||
| 206 | Shampoo | 3 – Colourful, with drawings of figures, but no special design | Container Stopper Tube near stripes |
- - |
0.2% |
| 207 | Bobble bath | 4 – Colourful, designed as a special figure. In soft plastic. |
Near valve | + | 30% |
| 208 | Bobble bath | 4 – Colourful, designed as a special figure. In soft plastic. |
+ |
+ means that a content of chlorine is found by means of Beilstein’s test
- means that no content of chlorine is found by means of Beilstein’s test
5.3.2.2 Content of heavy metals in the packaging
As mentioned, 10 products have been analyzed for content of heavy metals (metals) in the packaging. The results of the X-ray analysis are stated in the table below.
Table 5.13: Results of X-ray analysis for heavy metals in the packaging
Already during the study we knew that products no. 21, 27 and 32 were made of PVC (via plastic code). This is confirmed by the X-ray analysis.
Generally, the results show values below 10 ppm whereas the values especially for nickel are somewhat higher (max. 32 ppm). Furthermore, two values for chromium are 15 ppm and a single value for tin is significantly higher than the other values – 360 ppm. A possible explanation might be use of organic tin compounds as heat stabilizer in PVC. According to Plastics Additive Handbook (Hanser Publications, 2001) sulphurous organic tin compounds are among the most efficient and most widespread heat stabilizer.
In the European standard for safety for toys, EN 71-3, dealing with migration of special substances, limit values for migration of substances from toys materials are stated. These limit values are stated in the table below. For comparison the maximum analyzed values in the 10 cosmetic products for children are stated. The maximum measured values occur in all products with a special design of which some can be included in the Toys directive. Please note that these values are not migration values but on the contrary total values in the products.
Table 5.14: Limit values for migration of substances from toy materials
| Cr | As | Se | Cd | Sb | Ba | Hg | Pb | |
| Maximum migration of substances from toy materials in mg/kg (ppm) | 60 | 25 | 500 | 75 | 60 | 1000 | 60 | 90 |
| Maximum measured total value in the analysed cosmetic products | 15 | 1.4 | 1.1 | 3.4 | 12 | 52 | 2 | 4 |
Please note that the maximal analyzed values in the cosmetic products for children are total values and not migration values.
From Table 5.14 can be seen that all the measured total values are substantially below the limit values for migration of the substances from toys (between 4 or 450 times below the limit values). Thus the measured total values are not a cause for concern in relation to the existing limit values for migration of substances from toy materials. For the same reason the planned migration analyses (according to EN 71-3) of the packaging materials are not conducted.
5.3.2.3 Content of phthalates in the packaging material
10 products were selected for a quantitative analysis of phthalates in the packaging material where chlorine in the packaging material was proved or where statement on the packaging stated that it was made of PVC. Of the 10 product 3 are assessed to be toys (ID no. 21, 27 and 32) based on the indicative statement of the Danish Safety Technology Authority. The rest of the products are defined as child care articles. The results are stated in the table below.
Table 5.15: Content of phthalates in 10 selected packaging materials
| Analyzed content of phthalate in the packaging (in %) | |||||
| ID No. | Product type | Packaging description | DEHP | DINP | DEHIP1 (DnOP) |
| 5 | Body shampoo/bath gel | 4 – Colourful, designed as a special figure. In soft plastic. |
27 | ||
| 21 | Bobble bath | 4 – Colourful, special design | 0.4 | 15 | |
| 27 | Body shampoo/ bath gel |
4 – Colourful, special design | Content of phthalate < 0.05% Softened with citric acid tributylesteraetat (see text) |
||
| 32 | Body shampoo/ bath gel |
4 – Colourful, special design | 11 | ||
| 33 | Body shampoo/bath gel | 4 – Colourful, designed as a special figure. In soft plastic. |
26 | 1 | |
| 34 | Body shampoo/bath gel | 4 – Colourful, designed as a special figure. In soft plastic. |
27 | ||
| 82 | Bobble bath | 3 – Colourful, with drawings of figures, but no special design | < 0.05 | ||
| 128 | Body shampoo/bath gel | 4 – Colourful, designed as a special figure. In soft plastic. |
31 | ||
| 196 | Body shampoo/bath gel | 4 – Colourful, designed as a special figure. In soft plastic. |
28 | ||
| 207 | Bobble bath | 4 – Colourful, special design In soft plastic |
26 | ||
1: It is not an assured identification of DEHIP due to lacking reference substance. Here DEHIP is calculated as DEHP. But the found phthalate is most probably DEHIP or alternatively DnOP based on the spectras.
DEHP: Bis(2-ethylhexyl)phthalate CAS no.: 117-81-7
DINP: Di-isononyl phthalate (isomeric mixture) CAS no.: 28553-12-0
DEHIP: Bis(2-ethylhexyl)iso phthalate
DnOP: Di-n-octyl phthalate CAS no.: 117-84-0
According to the analyses, product no. 27 contains presumably acetyltributylcitrat (CAS no.: 77-90-7) as plasticizer but probably mixed with (CAS no. 77-94-1) and tributylaconitat (CAS no. 7568-58-3). Futhermore, several compounds in small quantities were found.
Product no. 82 had no detectable content of extractable plasticizers.
The analysis results confirm that the small bath products, designed as a particular figure and made in soft plastic, are made of PVC and have a high content of phthalates. For these figures the phthalate content is between 26 and 31% phthalates.
Three large bath products designed as a standing cartoon figure and made in somewhat harder plastic were also analyzed for the content of phthalates. One of the figures was not softened by use of phthalates (content < 0.05%) while the two other figures had a phthalate content of 11 and 15% respectively.
The identified types of phthalates are DEHP (Bis(2-ethylhexyl)phthalate) and DINP (Di-isononyl phthalate (isomeric mixture)). Furthermore, a content of something was found. From the spectra it seemed to be DEHIP[12] (Bis(2-ethylhexyl)iso phthalate). It might also be DnOP (di-n-octyl phthalate). This is not known for sure due to missing reference standard.
As described in chapter 3.2.2 ”Ban on phthalates in toys for children”, from April 2007 DEHP, DINP and DnOP are banned in toys for children up to 14 years and in child care articles in a concentration above 0.1% - for DINP and DnOP only in products which children might put into the mouth. Some of the analyzed products are thus in discrepancy with the new phthalate legislation per 16 April 2007. However, on the date of purchase all the analyzed products were legal (spring 2006).
6 Health assessment
6.1 Selection of substances for health assessment
As basis for the selection of substances for the health assessment the list of the most frequently used substances in cosmetic products for children is used. However, in the selection emphasis has been on substances with the most worrisome properties and primarily it has been chosen to emphasize on the risk of developing allergy.
Earlier health assessments of a number of the 26 fragrances mandatory to déclassé have been conducted for which reason these are not repeated in this project. Furthermore, parabens are under risk assessment in the EU for the moment and therefore they are also deselected at the health assessment of this project.
In the selection of fragrances for the health assessment emphasis has been on the analysis results and a fragrance is selected which is identified in the analyses in a concentration which requires a separate declaration of the substance. Therefore, the substances stated in the table below have been chosen for health assessment.
| INCI name | CAS No. | Function | In number of mapped products |
| Benzyl alcohol | 100-51-6 | Preservative. Fragrance. Solvent. |
20 |
| Sodium benzoate | 532-32-1 | 13 | |
| Methylchlorisothiazolinone and Metyhlisothiazolinone | MI 2682-20-4 CMI 26172-55-4 MI/CMI 55965-84-9 |
Preservative | 15 |
| Phenoxyethanol | 122-99-6 | Preservative | 50 |
| 5-Bromo-5-nitro-1,3-dioxane | 30007-47-7 | Preservative | 15 |
6.2 Fragrances
6.2.1 Benzyl alcohol
Occurrence and application
Yearly, 40,000 tons of benzyl alcohol are produced worldwide. The majority (30%) is applied as hardening agent in epoxy paints where it is chemically combined after reaction. Other important applications are as solvents in low concentrations in water-based paints (10%), as constituent in paint remover (10%), and as chemical intermediate in the synthesis of benzyl esters which are applied in aroma and in the perfume industry (10%) (OECD SIDS, 2001). Finally, benzyl alcohol is applied in many different cosmetic products as fragrance, preservative, solvent and viscosity decreasing agent (SCF 78, 2002). Benzyl alcohol has a bacteriostatic impact and therefore it is also applied as a preservative (Jensen AA, 1999).
In the mapped cosmetic products for children benzyl alcohol occurs in 20 of the 208 products, distributed on six hair dyes, six bath confetti/caviar/fizzle salt products, one tooth paste, one eau de toilette, one body shampoo/bath gel, one shampoo, one bobble bath products, one body lotion/cream and finally one product in the category others (body splash), that means in nearly all kind of cosmetic products being investigated in this project.
Benzyl alcohol is applied as a local anesthetic and a pharmaceutical remedy. Benzyl alcohol is a natural constituent in a number of different plants, including jasmine. Benzyl alcohol occurs naturally in for instance some edible fruits (up to 5 mg/kg) and in green and black tea (1-30 and 1-15 mg/kg respectively). Benzyl alcohol is also added to some food products and beverages as a flavouring agent (up to 400 mg/kg – however, for chewing gum 1,254 mg/kg) (SCF 78, 2002; Jensen AA, 1999). In Europe the yearly application of benzyl alcohol as an additive in food products is about 110 tons. WHO estimates that the daily intake of benzyl alcohol is 16,000 µg/day corresponding to 270 µg/kg bw/day in Europe (WHO, 2002).
Identification
| Chemical name | Benzyl alcohol |
| Synonyms | (Hydroxymethyl)benzene Benzenecarbinol a-Hydroxytoluene Phenylmethanol Phenylmethyl alcohol |
| INCI name | Benzyl alcohol |
| CAS No. | 100-51-6 |
| EINECS No. | 202-859-9 |
| Gross formula | C7H8O |
| Molecular structure |  |
| Legislation: The list of dangerous substances (Stat. Ord. 923, 2005) The list of harmful substances (Orientation from the Danish EPA no. 8, 2004) The Danish EPA’s self classification (Environmental project no. 635, 2001) Cosmetics Statutory Order (Stat. Ord. 422 , 2006) |
XN; R20/22. Conc. = 25%: XN; R20/22 R20/22 = ”Dangerous if inhaled and if swallowed”. Yes No. Maximum permitted concentration as preservative in cosmetic products is 1% (Appendix 5, serial number 4 VI, 1, 34). Is one of the 26 fragrances which by SCCNFP* is assessed to be allergenic in case of contact with skin. As fragrance the presence of the substance must be stated in the declaration of content when the concentration is higher than 0.001% in products which are not to be cleansed and when the concentration is higher than 0.01% in products which are to be cleansed. |
| International Fragrance Association (IFRA) | No guidelines from IFRA for this substance. |
* The Scientific Committee on Cosmetic Products and Non-Food Products
Physical chemical properties
The physical chemical properties of benzyl alcohol are found via the ChemIDplus database of TOXNET, Chemfinder and the OECD report on benzoates (OECD SIDS, 2001).
| Physical structure | Colourless liquid |
| Molar weight (g/mol) | 108.14 |
| Melting point | -15 °C |
| Boling point | 205 °C |
| Steam pressure | 0.13 hPa at 20 °C 0.3 hPa at 20 °C (according to IUCLID, 2000b) 0.02 kPa at 250 °C |
| Octanol water distribution coefficient (log POW) | 1.1 |
| Water solubility | 42.9 g/L 35 g/L at 20 °C |
According to HSDB the odour threshold of benzyl alcohol is 5.5. ppm.
Absorption and distribution
Benzyl alcohol is easily absorbed via the skin – the quantity which is not evaporated at first. In animal trials with rhesus monkeys, benzyl alcohol was applied to the skin of the monkeys. Under normal application only 32% was absorbed via the skin during 24 hours because the substance evaporated. Under circumstances where the benzyl alcohol was not able to evaporate so easily 56-80% of the substance was absorbed (IUCLID, 2000b).
Benzyl alcohol is easily absorbed in the gastrointestinal tract. After the absorption it is quickly transformed in the liver to benzoic acid which is separated within 24 hours, mainly as hippuric acid in the urine. At very high dosages (> 500 mg/kg/day via oral intake) a saturation of this process of metabolism might occur (Jensen AA, 1999; Nair B, 2001; OECD SIDS, 2001; WHO, 2002).
Acute toxicity
Earlier benzyl alcohol has been applied in concentrations of about 1% as preservative in infusion liquids with physiological salt water and in a number of injection preparations. In 1983 the Danish National Board of Health warned against this use as in the USA in the early 1980’ies reports told about brain damages, blood changes and several deaths among premature babies which were caused by these applications. Total dosage applied to the children who died was 130-405 mg benzyl alcohol per kg body weight per day. This corresponds to the intravenous LD50 value found in test animals. If swallowed benzyl alcohol is 25 times less acutely toxic (Jensen AA, 1999).
Intake of large quantities of benzyl alcohol leads to vomiting and diarrhea and depression of the central nervous system (HSDB). Similarly five premature babies got several injections added bacteriostatic water with 0.9% benzyl alcohol. Daily dosages were 99-234 mg benzyl alcohol per kg body weight. The seen impacts were gradually neurological destruction, serious acidosis (abnormally strong acidification in the body), severely gasping breathing, destruction of the skin, impacts on the blood, impact on liver and kidney function, low blood pressure and collapse of the cardiovascular system (Gershenk et al, 1982).
According to “The list of dangerous substances” benzyl alcohol is assessed to be harmful to health when inhaled and swallowed. Tabel 6-1 states the acute toxicity in test animals.
Tabel 6-1: Acute toxicy (LD50) of benzyl alcohol
| Species | Exposure | LD50 (mg/kg bw) |
Reference |
| Rabbit | Oral | 1000 | Graham & Kuizenga, 1945 |
| Rat | Oral | 1600 | Procter & Gamble, 1992 |
| Rat | Oral | 3100 | Smyth et al., 1951 |
| Rat | Oral | 2080 | Opdyke, 1973. Graham & Kuizenga, 1945 |
| Moapply | Oral | 1580 | Opdyke, 1973. Jenner et al, 1964. |
| Rat | Oral | 1610 | Loeser, 1978. |
| Rat | Oral | 1230 | RTECS |
| Rat | Oral | 1610 | IUCLID, 2000b |
| Rat | Oral | 1230 | IUCLID, 2000b |
| Moapply | Oral | 1150 | IUCLID, 2000b |
| Rabbit | Oral | 1040 | IUCLID, 2000b |
| Rabbit | Dermal | 2000 | NPIRI, 1974. |
| Guinea pig | Dermal | < 5000 | IUCLID, 2000b |
WHO has set the limit for the acceptable daily intake of benzyl alcohol to 5 mg/kg/day. In general, US FDA (US Food and Drug Administration) sees benzyl alcohol to be safe for apply in food products (Nair B., 2001). A WHO working group has examined benzyl alcohol and other similar additives for food products and concludes that benzyl alcohol does not give cause for concern in relation to the health at a continuous use at the present level (WHO, 2002).
In tests with rats no effects were seen at a dose of 500 mg/kg body weight/day (375 mg/kg body weight/day when adjustment is made for dose only given five times a week). The value is based on a study of 16 days where the effects at higher doses were increased mortality, reduction in body weight, haemorrhage and lethargy. Correspondingly, no effects were seen at a dose of 400 mg/kg body weight/day (286 mg/kg body weight/day adjusted for five-day dosing schedule) in a subchronic study of 13 weeks. There were neurotoxic effects at higher doses. At a corresponding test of 16 days and a subchronic test (13 weeks) with mice no effects were seen at 250 (188 when adjusted for five-day dosing schedule) and 400 (286 when adjusted for five-day dosing schedule) mg/kg body weight/day respectively. In the test of 16 days the effects were lethargy at a higher dose and at the test of 13 weeks there were neurotoxic effects at a higher dose (NTP, 1989). Based on these tests 400 (286) mg/kg body weight/day can be regarded as NOAEL value.
Local irritation
Benzyl alcohol is irritating on skin (in a 0.5% solution), mucous membranes and eyes (at 100 mg per animal) and is affecting the central nervous system (Jensen AA, 1999; IUCLID, 2000b; OECD SIDS, 2001).
Allergy
In patch tests on humans benzyl alcohol has shown sensitizing properties in some tests but also ambiguous and negative results are seen (IUCLID, 2000b; OECD SIDS, 2001). Scientific Committee on Cosmetic Products and Non-Food Products (SCCNFP) evaluates that benzyl alcohol is one of the fragrances which most frequently is reported as allergenic to consumers (SCCNFP 0017, 1999). Benzyl alcohol is one of the 26 fragrances mandatory to declare which according to the Cosmetics legislation must be declared separately, with the purpose that allergic persons can avoid the substance.
Prolonged, repeated impact and gene damaging effects
Generally, benzyl alcohol shows low toxicity at repeated impact (OECD SIDS, 2001). Long-term tests with rats and mice showed no serious impacts of benzyl alcohol – the impacts were limited to be reduced food intake and reduced growth at doses of more than 400 mg/kg body weight/day.
In a reproduction study with mice the impacts were lower maternal body weight and decreasing mean litter weights. In another study a decreasing fetal weight in relation to the control group was noted while a third study showed no difference between the control group and the group of animals exposed to benzyl alcohol (Nair B., 2001).
In animal tests with mice and rats there were no signs that benzyl alcohol is carcinogenic (Jensen AA, 1999; Nair B., 2001; WHO, 2002; NTP, 1989).
In long-term tests on mice and rats there were no signs of carcinogenic impact of benzyl alcohol neither in female nor male rats at dosages of 200 and 400 mg/kg in the feed. Correspondingly there were no signs of carcinogenic impact of benzyl alcohol neither in female nor male rats at a dosage of 100 and 200 mg/kg for two years (NTP, 1989). In the OECD SIDS report on benzoates, a NOAEL value for benzyl alcohol of =400 mg/kg/day for the rat test and =200 mg/kg/day for the mouse test is stated, based on the above NTP report (OECD SIDS, 2001).
In the OECD SIDS report, a NOAEL value for benzyl alcohol of 550 mg/kg bw/day for developmental toxicity for mice is stated (OECD SIDS, 2001).
Benzyl alcohol has not shown to be mutagenic in the Ames test and other bacterial tests or in cell cultures (Jensen AA, 1999; Nair B., 2001; WHO, 2002; NTP, 1989).
Critical impact
At prolonged exposure the critical impact of benzyl alcohol is assessed to be neurotoxic effects. Here a NOEAL value of 400 mg/kg body weight/day is stated. With regard to the acute toxicity of benzyl alcohol mortality is seen at dose of down to 100 mg/kg body weight/day for premature babies. Here the dose was given intravenously. If swallowed benzyl alcohol is 25 times less acute toxic. In tests with mice a NOAEL value of 188 mg/kg body weight/day is stated where the acute toxicity at higher levels was lethargy. The critical impact of benzyl alcohol is thus lethargy (NOAEL value 188 mg/kg body weight/day). This value is used in the risk assessment despite the fact that mortality is seen at intravenous influence at lower doses as the acute effect of benzyl alcohol intravenously is 25 times higher than if swallowed.
6.3 Preservatives
6.3.1 Sodium benzoate
Sodium benzoate is the sodium salt of benzoic acid which is also preserving (WHO, 2000). Sodium benzoate is transformed into benzoic acid in acidic environments. In general benzoic acid and sodium benzoate are assessed together in one health assessment – and this is also the case for the fragrance/preservative benzyl alcohol which is described above. Sodium benzoate and benzoic acid belong to a comparatively similar chemical group and benzyl alcohol can be oxidated into benzoic acid. Therefore, the three substances have similar health properties (OECD SIDS, 2001).
Restrictions of sodium benzoate in relation to cosmetic products are set as a maximum limit for “salts of benzoic acid” where the maximum permissible content in percentages is calculated as benzoic acid (Stat. Ord. 422, 2006). Therefore the physical chemical parameters are stated for both sodium benzoate and benzoic acid.
Occurrence and application
In the mapped cosmetic products for children sodium benzoate occurs in 13 of the 208 products, distributed on five tooth pastes, three shampoos, two bobble bath products, one body shampoo/bath gel, one solid soap and one balsam.
OECD estimates that yearly 75,000 tons of sodium benzoate are produced worldwide. The majority[13] is applied as a preservative in food products and beverages and a large part is applied as rust-preventative agent, especially as an additive to anti-freeze fluid for cars or other coolant. Sodium benzoate is applied for many other purposes too, such as stabilizer in photo processing, in medical drugs, in plastic as for instance polypropylene to improve strength and brightness, as well as in cosmetic products. In most applications the primary function is preservative (OECD SIDS, 2001; WHO, 2000).
Sodium benzoate is applied as preservative (E211 – “Atamon”) in marmalade, jam, juice, vegetables in pickle, soft drinks, fish products such as marinated herrings and caviar, surface treatment of dried meat products, fluid egg products etc. Sodium benzoate occurs naturally in fruits and berries. The only fruits which have a natural content of sodium benzoate of importance are cranberries and cloudberries. They contain between about 600 and 2,000 mg/kg (Biosite, 2006; the E number guide, 2004; the Danish Veterinary and Food Administration, 2004).
According to the Positive list the content of benzoic acid and its salts must not be more than 6 g/kg in special crustaceans and mollusks. Other examples of restrictions for benzoic acid in food products are 200 mg/l in juice, non-alcoholic beer and alcohol (below 15% alcohol), 500 mg/kg in jelly, dried fruit butter and marmalade, 600 mg/l in fluid fruit tea, 1 g/kg in spices, 1.5 g/kg in food products for special medical purposes, chewing gum and slimming diet products, 2 g/kg in diet supplement and fish, and 5 g/kg in fluid egg products (The Positive list, 2005).
HSDB gives an example of a concentration of sodium benzoate of 0.125% in a body lotion (HSDB).
According to JECFA (Joint FAO/WHO Expert Committee on Food Additives) we consume between 0.18 and 2.3 mg benzoate per kg body weight per day. The low value is from Japan and the high value illustrates the daily intake in the USA (WHO, 2000).
Identification
| Chemical name | Sodium benzoate | Benzoic acid |
| Synonyms | Benzoic acid, sodium salt Antimol Benzoate of soda |
Benzenecarboxylic acid Benzenemethanoic acid |
| INCI name | Sodium benzoate | Benzoic acid |
| CAS-No. | 532-32-1 | 65-85-0 |
| EINECS No. | 208-534-8 | 200-618-2 |
| Gross formela | C7H5NaO2 | C7H6O2 |
| Molecular structure |  |
 |
| Legislation: The list of dangerous substances (Stat. Ord. 923, 2005) The list of harmful substances (Orientation from the Danish PA no. 8, 2004) The Danish EPA’s self classification (Environmental project no. 635, 2001) |
No No No |
No No No |
| Cosmetics Statutory Order (Stat. Ord. 422 , 2006) |
Salts of benzoic acid are permitted in cosmetic products but in a maximum concentration of 0.5% ranked as acid (Appendix 5, serial number 3m VI, 1, 1). | |
Physical chemical properties
Information about the physical chemical properties of sodium benzoate is found via Chemfinder, the ChemIDPlus database of TOXNET, a report from OECD SIDS on benzoates and via safety data sheet on sodium benzoate (OECD SIDS, 2001; Malinckrodt Chemicals, 2004; Safety Emporium, 2004).
| Sodium benzoate | Benzoic acid | |
| Physical structure | White crystallic powder | White crystallic powder |
| Molar weight (g/mol) | 144.11 | 122.12 |
| Melting point | 331 °C | 122 °C |
| Boiling point | 465 °C | 249 °C |
| Steam pressure | < 0.001 hPa | 0.0004 mmHg at 25 °C |
| Octanol water distribution (log POW) | -2.27 | 1.87 |
| Water solubility | 556 g/l at 25 °C | 3,4 g/l at 25 °C |
Absorption and disribution
Sodium benzoate is easily absorbed in the gastrointestinal tract. Sodium benzoate is expected to ionize immediately to benzoic acid in acid environments and also in the stomach. The benzoic acid is bound to the amino acid glycine and is mainly separated as hippuric acid in the urine within 24 hours (Nair B, 2001; OECD SIDS, 2001).
Acute toxicity
WHO has set a limit for the acceptable daily intake (ADI) of sodium benzoate of 5 mg/kg/day. In general US FDA (US Food and Drug Administration) regards sodium benzoate as being safe for apply in food products (Nair B, 2001).
There is a limited amount of data on the acute toxicity of sodium benzoate and the available studies are not completed according to the present guidelines (WHO, 2000). The found LD50 values for sodium benzoate are stated in Table 6.2. The stated values mean that sodium benzoate shall not be classified as harmful to health if swallowed.
Table 6.2: Acute toxicity (LD50) of sodium benzoate
| Species | Exposure | LD50 (mg/kg bw) |
Reference |
| Rat | Oral | 3140 | Loeser, 1977. |
| Rat | Oral | 4070 | Smyth & Carpenter, 1948. |
| Rat | Oral | 2100* | Deuel HJ et al, 1954 |
| Rat | Oral | 3450* | Deuel HJ et al, 1954 |
* Calculated as benzoic acid
The primary effect at intake of moderate amounts of benzoates is irritation of gastrointestinal systems. Intake of large amounts of benzoates (> 1 mg/kg body weight/day) can cause low pH level in the blood (acidosis) (HSDB).
Local irritation
Sodium benzoate is not assessed to be irritating on skin but slightly irritating for the eyes (OECD SIDS, 2001; WHO, 2001).
Allergy
There are reported cases of urticaria, asthma, rhinitis (a year-round cold) and anaphylactic shock (intense allergic response with skin rash and breathing difficulties) after oral exposure, skin exposure or inhalation of sodium benzoate (WHO, 2000).
Sodium benzoate is not skin sensitizing in tests with animals but in humans exposed to patch tests there is a low incidence of skin sensitization (5 of 2,045 cases (0.2%) were positive) (OECD SIDS, 2001; WHO, 2000). It is estimated that there shall be an exposure of 50 mg benzoate at one time to provoke an allergic response at sensitive persons (The Danish Veterinary and Food Administration, 2004).
Prolonged, repeated impact and gene damaging effects
In long-term tests with rats and mice, the impacts were limited to be reduced food intake and reduced growth. No damaging impacts to reproduction or carcinogenic impacts of sodium benzoate are observed in tests with rats and mice.
In the literature, different NOAEL values of 1000, >1090, 1310 and 3145 mg/kg bw/day are stated (Deuel HJ et al, 1954; Onodera et al, 1978; Sodemoto & Enomoto, 1980; IUCLID, 2000c; SCCNFP 0532, 2002). The highest value is the oldest.
There are no reports about serious side-effects after therapeutic use of sodium benzoate at doses of between 250 and 500 mg/kg bw/day in humans even if a few cases of anorexia and vomiting have been observed (WHO, 2000).
The Scientific Committee on Cosmetic Products and Non-Food Products (SCCNFP) has assessed whether it is safe to use benzoic acid and its salts and esters for non-preserving purposes in a maximum concentration of 2.5% in cosmetic products which are rinsed off after use. SCCNFP concluded that the data were not sufficient to be able to conduct a risk assessment of benzoic acid and sodium benzoate for these non-preserving purposes in cosmetic products (SCCNFP0532, 2002).
Sodium benzoate has shown not to be mutagenic in Ames tests and other tests (Nair B., 2001; OECD SIDS, 2001; WHO, 2000).
Critical impact
The critical impact of sodium benzoate is assessed to be irritation of gastrointestinal system if swallowed. No exact NOAEL value for this impact is found but the NOAEL value is between 250 and 500 mg/kg body weight/day even if a single case of vomiting is seen here.
6.3.2 Kathon
Kathon is the commercial name and a collective designation for the two substances methylchlorisothiazolinone and methylisothiazolinone which are often abbreviated CMI and MI respectively. According to the Cosmetics Statutory Order (Stat. Ord. 422 §25, 2006) the highest permissible concentration of Kathon in cosmetic products is 0.0015% of a mixture in the ratio 3:1 of methylchlorisothiazolinone and methylisothiazolinone.
When Kathon is applied in the CMI/MI ratio 3:1 in cosmetic products it is also this mixture that is primarily assessed in the health assessment. Most studies have also been carried out on the mixture and not on the individual isothiazolinones.
Earlier a health assessment of Kathon in Environmental Project No. 615 from the Danish Environmental Protection Agency 2001 has been conducted. This health assessment has been updated with new data (Madsen T et al, 2001).
Occurrence and application
Kathon is applied in cosmetic products as preservative. Kathon is applied in both “leave-on” products (primarily body lotions and other kinds of creams) and “rinse-off” products, such as shampoo and body shampoo (SCCNFP 0625, 2003).
In the studied cosmetic products for children Kathon is in 15 of the 208 products, distributed on eight body shampoo/bath gel products, two shampoos, two bobble bath products, one liquid soap, one eau de toilette and one body lotion/cream.
Identification
In the table below the chemical identification of methylisothiazolinone, methylchlorisothiazolinone and a mixture of these two substances are summarized.
Table 6.3: Identification of Kathon
| Chemical name | 2-Methyl-4-isothiazolin-3-one | 5-Chloro-2-methyl-4-isothiazolin-3-one | Chloro-2-methyl-3(2H)-isothiazolone with 2-methyl-3(2H)-isothiazolone |
| Synonyms | 2-Methyl-3(2H)-isothiazolone 2-Methyl-2H-isothiazol-3-one 2-Methyl-2,3-dihydroisothiazol-3-one MI |
5-Chloro-2-methyl-2H-isothiazol-3-one 3(2H)-Isothiazolone, 5-chloro-2-methyl- Kathon CG 5243 Kathon DP Kathon LX Kathon UT Kathon WT MCI CMI |
Kathon 886 Kathon 886 W Kathon 886MW Kathon CG Kathon CG/ICP II Kathon LX Kathon RH 886 Kathon WT Kathon biocide MCI/MI CMI/MI |
| INCI name | Methylisothiazolinone | Methylchloroisothiazolinone | - |
| CAS No. | 2682-20-4 | 26172-55-4 | 55965-84-9 |
| EINECS No. | 220-239-6 | 247-500-7 | - |
| Gross formula | C4H5NOS | C4H4ClNOS | |
| Molecular structure |  |
 |
 |
| Classification according to the List of dangerous substances (Stat.Ord. 923, 2005) The List of unwanted substances (Orientation from the Danish EPA no. 8, 2004) Danish EPA Self-classification (Environmental project no. 635, 2001) The Cosmetics Statutory Order (Stat. Ord. 422 , 2006) |
No No R43 Highest permissible concentration of 0.0015% (15 ppm) (of a mixture in the ratio 3:1 of 5- chlor-2-methyl-4- isothiazolin- 3-on and 2- methyl-4-isothiazolin-3-on). |
No No R43 Highest permissible concentration of 0.0015% (15 ppm) (of a mixture in the ratio 3:1 of 5- chlor-2-methyl-4- isothiazolin- 3-on and 2- methyl-4-isothiazolin-3-on). |
T;R23/24/25 C;R34 R43 N;R50/53 In concentrations above 0.0015%, but below 0.06% the classification is Xi, R43 No No Highest permissible concentration of 0.0015% (15 ppm) (of a mixture in the ratio 3:1 of 5- chlor-2-methyl-4- isothiazolin- 3-on and 2- methyl-4-isothiazolin-3-on). |
Physical chemical properties
In the table below the physical chemical properties of Kathon are summarized. The information is based on information from SCCNFP (SCCNFP 0805, 2004) and (SCCNFP 0625, 2003) as well as information from the IUCLID database which only covers the properties for the mixture of the two substances.
Table 6.4: Physical chemical properties of Kathon
| Methylisothiazolinone | Methylchlorisothiazolinone | Kathon (the mixture) | |
| Physical structure | Raw white or light brown solid substance at room temperature | Fast stof | |
| Molar weight (g/mol) | 115.16 | 149.59 | |
| Density | 1,35 g/cm³ at 25 °C | 1,2556 g/cm³ at 20 °C | |
| Melting point | 46,7 – 48,3 °C | Ca. 50-55 °C | |
| Boiling point | Thermal decomposition at 155 °C | 106,5 °C at 1000 hPa | |
| Steam pressure | 0,73 Pa at 20 °C | 20,8 hPa at 20 °C | |
| Octanol water distribution (log POW) | -0,486 at 24 °C | -0,71 – 0,75 at 20 °C | |
| Water solubility | > 1000 g/l at 20 °C | > 5000 mg/l at 20 °C | |
| Flame point | > 100 °C |
Absorption and distribution
Kathon is readily excreted after absorption in the body. After oral administration of Kathon 886 to rats, the majority of MI and CMI was excreted without troubles with the urine or faeces (CIRP, 1992). Tests with C14 labelling of the chlorinated compound (CMI) have shown that more than 50% is excreted with faeces or the urine after 24 hours and about 70% (faeces 35%, urine 31% and CO2) after 96 hours (Debethizy et al, 1986).
Kathon 886 is distributed quickly to blood, liver, kidneys and testicles after an intravenous dose (0.8 mg/kg bw) (CIRP, 1992).
The half-life period for dermally absorbed compounds is found to be 13.1 days. This indicates an increased potential for accumulation in the body at repeated application or use (Conner et al, 1996).
Acute toxicity
Kathon (CMI og MI) is from moderate to very poisonous when swallowed (Table 6.5). Signs of toxicity are among others drowsiness, ataxia (loss of full control of body mobility) and serious gastric irritation. Signs of intoxications at doses from 75 mg MI/kg body weight/day are seen in female rats (150 mg MI/kg body weight/day for male rats) (Rohm and Haas, 1999; Rohm and Haas, 2000a; SCCNFP 0625, 2003; CIRP, 1992).
In vitro tests with normal human ceratinocyte cell cultures showed that a short exposure (10 minutes) with CMI/MI in concentrations between 0.001% and 0.1% causes a cytotoxic (cell-destroying) impact which is dependent on concentration (Ettorre A et al, 2003).
Table 6.5: Acute toxicity (LD50) of Kathon
| Substance | Species | Exposure | LD50 (mg/kg bw) |
Reference |
| MI | Rat | Oral | 183-235 | Rohm and Haas, 1999 |
| MI | Rat | Oral | 106-275 | Rohm and Haas, 2000a |
| MI | Rat | Dermal | > 485 | Rohm and Haas, 2000b |
| Neolone 950 (calculated as MI) |
Rat | Oral | 104 - 269 | Rohm and Haas, 2004b |
| Neolone 950 (calculated as MI) | Rat | Dermal | > 475 | Rohm and Haas, 2004b |
| Kathon CG (calculated as CMI/MI total) | Rat | Oral | 39,5-50,3 | Rohm and Haas, 2004a |
| Kathon CG (calculated as CMI/MI total) | ? | Dermal | 75 | Rohm and Haas, 2004a |
| CMI | Rat | Dermal | > 1008 | IUCLID, 2000a |
| CMI | Rat | Oral | 481 | IUCLID, 2000a |
| CMI | Rat/moapply | Oral | 53-60 | DFG, 1993 |
| Kathon CG | Rat | Oral | 3350* | CIRP, 1992 |
| CMI | Rabbit | Dermal | 80 | DFG, 1993 |
* Suggests that the value is of the sold product Kathon CG and is not calculated for the total content of CMI/MI.
Local irritation
Kathon is in its pure form corrosive for both skin and eyes. Solutions containing more than 0.5% active isothiazolinones are very skin irritating on humans and can cause corrosion of mucous membranes and corneas (DFG, 1993; CIRP, 1992).
It also applies to the two individual substances in Kathon that they individually are corrosive in pure form. MI is neither skin nor eye irritating (on rabbits) at 100 ppm (0.01%) active constituent. No found data indicate how irritating CMI is in undiluted form (Rohm and Haas, 2000d; IUCLID, 2000a; Rohm and Haas, 2000e).
Allergy
The sensitizing potential for Kathon (CMI/MI) for humans is well-described in the literature. There is a general agreement that CMI/MI is a sensitizer. At the beginning of the 1980’ies CMI/MI was introduced as a preservative in cosmetics and gave cause for an empidemic of allergy cases in Europe. The many cases of allergy have led to limitation of the use of CMI/MI. In 2005 1.6% of the eczema patients in Denmark were allergic to CMI/MI. On the whole this figure is unchanged from 2003 and 2004 and CMI/MI is still considered as one of the most frequent causes for preservative allergy in Denmark (Denmark’s National Allergy Research Centre, 2006b). The picture in Germany was the same when CMI/MI was introduced as a preservative in the 1980’ies. Here 5.2% of the patients were allergic to CMI/MI. Since reduction in the applied concentration of CMI/MI has halved this figure (Alexander BR, 2002).
In a memorandum to ECB (European Chemicals Bureau) on CMI/MI three tests with patch tests on children are described. The studies carried out in Norway (in 1991), in Germany (in 1990-1995) and in Italy (in 1988-1994) show that 5.2%, 1.1% and 5.7% respectively reacted positively to Kathon CG. The studies demonstrate that Kathon CG is sensitizing and that occupational exposure might be excluded. Kathon from cosmetic products is the most probable cause for the sensitization. In the same memorandum a survey in Japan is described where 0.9% reacted positively to a patch test in the period 1989 to 1992 and this despite the fact that Kathon CG is only applied in products being rinsed after use and in a maximum concentration of 15 ppm. A concentration of 15 ppm in products being rinsed after use represents thus a risk for sensitization of Kathon CG (ECB, 1999). Today Kathon is permitted in a maximum concentration of 15 ppm but also in product with the purpose of staying on the skin.
Dermatological studies have shown isothiazolone concentrations below 20 ppm may cause sensization and that allergic responses can be provoked in already sensitized persons with concentrations of 7-15 ppm (DFG, 1993).
It is primarily CMI which is the sensitizing substance in Kathon (CMI/MI) but MI has also sensitizing properties. SCCNFP (Scientific Committee on Cosmetic Products and Non-Food Products) assesses that there is at least a factor 30 in difference between the mixture CMI/MI and MI in the sensitizing potential of the two products (SCCNFP 0625, 2003). ECB (2002b) and characterizes the mixture CMI/MI as an extreme sensitizer and MI as a strong sensitizer based on the EC3 values[14] which are found in LLNA tests (Local Lymph Node Assay).
ECB’s Working Group on Sensitisation has set up a treshold limit of sensitization for Kathon CG of < 3 ppm based on several published Guinea Pig Maximazation Tests (ECB, 2002a). Correspondingly, Alexander (2002) has set up a threshold limit of sensitization for CMI/MI of 58 ppm based on animal tests and 10-20 ppm based on human tests. Regarding MI the corresponding threshold value of sensitization is 800 ppm, based on animal tests. The highest tested dose of the mixture CMI/MI not being sensitizing is found to be 36 ppm and 500 ppm for MI in animal tests (Alexander BR, 2002).
An article in the weekly journal of the Danish Medical Association describes that persons with known Kathon allergy got severe allergic responses when being in a room painted with water-based plastic paint preserved with Kathon (30 ppm). It is unknown whether the Kathon responses are due to inhalation or skin contact with Kathon in the air (The weekly journal of the Danish Medical Association, 2002).
Prolonged, repeated impact and gene damaging effects
In long-term tests with rats no effects are seen in oral tests with MI alone. In similar long-term tests with rats only a slight gastric irritation for the Kathon mixture is seen (Kathon 886) (Rohm and Haas, 2000c; CIRP 1992).
No damaging impacts on reproduction or carcinogenic impacts of Kathon are found in the relatively few tests which are referred in the literature (CIRP, 1992). No information about the damaging potential on reproduction or carcinogenic potential of the two individual substances MI and CMI is found.
In a long-term test where rats were exposed to MI via the drinking water there was no mortality at any of the doses. No treatment related impacts on body weight and food intake at up to the second-highest dose were found. Based on this test a NOAEL of 250 ppm active constituent was set up, equivalent to 19.0 and 24.6 mg a.i./kg bw/day for male and female rats respectively for MI (Rohm and Haas, 2000c). In view of this NOAEL the Scientific Committee on Cosmetic Products and Non-Food Products (SCCNFP) has assessed that MI (as single substance) will not present a risk for the consumers’ health when used as preservative in cosmetic products in a maximum concentration of 100 ppm (0.01%) (SCCNFP 0805, 2004).
A three months’ test on rats with CMI via dermal exposure showed only weak skin responses at the highest dose of 18.75 mg/kg/day (IUCLID, 2000a).
For the Kathon mixture (Kathon 886) a three months’ test on rats with oral intake via the drinking water showed only a slight gastric irritation at a dose of 20 mg/kg/day. NOEL was set to 8 mg/kg/day (CIRP, 1992).
Kathon mixtures (Kathon CG and Kathon) have been through a number of tests regarding the mutagenic properties. Several tests show that Kathon is mutagenic in Ames test whereas there are negative results in studies investigating the DNA damaging potential of Kathon (Madsen T et al, 2001). Similarly, the single substance CMI is tested mutagenic according to Ames test (IUCLID, 2000a) whereas MI does not show any signs of mutagenicity in test with bacteria. Based on in vitro tests (in vitro) SCCNFP assesses MI to be clastogenic. That means it may cause damages or changes on chromosomes (SCCNFP 0625, 2003).
Critical impact
The critical impact of Kathon if swallowed is ataxia and serious gastric irritation. NOEL value coming from a test at oral intake through drinking water is found to 8 mg/kg body weight/day. This value is used in the risk assessment of Kathon as it is the lowest value where impacts of Kathon are seen.
In the total scenario of the toxicity of Kathon the attention is drawn to the point that Kathon is assessed to be sensitizing. ECB’s Working Group on Sensitisation has set a threshold value for Kathon CG of < 3 ppm based on animal tests. Others state a threshold value for Kathon of 10-20 ppm based on human tests.
6.3.3 Phenoxyethanol
Occurrence and application
OECD has investigated the production of phenoxyethanol and has not identified more than one American producer. In 1999 6,400 tons phenoxyethanol were produced in the USA. A large number of applications of phenoxyethanol is reported in both the USA and Europe (see the table below). Paints, detergents and dyes seem to be the largest application areas (OECD SIDS, 2004). In cosmetics phenoxyethanol is used as a preservative (INCI, 2006).
The OECD report estimates that about 60% of the produced amount of phenoxyethanol is used in consumer products (OECD SIDS, 2004).
In the mapped cosmetic products for children phenoxyethanol is found in 50 of the 208 products, distributed on 22 body shampoo/bath gel, 10 shampoos, eight booble bath products, six body lotion/cream, two hairstyling products, one balsam and one facial cream. Thus phenoxyethanol is one of the most used preservative in the mapped products. Only methyl paraben and propyl paraben are more frequently used (in 79 and 70 of the mapped products respectively).
Table 6.6: Application areas reported for phenoxyethanol
| USA | Europa |
| Solvent for cellulose acetate Dyes Printing inks Resins Organic synthesis of plasticizers Germicides Pharmaceuticals Preservative for human anatomical specimens for dissection Fixative for perfumes As bactericide and insect repellant in organic synthesis Fixative for cosmetic products Textile dye carrier Chemical intermediate product Solvent for detergents Solvent for frimærkepuder Specialty inks and ball points |
Detergents Paints and varnishes Cutting fluids Non-agricultural pesticides and preservatives Reprographic substances Surfactant Cosmetic products plasticizer Glue and binder Surface treatment Welding and soldering agents Pharmaceuticals Colouring agents corrosion inhibitor Lubricant Stabilizer Solvent |
Identification
| Chemical name | 2-phenoxyethanol |
| Synonyms | 1-Hydroxy-2-phenoxyethane 2-Hydroxyethyl phenyl ether 2-Phenoxyethyl alcohol Ethylene glycol phenyl ether Phenoxethol Phenoxyethanol Phenoxyethyl alcohol Phenyl cellosolve Phenylmonoglycol ether Rose ether |
| INCI name | Phenoxyethanol |
| CAS No. | 122-99-6 |
| EINECS No. | 204-589-7 |
| Gross formula | C8H10O2 |
| Molecular structure |  |
| Legislation: The List of dangerous substances (Stat. Ord. 923, 2005) The List of unwanted substances (Orientation from the Danish EPA no. 8, 2004) Danish EPA Self-classification (Environmental project no. 635, 2001) The Cosmetics Statutory Order (Stat. Ord. 422 , 2006) |
XN; R22 XI; R36 R22 = Dangerous if swallowed. R36 = Irritates the eyes. No No Is permitted as a preservative in cosmestics products in a maximum concentration of 1.0%. (Appendix 5, serial number 39 VI, 1, 29). The substance may be used in other concentrations and for other purposes than preservation, for instance as deodorant in soaps or in dandruff hair shampoo. In such cases the purpose of the use of the substance must appear from the product itself. |
Physical chemical properties
Data on the physical chemical properties of phenoxyethanol are found via Chemfinder, ChemIDplus database of TOXNET, OECD SIDS report on phenoxyethanol as well as IUCLID data sheet for 2-phenoxyethanol. (Chemfinder; TOXNET; IUCLID, 2000d; OECD SIDS, 2004).
| Physical structure | Clear, colourless, thick liquid |
| Molar weight (g/mol) | 138.17 |
| Melting point | 14 °C |
| Boiling point | 245 °C |
| Steam pressure | 0.013 hPa at 20 °C |
| Octanol water distribution (log POW) | 1.16 |
| Water solubility | 26,700 mg/l at 20 °C |
Absorption and distribution
Human skin absorption of phenoxyethanol is tested in vitro and shows that about 60% of the substances is absorbed after 6 hours (Roper et al, 1997). OECD notes that it is uncertain whether the apply af methanol as a solution has had an impact on the absorption of phenoxyethanol in one or another direction (OECD SIDS, 2004).
Tests with rats show that more than 75% and up to 99% of the phenoxyethanol after either oral or dermal exposure can be found unchanged in the urine together with small quantities of two substances to which the phenoxyethanol has metabolized. One of the metabolism products is phenoxyacetic acid (Breslin et al, 1991).
Acute toxicity
Symptoms of poisoning with phenoxyethanol in animal tests are a slight to more serious reduction in activity, weakness, reflex impairment and breathing difficulties. At high doses comatose before death or recovery is seen and also lethargy, ataxia (loss of full control of bodily movements) and body tremors at high doses are seen. No abnormalities are found in the animals which died (Hill Top Research, 1980; Richold et al, 1982).
The found LD50 values (Table 6.7) support the classification of phenoxyethanol as harmful to health if swallowed.
Table 6.7: Acute toxicity (LD50) of phenoxy ethanol
| Species | Exposure | LD50 (mg/kg bw) |
Reference |
| Rat | Oral | 1386 - 2563 | Hill Top Research, 1980 |
| Rat | Oral | 2937 - 4013 | Gollapudi et al, 1988 |
| Rat | Oral | 1260 | Smyth et al, 1941. |
| Rat | Dermal | 14.300 | Davies, 1970 |
Local irritation
Phenoxyethanol is not skin irritating for humans but irritating for eyes in animal tests with rabbits. Phenoxyethanol must also be classified Xi R36 ”Irritates the eyes” (IUCLID, 2000d; OECD SIDS, 2004; BEK 923, 2005).
A survey of 2,736 patients who underwent patch tests with 1% phenoxyethanol in petrolatum showed that none of the patients had signs of irritating effects two or four days after exposure (Lovell et al, 1984).
Allergy
Both human data and data from animal tests show that phenoxyethanol is not sensitizing. A survey with patch tests showed only one positive response to phenoxyethanol (5% in petrolatum) corresponding to 0.2% out of the 501 patients (IUCLID, 2000d; De Groot et al, 1986). In another survey with patch test with 5% phenoxyethanol in petrolatum on 3,726 patients, only one patient showed allergic responses (CIR Review, 1990a). In the above-mentioned survey of 2,736 patients with patch tests none of the patients got allergic responses (Lovell et al, 1984).
Prolonged, repeated impact and gene damaging effects
No studies regarding the carcinogenic properties of phenoxyethanol are found but the Danish Environmental Protection Agency has applied FDA’s cancer models on phenoxyethanol with a negative result (OECD SIDS, 2004). This means that phenoxyethanol is not expected to be carcinogenic.
Phenoxyethanol has shown damaging impacts on reproduction and developmental toxicity in animal tests with mice. In several reproduction studies with mice the impacts were decreasing body weight on the mice and their progeny as well as increased liver weight at high doses of between 1,875 and 4,000 mg/kg bw/day. At the highest dose of between 3,700 and 4,000 mg/kg bw/day, phenoxyethanol showed damaging impacts on reproduction in the form of decrease in both the number of young per litter and young born alive (NTP, 1984; Heindel et al, 1990; OECD SIDS, 2004).
Heindel et al. (1990) states a NOAEL for impacts on both parents and foetus of 400 mg/kg bw/day in reproduction studies with mice. A 90 days’ repeated test with oral exposure of rats with the doses 80, 400 and 2,000 mg/kg bw/day showed no impacts at 80 mg/kg bw/day (NOAEL value). At 400 mg/kg bw/day kidney toxicity and changes in grooming behavior were seen. At a dose of 2,000 mg/kg bw/day toxicity towards red blood corpuscles was seen (Ben-Dyke et al, 1977).
A 90 days’ repeated test with skin exposure on rabbits showed no impacts besides erythema of the skin on the test spot at doses of 50 to 500 mg phenoxyethanol/kg bw/day. From this a NOAEL for systemic toxicity of 500 mg/kg bw/day is derived (Breslin et al, 1991).
Phenoxyethanol has shown not to be mutagenic in the Ames test. Other gene mutation tests were also negative (IUCLID, 2000d; OECD SIDS, 2004).
The recommendations in an OECD SIDS report on phenoxyethanol are that the substance is a candidate for future work regarding human health as the substance has properties indicating a hazard for human health (eye irritation and developmental toxicity at high doses which also gives maternal toxicity) (OECD SIDS, 2004).
Critical impact
The critical impact of phenoxyethanol is assessed to be kidney toxicity if swallowed. The NOAEL value is found to 80 mg/kg body weight/day.
6.3.4 5-bromo-5-nitro-1,3-dioxane
Occurrence and application
5-bromo-5-nitro-1,3-dioxane is used as a stabilizer, surfactant and bactericide, and in the immunology it is used to preserve antibodies in concentrations of between 0.1 and 0.5% (Wikipedia, 2006). According to the SPIN database 5-bromo-5-nitro-1,3-dioxane is used in detergents, in cosmetic products and for surface treatment in Denmark (SPIN2000).
Since the middle of the 1970’ies 5-bromo-5-nitro-1,3-dioxane has been used in cosmetic products as a preservative in shampoos, bobble bath etc. (Wikipedia, 2006). According to the Cosmetics Statutory Order the permitted maximum concentration is 0.1% and only in products which are cleansed after apply (Stat. Ord. 422, 2006).
In the mapped cosmetic products for children 5-bromo-5-nitro-1,3-dioxane occurs in 15 of the 208 products, distributed on 13 body shampoos/bath gels and two bobble baths.
Identification
| Chemical name | 5-Bromo-5-nitro-1,3-dioxan |
| Synonyms | 1,3-Dioxane, 5-bromo-5-nitro- 5-Bromo-5-nitro-m-dioxane Bronidox |
| INCI name | 5-Bromo-5-nitro-1,3-dioxane |
| CAS No. | 30007-47-7 |
| EINECS No. | 250-001-7 |
| Gross formula | C4H6BrNO4 |
| Molecular structure | 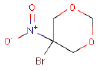 |
| Legislation: The List of dangerous substances (Stat. Ord. 923, 2005) The List of unwanted substances (Orientation from the Danish EPA no. 8, 2004) Danish EPA Self-classification (Environmental project no. 635, 2001) The Cosmetics Statutory Order (Stat. Ord. 422 , 2006) |
No No Xn; R22 R43 R22 = Dangerous swallowed R43 = Might give allergic reactions at contact with the skin Is permitted as a preservative in cosmetic products in a maximum concentration of 0.1%. May only be used in products which are cleansed of after apply. Formation of nitrosamines shall be avoided. Compulsory user’s instruction and warning on the label is: “Contains formaldehyde” if the concentration in the end product is above 0.05%. |
Physical chemical properties
Data on the physical chemical properties of 5-bromo-5-nitro-1,3-dioxane are found in various safety data sheets on the Internet (ChemicalLand21.com, 2006), (Wikipedia, 2006), as well as The Danish Information Centre for Environment & Health (The Danish Information Centre for Environment & Health, 2004).
| Physical structure | White crystallic powder |
| Molar weight (g/mol) | 212.06 |
| Melting point | 60 °C |
| Boiling point | - |
| Steam pressure | - |
| Octanol water distribution (log POW) | 0.25 |
| Water solubility | Insoluble |
Acute toxicity
In general, only few data on the toxicity of 5-bromo-5-nitro-1,3-dioxane are available. According to the list of dangerous substances 5-bromo-5-nitro-1,3-dioxane shall not be classified as hazardous but according to the guiding list for self-classification of the Danish Environmental Protection Agency the substance is hazardous to health (dangerous if swallowed) (Orientation from the Danish Environmental Protection Agency no. 8, 2004).
Table 6.8: Acute toxicity (LD50) of 5-Bromo-5-nitro-1,3-dioxan
| Species | Exposure | LD50 (mg/kg bw) |
Reference |
| Rat | Oral | 590 | (Wikipeida, 2006) |
| Rat | Oral | 455 | (Fette, Seifen, Anstrichmittel, 1976) |
| Moapply | Oral | 455 | (Wikipeida, 2006) |
| Rat | Dermal | 2.5 mg/24 hours | (Wikipeida, 2006) |
In safety data sheets 5-bromo-5-nitro-1,3-dioxane is described as being dangerous if swallowed. Reactions of toxicity when swallowed are impacts on the central nervous system, such as tremor, convulsions and excitement (ScienceLab, 2005; Wikipedia, 2006).
Local irritation
5-bromo-5-nitro-1,3-dioxane is skin irritating and irritating for the eyes in concentrations above 0.1% (ScienceLab, 2005; Wikipedia, 2006; CIR Review, 1990b).
Allergy
5-bromo-5-nitro-1,3-dioxane is assessed to be sensitizing for human even if the substance is not sensitizing in studies with guinea pigs (CIR Review, 1990b; Madsen T et al, 2001). According to the guiding list for self-classification of the Danish Environmental Protection Agency, 5-bromo-5-nitro-1,3-dioxane is allergenic and therefore the substance ought to be labelled with R43 “Might cause allergic reactions at contact with the skin (Environmental project no. 635, 2001).
5-bromo-5-nitro-1,3-dioxane is a so-called formaldehyde releaser, this means that the substance separates out formaldehyde. This is the way that 5-bromo-5-nitro-1,3-dioxane is preserving as formaldehyde is a preservative. According to the Danish Knowledge Centre for Allergy, about 2% of the persons with eczema are allergic towards formaldehyde (The Danish Knowledge Centre for Allergy, 2006c).
Prolonged, repeated impact and gene damaging effects
Only a little information about the carcinogenic, damaging to reproduction and gene damaging effects is available but according to the little found information 5-bromo-5-nitro-1,3-dioxane is neither carcinogenic, damaging to reproduction nor mutagenic (negative Ames test), (Roche Applied Science 2005; CIR Review, 1990b).
5-bromo-5-nitro-1,3-dioxane can react with amines and amides and form nitrosamines or nitrosamides which are assessed to be carcinogenic and may penetrate the skin (Madsen T et al, 2001, US FDA). Special guidelines regarding this substance are found in the Cosmestics Statutory Order stating that formation of nitrosamines should be avoided (Stat. Ord. 422, 2006). In practice this means that 5-bromo-5-nitro-1,3-dioxane must not be mixed with amines and amides in cosmetic products. Furthermore, the substance is only permitted to be used in cosmetic products being cleansed after use.
Critical impact
Detailed information about 5-bromo-5-nitro-1,3-dioxane is missing in order to assess the critical impact of the substance but there is no doubt that contact allergy at skin contact is a substantial impact.
7 Exposure assessment
Exposure assessments on Kathon and benzyl alcohol are conducted as an exact determination of the concentration in the analyzed products is available for these two substances. Quantitative analyses of the constituents phenoxyethanol, sodium benzoate or 5-bromo-5-nitro-1,3-dioxane are not undertaken in the same way and therefore no exposure assessment will be conducted for these substances.
The guidelines for the exposure assessment are stated in EU’s Technical Guidance Document (TGD) (European Commission, 2003) and SCNNFP’s guidelines (SCCNFP 0690, 2003).
However, no standard weight for a child is stated in the TGD. Children’s weight for a certain age can be found in official growth curves. Netdoktor.dk has a table of girls’ and boys’ weight (see extract below) dating from an older Scandinavian study. They emphasize that it is an older study and that in general children have become a little taller and heavier since then (Netdoktor, 2006).
Table 7.1: Overview of Scandinavian children’s weight (Netdoktor, 2006).
| The child’s age | Girls’ average weight | Boys’ average weight |
| 3 years | 15.0 kg | 15.4 kg |
| 6 years | 20.8 kg | 21.2 kg |
| 9 years | 27.5 kg | 28.5 kg |
| 12 years | 39.0 kg | 37.5 kg |
| 14 years | 50.0 kg | 48.0 kg |
The weight from 8 years and up must be taken with a reservation as the weight varies much in the puberty.
The target group of this project is children at the age 3-14 years. Worst case is thus a three-year-old girl’s weight, i.e. 15 kg. For a few situations, for instance bath situations where little brother or sister bathes together with the older siblings, it may be considered to apply an even lower weight as worst case.
In all exposure scenarios a child of 15 kilos is applied as a standard equivalent to an age of three years which is the lower age in the target group of the cosmetic products for children being studied in this project.
A standard MoS (Margin of Satety) of 100 is applied for the risk assessment. It is generally accepted that MoS must be at least 100 in order that a substance can be declared as safe to apply. If a credible animal study is available it can be justified to use a factor 10 for differences in kind (animals to humans) and a factor 10 to include sensitive groups in the population – in total a factor 100.
SCCNFP concludes in their guidelines for safety assessment of cosmetics constituents that there is no reason to apply an additional safety factor for children when talking of intact skin (SCCNFP 0690, 2003).
7.1 Kathon
In total 11 products are analyzed for a quantitative content of Kathon. The table below states the intervals of the measured concentrations of Kathon in the different product types being studied. Furthermore, the table is an overview of the product types in which Kathon is found via the study. The maximum permissible concentration of Kathon in cosmetic products is 15 mg/kg.
Table 7.2: Produkts with Kathon. The measured concentrations are stated for the analyzed products.
| Product type | Products analyzed | Products in total with Kathon |
Measured concentrations I mg/kg (ppm) |
| Body shampoo/bath gel | 6 | 9 | < 2 – 7.7 |
| Liquid soap | 1 | 1 | 10 – 12 |
| Bobble bath | 2 | 4 | 3.0 – 11 |
| Body lotion/cream | 1 | 1 | < 2 |
| Shampoo | 1 | 2 | 4.4 – 6.7 |
| Eau de toilette | 0 | 1 | No analyses conducted |
The detection limit is 2 mg/kg
For a cosmetics constituent, the daily exposure quantity – also called the internal exposure (in the EU called SED or Systemic Exposure Dosage) is the quantity of the substance being expected to penetrate into the bloodstream (and thus being systemic disposable) per kg body weight per day (SCCNFP 0690, 2003).
The calculated exposure scenarios for Kathon are worst case scenarios, calculated according to the guidelines and default values in TGD (Appendix II, Table 14) and retention factors from SCCMFP 0690 (2003). The retention factor is introduced by SCCNFP to consider products being diluted when they are used and cleansed off after use, i.e. for shampoo products, body shampoo and similar “rinse-off” products.
No data are available for liquid soap. Here the values from the consumer project “Survey and health and environmental assessment of hand soap”(Larsen JR et al, 2006) are applied. The factors being a part of the calculation are seen in Table 7-3.
Table 7-3. Applied factors in the exposure assessment.
| Product type | Number of appplications | Applied quantity per application | Concentra-tion of Kathon | Uptake through the skin | Retention factor | The child’s weight |
| Body shampoo/ Bath gel |
1-2 per day | 5.0 g | 7.7 mg/kg | 100% | 0.01 | 15 kg |
| Fluid soap | 6 per day | 1.0 g | 12 mg/kg | 0.01 | ||
| Bobble bath | 1-2 per week | 17.0 g | 11 mg/kg | 0.01 | ||
| Body lotion/cream | 1-2 per day | 7.5 g | 2 mg/kg | 1 | ||
| Shampoo | 2-7 per week | 12.0 g | 6.7 mg/kg | 0.01 | ||
| Eau de toilette | 1-2 per day1 | 3.0 g² | 15 mg/kg³ | 1 |
1 – assumption as no data in TGD or SCCNFP guidelines.
2 – the value for deodorant spray is applied. This value will be significantly higher than for eau de toilette (but there is no available value for eau de toilette in TGD).
3 – no eau de toilette products have been analyzed. Therefore, the maximum permissible concentration of 15 mg/kg is applied.
As seen in the table no data for eau de toilette are available. A density of 1 g/litre is assumed as eau de toilette mainly consists of water and about 8% fragrances according to the analyse of an eau de toilette in this project. It is assumed that eau de toilette is applied once per day. Typically, adults will have a use of 1-2 applications per day whereas very small children may be expected to apply significantly less. It may be assumed that the girls have a certain age before they start using eau de toilette. The older children can be expected to imitate the mother’s behaviour, i.e. 1-2 applications per day.
Regarding application quantity, no quantity is stated in TGD for eau de toilette. Instead the value for deodorant spray of 3.0 g per application is used. This value is probably significantly higher than the real value as experience shows that less eau de toilette than deodorant spray is used.
Correspondingly, no standard weight for a child is stated in TGD. As described in the beginning of the chapter, a worst-case value of 15 kg for a child’s weight is applied.
The daily exposure is calculated by use of the formula below where SED (Systemic Exposure Dosage) is the daily exposure, A is the quantity being applied daily, C is the concentration of the substance in the cosmetics product, DA (Dermal Absorption) is the absorption through the skin indicated in %, Rf is the retention factor (introduced by SCCNFP to consider “rinse-off” products) and bw (body weight) is the body weight of the child.
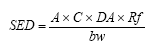
The daily exposure of Kathon when using body lotion on a three-year-old child can be calculated to:
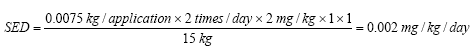
In the table below the daily exposure (SED) for Kathon is stated for the different product types.
Table 7.4: Daily exposure (SED) for Kathon for the different product types
| Applied quantity per application (in kg) | Number of daily applications | Concentration (mg/kg) |
Rf | SED (mg/kg/day) |
|
| Body shampoo/bath gel | 0.005 | 2 | 7.7 | 0.01 | 0.000051 |
| Liquid soap | 0.001 | 6 | 12 | 0.01 | 0.000048 |
| Bobble bath | 0.017 | 0.33 | 11 | 0.01 | 0.000041 |
| Body lotion/cream | 0.0075 | 2 | 2 | 1 | 0.0020 |
| Shampoo | 0.012 | 1 | 6.7 | 0.01 | 0.000054 |
| Eau de toilette | 0.003 | 2 | 15 | 1 | 0.0060 |
It is seen that the highest daily exposures occur when using eau de toilette and body lotion. It must be noted that the apply of eau de toilette is assessed to be much overestimated as the same exposure values as for normal deodorant are applied.
7.1.1 Risk assessment for Kathon
The risk assessment is conducted for the critical effect of Kathon which at acute toxicity is ataxia and serious stomach irritation. It is presumed that 100% of the Kathon to which the child is exposed will also be absorbed in the body. The applied NOAEL value is identified for Kathon in the health assessment in section 6.3.2, i.e. for the mixture MI/CMI. This value is 8 mg/kg bw/day CIRP, 1992).
When calculating Margin of Safety (MoS) for Kathon the NOAEL value and the calculated daily exposure (= SED) are placed in the following formula:
![]()
Margin of Safety for Kathon when using body lotion on a three-year-old child will be:
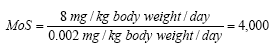
Table 7.5: Margin of safety for Kathon for the different product types
| SED (mg/kg/day) |
MoS | |
| Body shampoo/bath gel | 0.000051 | 155,844 |
| Liquid soap | 0.000048 | 166,667 |
| Bobble bath | 0.000041 | 194,458 |
| Body lotion/cream | 0.0020 | 4,000 |
| Shampoo | 0.000054 | 149,254 |
| Eau de toilette | 0.0060 | 1,333 |
Margin of Safety ought to be above 100 to take account of a safety factor of 10 for extrapolation of data from animals to humans and a safety factor of 10 to take account of especially sensitive human individuals.
The calculation shows that the margins of safety are substantially above 100 even if the calculation is made as a worst case calculation, i.e. for a three-year-old child with a high daily use of body lotion. However, it must be emphazied that MoS is not calculated for allergy but for another critical effect, cf. the health assessments in chapter 6.
7.2 Benzyl alcohol
In total 17 products are analyzed for a quantitative content of the 26 fragrances, including benzyl alcohol. Benzyl alcohol was found in 12 of the 17 analyzed products. The table below states the intervals of the measured concentrations of benzyl alcohol in the different product types being studied. Furthermore, the table is an overview of the product types in which benzyl alcohol is found via the study. The maximum permissible concentration of a preservative in cosmetic products is 1% according to the Cosmetics Statutory Order. As a fragrance there is no upper limit.
Through the analyses, more products with a content of benzyl alcohol are identified than found via the survey (applies to the product types body shampoo/bath gel and shampoo). This reflects the fact that there is benzyl alcohol in the products but in a low concentration not mandatory to declare on the product.
Table 7.6: Products with benzyl alcohol. The measured concentrations are stated for the analyzed products.
| Product type | Products with an analyzed content of benzyl alcohol | Products in total with benzyl alcohol | Measured concentrations in mg/kg (ppm) |
| Body shampoo/bath gel | 6 | 1 | 4 – 120 |
| Shampoo | 3 | 1 | 1 – 8 |
| Body lotion/cream | 1 | 1 | 780 – 790 |
| Eau de toilette | 1 | 1 | 5 |
| Bath confetti/caviar/fizzle salt | 1 | 6 | 2 |
| Bobble bath | - | 1 | No analyses conducted |
| Tooth paste | - | 1 | No analyses conducted |
| Hair dye (rinsing colour) | - | 7 | No analyses conducted |
| Others (body splash) | - | 1 | No analyses conducted |
The detection limit is 1 mg/kg
The calculated exposure scenarios for benzyl alcohol are worst case scenarios. The daily exposure quantity, Systemic Exposure Dosage, SED, is calculated on basis of the factors below which appear from TGD and SCCNFP 0690 (2003) (Table 7-7).
Table 7-7. Factors in the exposure assessment of benzyl alcohol.
| Product type | Number of applications | Applied quantity per application | Concentration of benzyl alcohol (worst case) |
Absorption through the skin (worst case) |
Retention factor | The child’s weight (worst case) |
| Body shampoo/ Bath gel |
1-2 per day | 5.0 g | 120 mg/kg | 100% | 0.01 | 15 kg |
| Body lotion/cream | 1-2 per day | 7.5 g | 790 mg/kg | 1 | ||
| Shampoo | 2-7 per week | 12.0 g | 8 mg/kg | 0.01 | ||
| Eau de toilette | 1-2 per day1 | 3.0 g² | 5 mg/kg | 1 | ||
| Bath caviar | 1 per week³ | 25.0 g4 | 2 mg/kg | 0.01 |
1 – assumption as no data in TGD or SCCNFP guidelines.
2 – the value for deodorant spray is applied. This value will be significantly higher than for eau de toilette (but there is no available value for eau de toilette in TGD).
3 – assumption as no data in TGD or SCCNFP guidelines.
4 – assumption as no data in TGD or SCCNFP guidelines.
There are no data for eau de toilette in TGD. A density of 1 g/litre is assumed as eau de toilette mainly consists of water and about 8% fragrances according to the analyse of an eau de toilette in this project. It is assumed that eau de toilette is applied once per day. Regarding application quantity, no quantity is stated in TGD for eau de toilette. Instead the value for deodorant spray of 3.0 g per application is used. This value is probably significantly higher than the real value as experience shows that less eau de toilette than deodorant spray is used.
Nor any data are found for bath caviar in TGD. Therefore, an educated guess of an applied frequency of 1 time per week is made and for the applied quantity per application a value of 25 g is used. In the purchased bath caviar products, the content varies between 25 and 100 g. In the smallest packages the content corresponds to a small handful. As worst case it is assumed that the whole package of 25 g is used.
Correspondingly, no standard weight for a child is stated in TGD. As described in the beginning of this chapter, a value of 15 kg for a child’s weight is applied.
The daily exposure (SED) for benzyl alcohol is calculated as described for Kathon and gives for instance the following daily exposure when using body lotion on a three-year-old child:
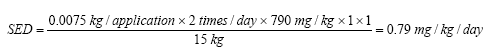
In the table below the daily exposure (SED) for benzyl alcohol is stated for the different product types.
Table 7.8: Daily exposure (SED) for benzyl alcohol for the different product types
| Applied quantity per application (in kg) | Number of daily applications | Concentration (mg/kg) |
Rf | SED (mg/kg/day) | |
| Body shampoo/ Bath gel |
0.005 | 2 | 120 | 0.01 | 0.00080 |
| Body lotion/cream | 0.0075 | 2 | 790 | 1 | 0.79 |
| Shampoo | 0.012 | 1 | 8 | 0.01 | 0.000064 |
| Eau de toilette | 0.003 | 2 | 5 | 1 | 0.0020 |
| Bath caviar | 0.025 | 0.14 | 2 | 0.01 | 0.0000048 |
It is seen that the highest daily exposure occurs when using body lotion.
7.2.1 Risk assessment for benzyl alcohol
The risk assessment for benzyl alcohol is conducted for the critical effect of benzyl alcohol which is lethargy at acute toxicity. It is presumed that 100% of the benzyl alcohol to which the child is exposed will also be absorbed in the body.
Several NOAEL values for benzyl alcohol from different studies are stated in section 6.2.1 in the health assessment of benzyl alcohol. For these worst case calculations, the lowest NOAEL value found in the literature for benzyl alcohol is applied, i.e. the value 188 mg/kg bw/day found in tests with acute toxicity with mice (OECD SIDS, 2001).
When calculating Margin of Safety (MoS) for benzyl alcohol the calculated daily exposure (= SED) is placed in the following formula:
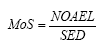
Margin of Safety for benzyl alcohol when using body lotion on a three-year-old child will be:
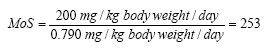
Tabel 7.9: Margin of safety for benzyl alcohol for the different product types
| SED (mg/kg/day) |
MoS | |
| Body shampoo/bath gel | 0.00080 | 235,000 |
| Body lotion/cream | 0.79 | 238 |
| Shampoo | 0.000064 | 2,937,000 |
| Eau de toilette | 0.0020 | 94,000 |
| Bath caviar | 0.0000048 | 39,480,000 |
Margin of Safety ought to be above 100 to take account of a safety factor of 10 for extrapolation of data from animals to humans and a safety factor of 10 to take account of especially sensitive human individuals. The calculation above shows that Margin of Safety is not exceeded in any of the concrete examples. However, for the body lotion product Margin of Safety is relatively close to 100 whereas for the other products there is an even very large margin of safety.
However, it must be emphazied that MoS is not calculated for allergy but for another critical effect, cf. the health assessments in chapter 6.
It must also be emphazied that the mapped body lotion/cream is close to the limit to represent a health risk on basis of the content of benzyl alcohol. Therefore, it cannot be denied that other non-analyzed products may have a higher content of benzyl alcohol and thus form a potential health risk. It may be stay-on products such as body lotion/cream which may represent a potential health risk in the worst-case scenario. However, the calculated Margin of Safety shows that there is no health risk in using the products in relation to the content of benzyl alcohol.
8 Discussion
- 8.1 Risk assessment based on the analyse results
- 8.2 The analyse results for packaging
- 8.3 General health assessment of the cosmetic products for children
- 8.4 Recommendations on purchase and apply of cosmetic products for children
8.1 Risk assessment based on the analyse results
Based on the results of the quantitative analyses of cosmetic products for children, a risk assessment of the analyzed products for their content of Kathon and benzyl alcohol is conducted. The risk assessment is conducted for the acute toxicity of Kathon and benzyl alcohol.
The risk assessment shows that the Margin of Safety (MoS) is far from being exceeded, with one exception where a body lotion/cream has a content of benzyl alcohol resulting in a margin of safety of approx. 240. I.e. there is still no health risk in using the products in relation to the content of benzyl alcohol.
In general, the following applies for the studied products that there is no health risk based on the analyzed content of Kathon and benzyl alcohol in the products. However, it must be emphazised that the risk assessment is solely based on the content of either Kathon or benzyl alcohol in the analyzed products. Furthermore, it must also be emphazised that the assessment is not based on allergy as the critical effect, cf. the health assessments in chapter 6.
8.2 The analyse results for packaging
Selected products were analyzed for their content of heavy metals in the packaging material. All the measured total values of heavy metals in the packaging materials are significantly below the threshold limits indicating how large quantities of metals that are permitted to migrate from the toys when used. Thus the measured total values do not give cause for health concern in relation to the existing threshold limits for migration of substances from toys materials. For the same reason no migration analyses of selected products have been conducted.
Other products were tested for a content of phthalates in the packaging material. The phthalates being identified were primarily DEHP and DINP. The total content of phthalate in the studied packaging materials was as a maximum 31%. All the studied products with a content of phthalates are designed as a cartoon figure, animals or similar of which some are assessed to be comprised by the rules for toys based on the indicative statement of the Danish Safety Technology Authority, while the rest are defined as child care articles. DEHP and DINP were banned from April 2007 in toys for children up to 14 years in a concentration above 0.1% - in the case of DINP only in toys which children may put into the mouth. Cosmetic products which are also comprised by the rules for toys and where the packaging contains phthalates are thus in discrepancy with the new phthalate legislation. All products were legal at the time of purchasing (spring 2006).
8.3 General health assessment of the cosmetic products for children
A majority of the mapped products contains allergenic substances in the form of fragrances, preservatives and/or colouring agents. 74% of all products contain perfume but if the products without the declaration of content and the non-perfumed tooth paste products are kept out of the calculation 86% of the remaining products have a content of perfume. Approximately one third of the products contains one or more of the 26 fragrances mandatory to declare.
More than the half (63%) of the products contains preservatives. Several preservatives are considered to be allergenic just like fragrances. 12 of the applied 23 preservatives are considered to be allergenic or potentially allergenic. Among the six most frequently applied preservatives five parabens are found which in rare instances are known to develop allergy (Denmark’s National Allergy Research Centre, 2006d) but of which some of them are being discussed for endocrine-disrupting effects.
Correspondingly, more than the half (57%) of the products contains colouring agents. Some colouring agents are also considered to be allergenic. A few colouring agents which are considered to be allergenic are found in the cosmetic products for children and these colouring agents are only found in the hair dye products (rinsing colour) – seven products in total. The EU commission bans 22 substances in hair dyes as per 1 December 2006. None of these hair dyes are found in the mapped products for children.
Even if the risk assessment shows that there is no health risk when using the analyzed products with a content of Kathon and benzyl alcohol respectively the risk assessment does not consider the risk of allergy. Both Kathon and benzyl alcohol are allergenic substances and products with these and other allergenic substances such as the other 25 fragrances mandatory to declare, other preservatives (such as 5-bromo-5-nitro-1,3-dioxane) and a few colouring agents represent a risk of sensitization.
Therefore it is relevant to avoid hair dyes and perfumed cosmetic products for children if the risk of sensitization shall be minimized.
8.4 Recommendations on purchase and apply of cosmetic products for children
Summarizing, the results of this project can result in a number of recommendations to consumers about cosmetic products for children. It is difficult to present useful recommendations to the ordinary consumer who not necessarily can understand nor remember long chemical names which should be avoided. However, to some extent it can be necessary as it can be difficult or nearly impossible to find products without perfume in some product groups.
As an example all the identified soaps for children contain perfume and a general recommendation on avoiding perfume is therefore problematic.
Recommendations to consumers
- If possible, buy cosmetic products without perfume or as a minimum avoid products without a content of the 26 allergenic fragrances.
- If you buy cosmetic products designed as for instance cartoon figures, be aware that they are CE labelled which means that they comply with the health requirements and safety requirements on toys.
9 References
Alexander BR, 2002. ”An assessment of the comparative sensitization potential of some common isothiazolinones”. B. R. Alexander, Thor Group Management Limited, Margate, Kent, United Kingdom. Contact Dermatitis 2002, p. 191-196.
Ben-Dyke et al, 1977. ”Phenoxyetol: Toxicity in oral administration to rats for thirteen weeks”. Ben-Dyke R, Ashby R, Bhatt A, Newman AJ. Life Science Research Report No. 77/NLL5/375 to Nipa Laboratories, 21 November 1977. (Not published). As described in (OECD SIDS, 2004).
BioSite, 2006. BioSite om benzoesyre. Found on http://www.biosite.dk/leksikon/benzosy.htm. Latest update 4.6.2006.
Breslin et al, 1991. ”Hemolytic activity of ethylene glycol phenyl ether (EGPE) in rabbits”. Breslin WJ, Phillips JE, Lomax LG et al. Fund. Appl. Toxicol. 17: p.466-481, 1991. As described in (OECD SIDS, 2004).
Chemfinder. Chemfinder.com. Found on: http://chemfinder.cambridgesoft.com/.
ChemicalLand21.com, 2006. ”5-bromo-5-nitro-1,3-dioxane”, ChemicalLand21.com. http://www.chemicalland21.com/lifescience/phar/5-BROMO-5-NITRO-1,3-DIOXANE.htm
CIRP, 1992. ”Final report on the safety assessment of methylisothiazolinone and methylchloroisothiazolinone”. Cosmetic Ingredient Review Program, Vol. 11, No. 1, 1992. As described in (Madsen T et al, 2001).
CIR Review, 1990a. ”Final report on the safety of phenoxyethanol”. J. Am. Coll. Toxicol. 9(2): p.259-277. As described in (OECD SIDS, 2004).
CIR Review, 1990b. Final report on the safety assessment of 5-bromo-5-nitro-1,3-dioxane. Cosmetic Ingredient Review, Vol. 9, No. 2. As described in (Madsen T et al, 2001).
Conner et al, 1996. “Mutagenicity of cosmetic products containing Kathon”. Connor, T.H., P.G. Tee, M. Afshar, K.M. Connor. Environmental and Molecular Mutagenesis, 28, 127-132, 1996. As described in (Madsen T et al, 2001).
The Danish Knowledge Centre for Allergy, 2006a. ”Om allergi. Kathon/isothiazolinone”. Beskrivelse fundet i september 2006 på hjemmeside for Videncenter for Allergi. http://www.videncenterforallergi.dk/1_om_allergi/
Konserveringsmiddel_Al/Kathon/Kathon.html
The Danish Knowledge Centre for Allergy, 2006b. ”Nationale allergidata. Aktuelle allergidata” Description found in October 2006 on the homepage of the Danish Knowledge Centre for Allergy. http://www.videncenterforallergi.dk/4_
nationale_data/aktuelledata/aktueldata.html
The Danish Knowledge Centre for Allergy, 2006c. “Formaldehyd og formaldehydfrigørere”. Desription found in November 2006 on the homepage of the Danish Knowledge Centre for Allergy. http://www.videncenterforallergi.dk/Default.aspx?ID=106
The Danish Knowledge Centre for Allergy, 2006d. “Parabener”. Description found in November 2006 on the homepage of the Danish Knowledge Centre for Allergy. http://www.videncenterforallergi.dk/Default.aspx?ID=111
The Danish Veterinary and Food Administration, 2004. “Overfølsomhed og tilsætningsstoffer i mad”. Ministeriet for familie- og forbrugeranliggender. Fødevarestyrelsen, December 2004, 3. oplag. http://www.astma-allergi.dk/media/filebank/org/tilsaet.pdf
Davies, 1970. ”Acute percutaneous toxicity of phenoxetol to rats”. Davies RE, 1970. Fra COLIPA, 1980, “Summaries of submissions I and II on phenoxyehtanol”. COLIPA report No. 5/70/D57. As described in (OECD SIDS, 2004).
Debethizy et al, 1986. “Absorption and disposition of 14C-labelled Kathon biocide, a mixture of 5-chloro-2-methyl-4-isothiazolin-3-one and 2-methyl-4-isothiazolin-3-one, following intravenous or dermal administration to male sprague-Dawley rats”. Debethizy, J.D., S.L. Longacre, R.B. Steigerwalt, F.W. Deckert, J.N. Moss, A.W. Hayes, J.M. Smith, H.E. Scribner. Fd. Chem. Toxicol., 24 ( 1), 43-49, 1986. As described in (Madsen T et al, 2001).
De Groot et al, 1986. De Groot AC, Bos JD, Jagtman BA, Bruynzeel DP, van Joost T, Weyland JW. Contact Dermatitis 15: p.218-222, 1986. As described in (IUCLID, 2000d).
Deuel HJ et al, 1954. ”Sorbic acid as a Fungistatic Agent for Foods. I. Harmlessness of Sorbic Acid as a Dietary Component”. Food Res. 19: p. 1-12. 1954. As described in (SCCNFP 0532, 2002) and in (OECD SIDS, 2001).
DFG, 1993. ”Toxicants. Critical data evaluation for MAK values and classification of carcinogens”. Deutsche Forschungsgemeinschaft (DFG). Occupational Vol 5, VCH Verlagsgesellschaft mbH, Weinheim, Germany, 1993. As described in (Madsen T et al, 2001).
DS/EN 71-3. Toys. Safety requirements. Part 3: Migration of special substances. Dansk Standard, 2nd edition, approved 1995-08-09.
ECB, 1999. ”Classification of chemical products containing methylchloroisothiazolinone/methylisothiazolinone (MCI/MI)”. ECBI/46/95 – Add.70. 21 January 1999, Birgitaa Gruvberger PhD, Department of Occupational and Environmental Dermatology, University Hosptial, Malmö, Sweden. http://ecb.jrc.it/classlab/4695a70-I.doc
ECB, 2002a. “Draft Report from the Working Group on Sensitisation”, Ispra, 18-19 April 2002. ECBI/13/02 Add.1. European Commission, DG JRC, Joint Research Centre, Insitute for Health and Consumer Protection, Unit: Toxicology and Chemical Substances, European Chemicals Bureau.
http://ecb.jrc.it/classlab/1302a1_Report.doc
ECB, 2002b. “Meeting of the Sensitisation Expert Group, Ispra 4-6 November 2002”. ECBI/81/02, 16 December 2002. European Commission, DG JRC, Joint Research Centre, Insitute for Health and Consumer Protection, Unit: Toxicology and Chemical Substances, European Chemicals Bureau. http://ecb.jrc.it/classlab/8102_Sensitisation_1102_report.doc
The E number guide, 2004. Description of the different E numbers on the homepage www.e-nummer.dk. Published with support from Danish Food and Allied Workers' Union , the Food and Nutrition Union, and the Restaurant Business Union.
Environmental Project No. 615, 2001. Environmental and Health Assessment of Substances in Hoapplyhold Detergents and Cosmetic Detergent Products, Environmental Project No. 615, the Danish Environmental Protection Agency 2001. http://www.mst.dk/udgiv/Publications/2001/
87-7944-596-9/html/default_eng.htm
Environmental project no. 635, 2001. ”Rapport om vejledende liste til selvklassificering af farlige stoffer”. Environmental project no. 635, 2001, the Danish Environmental Protection Agency.
Ettorre A. et al, 2003. Involvement of Oxidative Stress in Apoptosis Induced by a Mixture of Isothiazolinones in Normal Human Keratinocytes. Anna Ettorre et al, Department of Molecular Biology, University of Siena, Siena, Italy. The Journal of Investigateve Dermatology p. 328-336, 2003, The Society for Invenstigative Dermatology, Inc.
European Commission, 2003. “Technical Guidance Document on Risk Assessment in support of Commission Directive 93/67/EEC on Risk Assessment for new notified substances. Commission Regulation (EC) No 1488/94 on Risk Assessment for existing substances. Directive 98/8/EC of the European Parliament and of the Council concerning the placing of biocidal products on the market”. Part I. European Commission, Joint Research Centre, Institute for Health and Consumer Protection, European Chemicals Bureau, 2003.
Fette, Seifen, Anstrichmittel, 1976. Fette, Seifen, Anstrichmittel. Vol. 78, p. 269, 1976. As described in TOXNET’s ChemIDplus database.
Gershanik et al, 1982. ” The gasping syndrome and benzyl alcohol poisoning”. Gershanik J., Boecler, B., Ensley, H., McCloskey, S. & George, W. (1982). New Engl. J. Med., 307, 1384-1388. As described in (WHO, 1996)
Gollapudi et al, 1988. ”Evaluation of 2-phenoxyethanol in the rat bone marrow chromosomal aberration assay”. Gollapudi BB, Linscombe VA, Bruce RJ. Dow Chemical Company Report TXT:K-000111-020, February 4, 1980.
Graham & Kuizenga, 1945. Graham, B.E. & Kuizenga, M.H. “Toxcity studies on benzyl benzoate and related benzyl compounds”. J. Pharmacol. Exp. Ther., 84, 358–362, 1945. As described in (WHO, 2002).
Hanser Publishers, 2001. Plastics Additive Handbook. 5th Edition. Hanser Publishers, Munic. Hanser Gardner Publications, Inc., Cincinatti, 2001.
Heindel et al, 1990. “Assessment of ethylene glycol monobutyl and monophenyl ether reproductive toxicity using a continuous breeding protocol in Swiss CD-1 mice”. Heindel JJ, Gulati DK, Russell VS, Reel JR, Lawton AD, Lamb JC. Fun. Appl. Toxicol. 15: p.683-696, 1990. As described in (OECD SIDS, 2004).
Hill Top Research, 1980. “Acute oral and acute dermal toxicity, and acute eye irritation potential of samle 2219-93” (cosmetic grade phenoxyethanol). Report No. 80-479-21 to CFA, June 18, 1980. (Not published). As described in (OECD SIDS, 2004).
HSDB. Hazardous Substance Data Bank. TOXNET. Extraction from HSDB of the different substances on CAS no. http://toxnet.nlm.nih.gov/cgi-bin/sis/htmlgen?HSDB
Howes, 1988. Cosmetic Science 88, 15th IFSCC Int. Congress. Howes D., 1988. As described in (OECD SIDS, 2004)
IFRA. Internatiol Fragrance Association. Codes and Standards http://www.ifraorg.org/GuideLines.asp
ILO, 1973. Internation Labour Organisation, C138 Minimum Age Convention, 1973. http://www.ilo.org/ilolex/cgi-lex/convde.pl?C138
INCI, 2006. List of cosmetic ingredients ordered by INCI name, European Commission – Enterprise and Industry Directorate General – Consumer Goods – Cosmetics. Last updated 24.2.2006. Found on http://ec.europa.eu/enterprise/cosmetics/html/cosm_inci_index.htm.
The Information Centre for Environment & Health, 2004. Data on 5-bromo-5-nitro-1,3-dioxane in the Chemical database from the Information Centre for Environment & Health. Data latest updated 11.3.2004. http://192.38.244.140/kd_vis_stof.asp?stof_id=153.
IUCLID, 2000a. “IUCLID Datasheet. Substance ID: 26172-55-4, 5-chloro-2-methyl-2H-isothiazol-3-one”. IUCLID (International Uniform Chemical Information Database). European Commission, Joint Research Centre, Institute for Health and Consumer Protection, European Chemicals Bureau, 2000. Found on http://ecb.jrc.it/esis.
IUCLID, 2000b. “IUCLID Datasheet. Substance ID: 100-51-6, benzyl alcohol“. IUCLID (International Uniform Chemical Information Database). European Commission, Joint Research Centre, Institute for Health and Consumer Protection, European Chemicals Bureau, 2000. Found on http://ecb.jrc.it/esis.
IUCLID, 2000c. “IUCLID Datasheet. Substance ID: 532-32-1, Sodium benzoate “. IUCLID (International Uniform Chemical Information Database). European Commission, Joint Research Centre, Institute for Health and Consumer Protection, European Chemicals Bureau, 2000. Found on http://ecb.jrc.it/esis.
IUCLID, 2000d. “IUCLID Datasheet. Substance ID: 122-99-6, 2-phenoxyethanol “. IUCLID (International Uniform Chemical Information Database). European Commission, Joint Research Centre, Institute for Health and Consumer Protection, European Chemicals Bureau, 2000. Found on http://ecb.jrc.it/esis.
Jenner et al, 1964. Fd. Cosmet. Toxicol. 2: p.327-343. Jenner PM et al, 1964. As described in (OECD SIDS, 2001).
Jensen AA, 1999. ”Fokus på farlige stoffer nr. 77. Benzylalkohol og dens estere”. Working Environment 1999; 15(11): 49-50.
Larsen JR et al, 2006. ”Survey and health and environmental assessment of hand soap”, Jette Rud Larsen – Dansk Toksikologi Center, Trine Thorup Andersen og Dorte Rasmussen – DHI – Institut for Vand og Miljø. Mapping of chemical substances in consumer products, no. 69 2006. Danish Ministry of the Environment, the Danish Environmental Protection Agency.
Loeser, 1978. Bayer AG data, short report, 3.11.1978. As described in (OECD SIDS, 2001).
Lovell et al, 1984. Contact Dermatitis 11: p.187. Lovell CR et al, 1984. As described in (OECD SIDS, 2004).
Madsen T et al, 2001. ”Environmental and Health Assessment of Substances in Hoapplyhold Detergents and Cosmetic Detergent Products”, Environmental project no. 615, 2001. CETOX, Madsen T et al. The Danish Environmental Protection Agency.
Malinckrodt Chemicals, 2004. Materials Safety Data Sheet, Sodium Benzoate. Malinckrodt Baker Inc., 222 Red School Lane, Phillipsburg, NJ 08865. 02.19.04. Found on http://www.jtbaker.com/msds/englishhtml/S2930.htm.
Matissek R, 1985. “Zur Analytik mikrobiocider Isothiazolone”, Matissek, R; Fresenius Z Anal. Chem. (1985) 322: 465-469.
Mærkningsguiden, 1997. ”Mærkningsguiden”, Pjece nr. 4, december 1997, Statens Husholdningsråd, Forbrugerstyrelsen (The National Consumer Agency).
Nair B, 2001. ”Final Report on Safety Assessment of Benzyl Alcohol, Benzoic Acid, and Sodium Benzoate”, Nair B. International Joundal of Toxicology, Vol. 20, No. 4, Supplement 3, January 03, 2001, p. 23-50. Taylor & Friends.
Netdoktor, 2006. ”Drenges højde og vægt i forhold til alder” og ”Pigers højde og vægt i forhold til alder”. Found on Netdoktor’s homepage in October, 2006. http://www.netdoktor.dk/boern/fakta/drengevaeksttabel.htm and http://www.netdoktor.dk/Sunderaad/fakta/pigevaeksttabel.htm.
NPIRI, 1974. ”Raw Materials Data Handbook”. Vol I: 6. 1974. As described in (OECD SIDS, 2001).
NTP, 1984. “Reproduction and Fertility Assessment of Ethylene Glycol Monophenyl Ether (CAS #122-99-6) in CD-1 Mice When Administered in Feed”. Department of Health & Human Services/National Institute of Environmental Health Sciences, National Toxicology Program. NTP Study No. RACB83101, November 1984. http://ntp.niehs.nih.gov/index.cfm?
objectid=071C4D74-FB0B-FF73-89C8AEEC022285F3.
NTP, 1989. ”Toxicology and carcinogenesis studies of benzyl alcohol in F344/N rats and B6C3F1 mice (gavage studies)”. NTP-TR-343, NIH Publication No. 89-2599. US Department of Health and Human Services, Public Health Service, National Institute of Health, June 1989. http://ntp-server.niehs.nih.gov/ntp/htdocs/LT_rpts/tr395.pdf
OECD SIDS, 2001. ”SIDS Initial Assessment Report for 13th SIAM (Bern, 7th - 9th November 2001) – Benzoates”. UNEP Publications.
OECD SIDS, 2004. “SIDS Initial Assessment Report for SIAM 18 (Paris, France, 20-23 April 2004) – Ethylene Glycol Phenyl Ether”. UNEP Publications.
Onodera et al, 1978. “Studies on effects of sodium benzoate on fetapplys and offspring of Wistar rats”. Onodera et al, Eisei Shikensho Hokoku, 96: p.47-55 (på Japansk). As described in (WHO, 2000).
Opdyke, 1973. “Monograph on Fragrance Raw Materials”. Fd. Cosmet. Toxicol. 11: p. 1011-1013. Opdyke, DLJ. 1973. As described in (OECD SIDS, 2001).
Orientation from the Danish Environmental Protection Agency no. 8, 2004. ”Listen over uønskede stoffer 2004”. the Danish Environmental Protection Agency 2004.
The Positive list), 2005. ”Positivlisten. Fortegnelse over tilsætningsstoffer til fødevarer”, The Danish Veterinary and Food Administration, 2005. http://www.foedevarestyrelsen.dk/NR
/exeres/66BFDC97-3D7C-46CD-A90B-3E430628410D.htm?
Procter & Gamble, 1992. “Acute oral toxicity study (LD50) in the rat. Benzyl alcohol”. Unpublished report No. 2131-110/233 from Hazleton, Harrogate, England. Submitted to WHO by Flavor and Extract Manufacturers Associations, USA, 1992. As descriped in (WHO, 2002).
Richold et al, 1982. “Micronucleus test on phenoxyethanol”. Huntingdon Research Center report NPA 19/82966 for NIPA Laboratories Ltd., 19 November 1982. (Ikke publiceret). As described in (OECD SIDS, 2004).
Roche Applied Science, 2005. “5-bromo-5-nitro-1,3-dioxane (BND)”, Roche Applied Science 2005/2006, edited September 2005. http://www.roche-applied-science.com/indbio_neu/
industry/clinical/pdf/biocides_buffers_detergent/136-137.pdf
Rohm and Haas. “RH-573T – Skin Irritation Study in Rabits”. Report No. 96R-123.
Rohm and Haas, 1999. “RH-573 Technical: Acute oral toxicity study in male and female rats”, Rohm and Haas Report No. 98R-212, 1999.
Rohm and Haas, 2000a. “Neolone™ 950 Preservative Acute Oral Toxicity study in Male and Female rats”. Report No. OOR-004, 2000.
Rohm and Haas, 2000b. “Neolone™ 950 Preservative Acute Dermal Toxicity Study in Male and Female rats”. Report No. OOR-005, 2000.
Rohm and Haas, 2000c. “RH-573 Technical: Three-Month Drinking Water Toxicity Study in Rats”. Report No. 99R-135, 2000.
Rohm and Haas, 2000d. “Neolone™ 950 Preservative (100 ppm aqueous solution) Skin Irritation Study in Rabbits”. Report No. OOR-006.
Rohm and Haas, 2000e. “Neolone™ 950 Preservative (100 ppm aqueous solution) Eye Irritation Study in Rabbits”. Report No. OOR-007.
Rohm and Haas, 2004a. “Kathon™ CG. A safe, effective, globally approved preservative for rinse-off products”. Rohm and Haas Personal Care, February 2004.
Roper et al, 1997. “Percutaneous penetration of 2-phenoxyethanol via rat and human skin”. Roper CS, Howes D, Blain PG, Williams FM. Food Chem. Toxicol. 35: p.1009-1016, 1997. (As descriped in OECD SIDS, 2004).
RTECS. Registry of Toxic Effects of Chemical Substances. Canadian Centre for Occupational Health and Safety. http://ccinfoweb.ccohs.ca/rtecs/search.html
Safety Emporium, 2004. Sample MSDS for Benzoic Acid. Safety Emporium, Laboratory and Safety Supplies. 25.5.2004. Found on http://www.ilpi.com/msds/benzoic.html.
SCCNFP 0017, 1999. The Scientific Committee on Cosmetic Products and Non-Food Products intended for Consumers. “Opinion concerning Fragrance allergy in consumers. A review of the problem. Analysis of the need for appropriate consumer information and identification of consumer allergens”. Adopted by the SCCNFP during the plenary session of 8 December 1999. SCCNFP/0017/98, final.
SCCNFP 0532, 2001. The Scientific Committee on Cosmetic Products and Non-Food Products intended for Consumers. “Concerning Benzoic Acid and Sodium Benzoate”. Adopted by the SCCNFP during the 20th plenary meeting of 4 June 2002. SCCNFP/0532/01, final.
SCCNFP 0625, 2003. The Scientific Committee on Cosmetic Products and Non-Food Products intended for Consumers. “Opinion concerning methylisothiazolinone”. COLIPA No. P94. Adopted by the SCCNFP during the 23rd plenary meeting of 18 March 2003. SCCNFP/0625/02, final.
SCCNFP 0670, 2003. The Scientific Committee on Cosmetic Products and Non-Food Products intended for Consumers. “Opinion concerning Update of entry No. 39 of Annex VI to Directive 76/768/EEC on Cosmetic Products: Mixture of 5-chloro-2-methyl-isothiazolin-3(2H)-one and 2-methylisothiazolin-3(2H)-one”. COLIPA No. P56. Adopted by the SCCNFP during the 23rd plenary meeting of 24-25 June 2003. SCCNFP/0670/03, final.
SCCNFP 0690, 2003. The Scientific Committee on Cosmetic Products and Non-Food Products intended for Consumers. “The SCCNFP’s Notes of Guidance for the Testing of Cosmetic Ingredients and Their Saftey Evaluation, 5th Revision”. Adopted by the SCCNFP during the 25th pleanary meeting of 20 October 2003. SCCNFP/0690/03 Final.
SCCNFP 0805, 2004. The Scientific Committee on Cosmetic Products and Non-Food Products intended for Consumers. “Opinion concerning methylisothiazolinone”. COLIPA No. P94. Adopted by the SCCNFP on 23 April 2004 by means of written procedure.
SCCP 0849, 2004. Scientific Committee on Consumer Products SCCP. “Clarification of the SCCNFP Opion on the Update of entry No. 39 of Annex VI to Directive 76/768/EEC on Cosmetic Products: Mixture of 5-chloro-2-methyl-isothiazolin-3(2H)-one and 2-methylisothiazolin-3(2H)-one”. COLIPA No. P56. Adopted by the SCCP during the 2nd plenary meeting of 7 December 2004. SCCP/0849/04.
SCF 78, 2002. “Opinion of the Scientific Committee on Food on Benzyl Alcohol (expressed on 24 September 2002)”. European Commission, Health & Consumer Protection Directorate-General, Directorate C – Scientific Opinions, C2 – Management of scientific committees II; scientific co-operation and networks. Scientific Committee on Food. SCF/CS/ADD/FLAV/78 Final, 17. Sept. 2002.
ScienceLab, 2005. “Material Safety Data Sheet 5-bromo-5-nitro, 1,3-dioxane MSDS”. ScienceLab.com Chemicals & Laboratory Equipment. Latest update 10.10.2005. http://www.sciencelab.com/xMSDS-5_
Bromo_5_nitro_1_3_dioxane-9927112
Smyth et al, 1941. Smyth HF et al. J. Ind. Hyg. Toxicol. 23: p. 259-263, 1941. As described in (IUCLID, 2000d).
Smyth & Carpenter, 1948. Smyth HF & Carpenter CP. J. Ind. Hyg. Toxicol. 30: p. 63-68, 1948. As described in (OECD SIDS, 2001).
Smyth et al., 1951. Smyth, H.F., Carpenter, C.P. & Weil, C.S. “Range finding toxicity data: List IV”. Arch. Ind. Hyg. Occup. Med., 4,119–122, 1951. As described in (WHO, 2002).
Sodemoto & Enomoto, 1980. ”Report of carcinogenesis bioassay of Sodium benzoate in rats: Absence of carcinogenicity of sodium benzoate in rats”. Sodemoto Y & Enomoto M. J. Environ. Pahtol. Toxicol. 4: p.87-95, 1980. As described in (SCCNPF 0532, 2002)
SPIN2000. “SPIN Substances in Preparations in Nordic countries”. www.spin2000.net.
Stat. Ord. 151, 1999. Bekendtgørelse om forbud mod phthalater i legetøj til børn i alderen 0-3 år samt i visse småbørnsartikler m.v. BEK nr. 151 af 15.3.1999. The Danish Ministry of the Environment and Energy.
Stat. Ord. 975, 2002. Bekendtgørelse om ændring af bekendtgørelse om forbud mod phthalater i legetøj til børn i alderen 0-3 år samt i visse småbørnsartikler m.v. BEK nr. 975 af 27.11.2002. The Danish Ministry of the Environment.
Stat. Ord. 1116, 2003. Bekendtgørelse om sikkerhedskrav til legetøj og produkter, som på grund af deres ydre fremtræden kan forveksles med levnedsmidler. BEK nr. 1116 af 12.12.2003. The Danish Safety Technology Authority.
Stat. Ord. 923, 2005. Bekendtgørelse om listen over farlige stoffer. BEK nr. 923 af 28.09.2005. The Danish Ministry of the Environment.
Stat. Ord. 422, 2006. Bekendtgørelse om kosmetiske produkter. BEK nr. 422 af 4.5.2006. The Danish Ministry of the Environment.
Stat. Ord. 786, 2006. Bekendtgørelse om forbud mod ftalater i legetøj og småbørnsartikler. BEK nr. 786 af 11.7.2006. The Danish Ministry of the Environment.
Stat. Ord. 877, 2006. Bekendtgørelse om ændring af bekendtgørelse om kosmetiske produkter. BEK nr. 877 af 17.8.2006. The Danish Ministry of the Environment.
Stat. Ord. 1074, 2006. Bekendtgørelse om ændring af bekendtgørelse om forbud mod ftalater i legetøj og småbørnsartikler. BEK nr. 1074 af 3.11.2006. The Danish Ministry of the Environment.
TOXNET. TOXNET Toxicology Data Network. United States National Library of Medicine. Found on http://toxnet.nlm.nih.gov/.
US FDA. ”Guide to inspections of cosmetic product manufactures”. US Food and Drug Administration. Office of Regulatory Affairs. http://www.fda.gov/ora/Inspect_ref/igs/cosmet.html
The weekly journal of the Danish Medical Association, 2002. “Luftbåren kahonkontaktallergi udløst af vandbaseret plasticmaling”. The weekly journal of the Danish Medical Association 2002, 164 (18): 2411. Diana Kristensen, Hase Ole Hein & Kaare Weismann.
WHO, 1996. ”Safety Evaluation of Certain Food Additives and Contaminants. Benzyl acetate, benzyl alcohol, benzaldehyde, and benzoic acid and its salts”. WHO Food Additivies Series: 37. First draft prepared by Vavasour E, Chemical Health and Hazard Assessment Division, Bureau of Chemical Safety, Food Directorate, Health Protection Branch, Health Canada, Ottawa, Ontario, Canada. 1996. Found on IPCS INCHEM homepage. http://www.inchem.org/documents/jecfa/jecmono/v37je05.htm.
WHO, 2000. “Benzoic Acid and Sodium Benzoate”. Concise International Chemical Assessment Document 26. Corrigenda published by 12 April 2005 have been incorporated in this file. First draft prepared by Dr. A. Wibbertmann et al, Fraunhofer Institute for Toxicology and Aerosol Research, Hanover, Germany. World Health Organisation, Geneva, 2000.
WHO, 2002. ”Safety Evaluation of Certain Food Additives and Contaminants. Benzyl derivatives”. WHO Food Additivies Series: 48. First draft prepared by Dr. J. Gry et al. Institute of Food Safety and Nutrition, Danish Veterinary and Food Administration, Ministry of Food, Agriculture and Fisheries. 2002. Found on IPCS INCHEM homepage. http://www.inchem.org/documents/jecfa/jecmono/v48je01.htm.
Wikipedia, 2006. ”Bronidox”. Description of Bronidox (5-bromo-5-nitro-1,3-dioxane). Wikipeida, the free encyclopedia. Found on http://en.wikipedia.org/wiki/bronidox. Latest update 5 August 2006.
Footnotes
[1] Kathon is a mixture of the two substances methylchlorisothiazolinone and methylisothiazolinone in the ratio 3:1
[2] Massage oils for children are studied in an earlier survey project. Therefore they are only included if they are not studied in the project in question.
[3] Press release “Commission bans 22 hair dye substances to increase consumer safety”, 20.07.2006. IP/06/1047. Found on http://europa.eu.int. http://europa.eu.int/rapid/pressReleasesAction.
do?reference=IP/06/1047&format=HTML&aged=0&
language=EN&guiLanguage=en
[4] According to Statutory Order no. 877 of 17.8.2006 (Stat. Ord. 877, 2006) the date of the temporary permission of the substance is changed from 31.12.2006 to 31.12.2007.
[5] According to Statutory Order no. 877 of 17.8.2006 (Stat. Ord. 877, 2006) the date of the temporary permission of the substance is changed from 31.12.2006 to 31.12.2007.
[6] According to Statutory Order no. 877 of 17.8.2006 (Stat. Ord. 877, 2006) the date of the temporary permission of the substance is changed from 31.12.2006 to 31.12.2007.
[7] According to Statutory Order no. 877 of 17.8.2006 (Stat. Ord. 877, 2006) the date of the temporary permission of the substance is changed from 31.12.2006 to 31.12.2007.
[8] According to Statutory Order no. 877 of 17.8.2006 (Stat. Ord. 877, 2006) the date of the temporary permission of the substance is changed from 31.12.2006 to 31.12.2007.
[9] Please note that this product is a stay-on product; that means the limit for separate declaration of the 26 fragrances is now 10 mg/kg.
[10] Please note that this product is a stay-on product; that means the limit for separate declaration of the 26 fragrances is now 10 mg/kg.
[11] Please note that the maximum measured concentration is only based on a few analysis results of products where the individual fragrance occurs. For the majority of the products it is unknown in which concentration the fragrances occur (only that it is above 100 mg/kg or 0.001%).
[12] Please note that DEHIP (Bis(2-ethylhexyl)isophthalate) is not an ortho-phthalate but a meta-phthalate.
[13] The different sources disagree a little on how large quantities of sodium benzoate that are applied for the different purposes but preservative for food products and beverages seems to be more than 50% of the total produced quantity.
[14] EC3 value is the estimated concentration which is needed to provoke a three times increase in the activity of lymph gland cell.
Appendix A: Extractions from the database of cosmetics products for children
- 1 Overview of the content of the database
- 2 All chemical substances found in the mapped products
- 3 Chemical substances in the mapped products distributed on the type of products
- 4 Constituents with restrictions
1 Overview of the content of the database
This appendix contains a description of the content of the created database of cosmetics products for children.
1.1 The database
A database in Microsoft Access 2000 has been created for the Danish Environmental Protection Agency covering the in total 208 mapped cosmetics products for children. The database is shortly presented when it is opened (see figure 1).
Figure 1: Front page of the database
The database is based on a product overview where the following is recorded for each product:
- Content of chemical substances
- Various information about the products (type, place of purchase etc.)
1.2 Product overview
For each product information has been keyed into the database about which constituents are forming a part of the products but also in which ranking the constituents are listed on the product. Thus, the ranking is an indication of the relative concentration of the constituents in the products. A low number (high ranking) expresses that the substance enters as a main constituent in the product whereas a high number (low ranking) indicates that the substance enters as an additive, for instance preservatives.
Furthermore, the database contains a wide range of other information about the products.
Figure 2: Product information
The following information is recorded in the database for all the purchased products (see figure 2):
| Type information | Remarks |
| Product name | |
| Product type | The database operates with the following product types: Shampoo, balsam, body shampoo/bath gel, bobble bath, soap solid or liquid, bath confetti/bath caviar/fizzle salt, tooth paste, eau de toilette/perfume, deodorant, body lotion/cream, hair dyes (rinsing colours), hair styling products, massage oil, bath oil or other things |
| The chemical substances contained in the products including ranking order | The ranking order states the order of the substances in the list of constituents |
| Number of chemical substances (constituents) contained in the product in total | |
| Specification of how the product is procured | That means whether the product is bought or the declaration of contents is sent (or downloaded via the Internet) |
| Specification of where the product is bought or procured | The name of the convenience store chain and in which town |
| Specification whether the product is bought in a shop, at a hair dresser or via the Internet | |
| The name of the producer or importer | |
| When the product is bought or received | Stated by date. For products bought on the Internet the recorded date is the date when the products are ordered. Regarding declarations of contents the recorded date is the date where the declarations either are received or printed from the Internet |
| The bar code on the product | If available |
| Batch number | If available |
| Specifications of packaging material | That means a description of the type of packaging in question – for instance whether it is neutral or designed as a special figure |
| Description of the packaging material | For instance, whether the bottle is designed as Winnie the Pooh sitting in an umbrella. This description makes it much more easier to identify the products |
| Description of plastic type of the packaging via specification of plastic number if such a number is stated on the packaging | If the packaging is not made of plastic this field has no relevance |
| Whether the product is CE marked | |
| Description of the scent of the product, i.e. whether it contains perfume. | If perfume is stated in the declaration of contents it is only recorded as ”Perfume” in the database |
| Description whether the products are marketed directly to children | |
| Possible comments | For instance, whether products are sold in twin pack or notes about clarification of INCI names |
2 All chemical substances found in the mapped products
This chapter is an overview of all chemical substances found in the mapped cosmetics products for children. In total 461 different substances are found in the 208 mapped products.
Furtheremore, the chapter shows how often the different chemical substances are found and with which average ranking they are found (that means in which ranking the substances are listed on the products). The ranking is an indication of the relative concentration of the constituents in the products. A low number (high ranking) indicates that the substance is a main constituent in the product whereas a high number (low ranking) indicates that the substance is an additive, for instance a preservative.
At the same time the table shows how many of the 208 products of which the substances form a part as well as which average ranking they have.
2.1 Stated after falling frequency
2.2 Alphabetical overview
3 Chemical substances in the mapped products distributed on the type of products
This chapter shows the chemical substances in the cosmetics products for children distributed on the type of product. That means the substances which are found in bath confetti, balsom, body lotion/cream, body shampoo/bath gel, shampoo, bobble bath and tooth paste. The constituents of the different types of products are only listed for the seven types of products, mentioned above. For the other product types, the database must be used. At the same time the chapter shows with which average ranking the substances form a part of the mentioned product type – illustrating the average relative concentration of the substances in the product.
3.1 Bath confetti/caviar/fizzle salt
3.2 Balsam
3.3 Body lotion/cream
3.4 Body shampoo/bath gel
3.5 Shampoo
3.6 Bobble bath
3.7 Tooth paste
4 Constituents with restrictions
This chapter is an overview of the 66 constituents, which have restrictions in relation to the Cosmetics Statutory Order, out of the in total 461constituents found in the 208 mapped products. Please note that the latest version of the INCI list from the beginning of the project (from 24 February 2006) is used. This means that the latest changes with colouring agens and partly also the 26 fragrances are not included in the INCI list in the database.
Table 4.1: List of constituents in the cosmetics products for children with restrictions in relation to the Cosmetics Statutory Order.
| INCI Name | CAS N. | Function | Is in number of products | Average ranking | Restriction |
| METHYLPARABEN | 99-76-3 | preservative | 79 | 11.2 | VI/1,12 |
| PROPYLPARABEN | 94-13-3 | preservative | 70 | 13.2 | VI/1,12 |
| CI 42090 | 3844-45-9 | cosmetic colorant | 62 | 15.6 | IV/1 |
| CI 17200 | 3567-66-6 | cosmetic colorant | 50 | 13.7 | IV/1 |
| PHENOXYETHANOL | 122-99-6 | preservative | 50 | 9.2 | VI/1,29 |
| BUTYLPARABEN | 94-26-8 | preservative | 48 | 14.1 | VI/1,12 |
| ETHYLPARABEN | 120-47-8 | preservative | 46 | 11.6 | VI/1,12 |
| CI 19140 | 1934-21-0 | cosmetic colorant | 40 | 16.3 | IV/1 |
| ISOBUTYLPARABEN | 857-25-9 | preservative | 39 | 15.0 | VI/1,12 |
| BENZYL ALCOHOL | 100-51-6 | preservative / solvent | 20 | 16.3 | III/1,45 - VI/1,34 |
| CI 16255 | 2611-82-7 | cosmetic colorant | 16 | 19.8 | IV/1 |
| 5-BROMO-5-NITRO-1,3-DIOXANE | 30007-47-7 | preservative | 15 | 6.0 | VI/1,20 |
| METHYLISOTHIAZOLINONE | 2682-20-4 | preservative | 15 | 15.6 | VI/1,39 |
| METHYLCHLOROISOTHIAZOLINONE | 26172-55-4 | preservative | 15 | 14.7 | VI/1,39 |
| CI 16035 | 25956-17-6 | cosmetic colorant | 14 | 19.6 | IV/1 |
| SODIUM BENZOATE | 532-32-1 | preservative | 13 | 11.7 | VI/1,1 |
| CI 14700 | 4548-53-2 | cosmetic colorant | 12 | 18.9 | IV/1 |
| DMDM HYDANTOIN | 6440-58-0 | preservative | 11 | 9.2 | VI/1,33 |
| SODIUM FLUORIDE | 7681-49-4 | oral care / antiplaque | 11 | 10.1 | III/1,31 |
| CI 77891 | 13463-67-7 | cosmetic colorant | 10 | 12.9 | IV/1 |
| CETRIMONIUM CHLORIDE | 112-02-7 | preservative | 9 | 21.3 | VI/1,44 |
| TRIETHANOLAMINE | 102-71-6 | buffering | 8 | 9.0 | III/1,62 |
| TITANIUM DIOXIDE | 13463-67-7 | opacifying / uv absorber | 8 | 8.4 | IV/1 |
| SODIUM HYDROXIDE | 1310-73-2 | buffering / denaturant | 8 | 13.0 | III/1,15a |
| BEHENTRIMONIUM CHLORIDE | 17301-53-0 | preservative / hair conditioning | 7 | 3.0 | VI/1,44 |
| IMIDAZOLIDINYL UREA | 39236-46-9 | preservative | 7 | 21.1 | VI/1,27 |
| CI 73360 | 2379-74-0 | cosmetic colorant | 7 | 14.0 | IV/1 |
| TETRASODIUM ETIDRONATE | 3794-83-0 | chelating / emulsion stabilising / viscosity controlling | 6 | 9.0 | III/1,53 |
| CI 47005 | 8004-92-0 | cosmetic colorant | 6 | 17.3 | IV/1 |
| 2-BROMO-2-NITROPROPANE-1,3-DIOL | 52-51-7 | preservative | 6 | 12.7 | VI/1,21 |
| CI 77007 | 1302-83-6 | cosmetic colorant | 6 | 13.5 | IV/1 |
| CI 59040 | 6358-69-6 | cosmetic colorant | 5 | 12.6 | IV/1 |
| SODIUM BORATE | 1330-43-4 | buffering | 5 | 4.6 | III/1,1a / III/1,1b |
| CI 16185 | 915-67-3 | cosmetic colorant | 5 | 14.4 | IV/1 |
| CI 75810 | 11006-34-1 | cosmetic colorant | 5 | 20.2 | IV/1 |
| CI 12085 | 2814-77-9 | cosmetic colorant | 5 | 8.8 | IV/1 |
| CI 61570 | 4403-90-1 | cosmetic colorant | 5 | 21.0 | IV/1 |
| CI 15985 | 2783-94-0 | cosmetic colorant | 5 | 12.0 | IV/1 |
| TALC | 14807-96-6 | absorbent / bulking | 4 | 5.3 | III/1,59 |
| DIAZOLIDINYL UREA | 78491-02-8 | preservative | 4 | 5.3 | VI/1,46 |
| CI 45380 | 17372-87-1 | cosmetic colorant | 4 | 13.0 | IV/1 |
| CI 45100 | 3520-42-1 | cosmetic colorant | 4 | 17.8 | IV/1 |
| BENZOPHENONE-4 | 4065-45-6 | uv filter / uv absorber | 4 | 11.0 | VII/1,22 |
| ISOPROPYLPARABEN | 4191-73-5 | preservative | 4 | 15.0 | VI/1,12 |
| CI 45430 | 16423-68-0 | cosmetic colorant | 4 | 13.5 | IV/1 |
| CI 77499 | 12227-89-3 | cosmetic colorant | 3 | 12.3 | IV/1 |
| CI 77491 | 1309-37-1 | cosmetic colorant | 3 | 13.7 | IV/1 |
| CI 15510 | 633-96-5 | cosmetic colorant | 2 | 9.5 | IV/1 |
| BETA VULGARIS EXTRACT | 89957-89-1 | cosmetic colorant | 2 | 10.0 | IV/1 |
| CI 77289 | 12001-99-9 | cosmetic colorant | 2 | 12.0 | IV/1 |
| SODIUM MONOFLUOROPHOSPHATE | 10163-15-2 | oral care / antiplaque | 2 | 9.5 | III/1,27 |
| QUATERNIUM-15 | 4080-31-3 | preservative | 2 | 15.0 | VI/1,31 |
| LAURTRIMONIUM CHLORIDE | 112-00-5 | preservative / antistatic / emulsifying | 2 | 10.5 | VI/1,44 |
| CI 77492 | 51274-00-1 | cosmetic colorant | 2 | 14.5 | IV/1 |
| CI 75120 | 1393-63-1 | cosmetic colorant | 2 | 18.5 | IV/1 |
| CI 74160 | 147-14-8 | cosmetic colorant | 2 | 15.5 | IV/1 |
| CI 45410 | 18472-87-2 | cosmetic colorant | 2 | 15.0 | IV/1 |
| CI 74260 | 1328-53-6 | cosmetic colorant | 1 | 14.0 | IV/1 |
| CI 42051 | 3536-49-0 | cosmetic colorant | 1 | 14.0 | IV/1 |
| CI 75470 | 1328-60-5 | cosmetic colorant | 1 | 20.0 | IV/1 |
| IODOPROPYNYL BUTYLCARBAMATE | 55406-53-6 | preservative | 1 | 21.0 | VI/1,56 |
| POTASSIUM SORBATE | 24634-61-5 | preservative | 1 | 14.0 | VI/1,4 |
| CI 45350 | 518-47-8 | cosmetic colorant | 1 | 8.0 | IV/1 |
| SODIUM METHYLPARABEN | 5026-62-0 | preservative | 1 | 10.0 | VI/1,12 |
| CI 42053 | 2353-45-9 | cosmetic colorant | 1 | 18.0 | IV/1 |
| ZINC OXIDE | 1314-13-2 | bulking / uv absorber / skin protecting | 1 | 7.0 | IV/1 |
Appendix B: The 26 fragrances
The table below is an overview of the 26 fragrances which according to ”The statutory order on cosmetics products” – Stat. Ord. no. 422 of 4.5.2006 (Stat. Ord. 422, 2006) must be declared separately in the declaration of content when the concentration is higher than 0.001% in products which are not to be cleansed after use and 0.01% in products which are to be cleansed after use.
| The 26 fragrances (stated with their INCI name) |
CAS numbers |
| Anise alcohol | 105-13-5 |
| Amyl cinnamal | 122-40-7 |
| Amylcinnamyl alcohol | 101-85-9 |
| Benzyl alcohol | 100-51-6 |
| Benzyl benzoate | 120-51-4 |
| Benzyl cinnamate | 103-41-3 |
| Benzyl salicylate | 118-58-1 |
| Cinnamyl alcohol | 104-54-1 |
| Cinnamal | 104-55-2 |
| Citral | 5392-40-5 |
| Citronellol | 106-22-9 |
| Coumarin | 91-64-5 |
| Eugenol | 97-53-0 |
| Farnesol | 4602-84-0 |
| Geraniol | 106-24-1 |
| Hexyl cinnamal | 101-86-0 |
| Hydroxycitronellal | 107-75-5 |
| a-Isomethylionone | 127-51-5 |
| Butylphenyl methylpropional (Lillial) | 80-54-6 |
| Limonene | 5989-27-5 |
| Linalool | 78-70-6 |
| Hydroxyisohexyl 3-cyclohexen carboxaldehyde (Lyral) | 31906-04-4 |
| Isoeugenol | 97-54-1 |
| Methyl 2-Octynoate | 111-12-6 |
| Evernia Prunastri (Oakmoss) Extract | 90028-68-5 |
| Evernia Furfuracea (Treemoss) Extract | 90028-67-4 |
Version 1.0 October 2007, © Danish Environmental Protection Agency