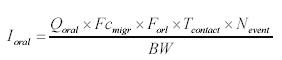|
Survey of Chemical Substances in Consumer Products, No. 60, 2005 Migration and health assessment of chemical substances in surface treated wooden toysContents
PrefaceThis report on migration and health assessment of chemical substances in surface treated toys made of wood is a project under the programme "Survey of chemical substances in consumer products" performed by the Danish Environmental Protection Agency. The purpose of the project was the continuation of a previous survey project on natural toys ("Report of natural toys made of plant fibres, woollen fibres and solid wood. Ferdinand et al. 2003) which included the consumption of among other wooden toys. The study also showed that wooden toys might be surface treated with health problematic substances. Therefore the purpose of this project specifically is to survey which chemical substances that migrates from the surface and to which the consumer might be exposed. The project is performed by the Danish Technological Institute, Chemistry and Water technology. Performing persons are:
The project was supervised by a steering committee including: Lise Emmy Møller, Danish Environmental Protection Agency Eva Pedersen, Danish Technological Institute Ole Christian Hansen, Danish Technological Institute Sammenfatning og konklusionerSom et led i Miljøstyrelsens kortlægning af kemiske stoffer i en række forbrugerprodukter ønskes viden om, hvilke stoffer der afgives og en sundhedsmæssig vurdering af kemiske stoffer i overfladebehandlet trælegetøj. Projektet "Afgivelse og sundhedsmæssig vurdering af kemiske stoffer i overfladebehandlet trælegetøj" er en fortsættelse af et tidligere projekt, hvor forbruget af bl.a. trælegetøj blev kortlagt (Kortlægningsrapport nr. 33). Konklusionen fra den tidligere kortlægningsrapport var, at forbruget af trælegetøj var ca. 50 mio. kroner eller 2,3% af omsætningen af legetøj i Danmark. En opdatering baseret på samme kilde antyder, at forbruget er øget fra ca. 370 tons i 2001 til 420 tons trælegetøj i 2003. Ifølge Legetøjsbekendtgørelsen har producenten eller importøren pligt til at vurdere, om legetøjet er farligt. Herudover er der fastsat yderligere krav i henhold til gældende standarder. Standarden fastsætter grænseværdier for afgivelse af otte tungmetaller fra alle typer af legetøj nemlig antimon, arsen, barium, bly, cadmium, chrom, kviksølv og selen. Disse metaller anses for at være dem, der har væsentligst sundhedsmæssig betydning. I Legetøjsbekendtgørelsen er anført, at legetøj til børn ikke må indeholde stoffer eller præparater som defineret i Direktiv 67/548/EØF og 88/379/EØF kan skade sundheden for de børn, der bruger det. Er disse stoffer til stede, skal der udføres en vurdering af, om de optræder i mængder, der kan være til skade for barnets helbred Denne undersøgelse omfatter derfor en migrationsanalyse af udvalgte stykker overfladebehandlet trælegetøj samt en sundhedsmæssig vurdering af de fundne identificerede stoffer. Der er fokuseret på de metaller, der er omtalt i Legetøjsbekendtgørelsen samt organiske stoffer, der er klassificeret i Listen over farlige stoffer. Af de fundne stoffer blev der, baseret på klassificering og forekomst, udvalgt et antal stoffer til nærmere vurdering af en eventuel sundhedsrisiko for forbrugerne. Forbrugerne er her defineret som børn i alderen 0-3 år. Af de 125 identificerede stoffer var 43 klassificeret i Listen over farlige stoffer, og yderligere 16 kunne selvklassificeres efter Miljøstyrelsens Vejledende liste til selvklassificering. Af de klassificerede stoffer var 2 klassificeret at kunne være kræftfremkaldende (carcinogene, Carc. cat. 3), 1 stof at kunne være mutagent (Mut. cat. 3), og 9 stoffer reproduktionstoksiske (Repr. cat. 2-3). Desuden var 5 stoffer klassificeret sensibiliserende. På trods af, at klassificeringen af enkelte af de identificerede stoffer var af en alvorlig karakter, kunne ingen af de fundne stoffer vurderes at udgøre en umiddelbar sundhedsmæssig risiko for forbrugeren (i dette tilfælde små børn der putter legetøjet i munden). Vurderingen er baseret på, at barnet putter legetøjet i munden i maksimalt 3 timer per dag. Vurderingerne er i de fleste tilfælde vurderet ved sammenligninger med data fra langtidsforsøg eller ligefrem kroniske data. Da lysten til at putte legetøj i munden må antages at være overstået efter nogle få år, skulle konklusionerne derfor være acceptable. Det konkluderes derfor, at ingen af de undersøgte stykker legetøj udgør en sundhedsmæssig risiko for børn, der putter dem i munden. Det bemærkes dog, at vurderingen ikke tager højde for at eksponeringen for enkeltstoffer øges ved samtidig leg med flere stykker trælegetøj. Desuden kan der være andre kilder til forekomsten af samme stof. Summary and conclusionsAs a part of the Danish Environmental Protection Agency's programme on survey of chemical substances in consumer products a project was initiated on a survey on which chemical substances were released (migrated) from surface treated (coated) wooden toys and an evaluation of the health risk. The project "Migration and health assessment of chemical substances in surface treated wooden toys" is a continuation of a previous project where the consumption of natural toys including wooden toys was surveyed (Survey report no. 33). The conclusion from the previous report was that the consumption of wooden toys was approx. 50 million DKK or 2.3% of the turnover of toys in Denmark. An update based on the same source indicates that the consumption has increased from approx. 370 tonnes in 2001 to approx. 420 tonnes of wooden toys in 2003. According to the Directive on safety of toys, the manufacturer or the importer has the obligation to evaluate whether the toy is hazardous to children. It furthermore includes requirements in accordance to standards in force. The standard sets the threshold limit values for the migration from all types of toys of eight heavy metals: antimony, arsenic, barium, cadmium, chromium, lead, mercury and selenium. In the Toy Directive it is further noted that toys must not contain dangerous substances or preparations within the meaning of Directive 67/548/EEC and 88/379/EEC in amounts which may harm the health of children using them. Are such substances present, an evaluation should be performed to establish whether the amount migrating from the toys might cause harm to the health of the child. This study therefore includes a migration analysis of selected surface coated wooden toys and a health evaluation of the identified substances. A focus has been on the metals mentioned in the Directive on toy safety and organic chemical substances classified in the Annex I to Directive 67/548/EEC (Dangerous substances). Of the identified substances and based on classification and numbers a number of substances were selected for further evaluation of potential health risk to the consumer. In this case, consumers are defined as children aged 0 to 3 years. Of the 125 identified substances 43 were classified in Annex I to Directive 67/548/EEC and further 16 could be self classified according the Advisory list for self classification of dangerous substances from the Danish Environmental Protection Agency. Of the Annex I classified substances 2 were classified carcinogenic (Carc. cat. 3), 1 substance mutagenic (Mut. cat. 3), and 9 substances reproduction toxic (Repr. cat. 2-3). Besides, 5 substances were classified sensitising. Despite the severe classification of a number of the identified substances, none of the substances was evaluated to present any immediate risk to the consumer (in this situation children mouthing the toy). The assessment is based on a child mouthing the toy for a maximum of 3 hours per day. The assessments are mostly evaluated by comparison to data from subchronic or even chronic effect data. As the urge to put toys in the mouth is assumed to decline after a few years, the conclusions should be acceptable. The conclusion from this study is, therefore, that none of the examined pieces of wooden toys are considered to be a health risk to children mouthing the toys. It is noted that the evaluation does not consider that the exposure to indicidual substances is increased when several toys are played with at the same time. Besides other sources to the same substance may exist. 1 Introduction1.1 BackgroundThe purpose of the study is an analysis of the migration of heavy metals and organic chemical substances from surface treated wooden toys and a health assessment of selected chemical substances migrating from surface treated wooden toys. The project specifically targets wooden toys aimed at children less than 3 years of age and that is surface coated with paint, wood stain, lacquers or similar. Particularly for young children/toddlers a risk of exposure exists when the children places the toy in the mouth. The study is a follow-up of a previous project by the Danish Environmental Protection Agency (Survey no. 33, 2003). The conclusion of the survey was that the release of health hazardous substances from the surface treated wooden toys was difficult to evaluate since the migration was not examined in that project. The manufacturer or the importer has an obligation to evaluate whether the toy is hazardous. Furthermore, additional requirements in relation to current standards are set. The current regulation sets threshold limit values for the migration of 8 metals from all types of toys: The metals are antimony, arsenic, barium, cadmium, chromium, lead, mercury and selenium. These metals are considered to be of most essential health concern/relevance. In the Statutory Order on toys (Bkg. 1116, 2003) is further noted that toys to children must not contain substances or preparations as defined in Directive 67/548/EEC and 88/378/EEC may be hazardous to the health of children using it. Therefore, some of the detected substances that might pose a health problem are evaluated. 1.2 PurposeThe study aims to show whether a migration of health hazardous substances takes place from surface treated toys intended for children below 3 years of age and whether this may cause a potential health problem. 1.3 Product descriptionThe products included in the study are primarily wooden toys labelled suitable to children 0 to 3 years of age. Wooden toys surface treated with paint, colorants and lacquers are intentionally selected. A further description of the selected toys is presented in section 2.6. 1.4 Target groupConsumers of surface treated toy products are usually young children / toddlers. Because the group of children aged between 0 and 3 years may be especially sensitive to chemical influences toys for this particular group are the ones focused on in selection and analyses. 1.5 Project developmentThe project is divided into 2 phases: Phase 1 Based on the survey in the survey report no. 33 the apparently most problematic toy categories from the information on the preparations used for surface treatment/coating are selected. A selection of 15 toys for children less than 3 years of age is sampled. The toy is selected among toys, which based on appearance, size or function might be expected to be put into the mouth, e.g. rattles, wooden blocks, pieces of puzzles. Toys stained, oiled, lacquered or painted were focused on. The migration of heavy metals was determined according to the EN71-3 standard where the simulant is 0.07 M HCl (simulating artificial saliva). The migration of organic chemical substances from the toys was determined qualitatively by a screening analysis in relation to the preliminary standards on toys (EN 71-9, -10 and –11, cf ref.list). In these standards the migration takes place in the simulant at 37°C for 120 minutes. Pretreatment of the test samples was performed according to standard prEN 71-10. In an EU-report (CEN/TC 52, 2003) prepared by the working group on safety of toys, the migration from a plastic film was measured using several extraction media (artificial saliva, hydrochloric acid (0.07 M HCl), distilled water at pH 7, distilled water containing 10% ethanol, and albumin protein in a sodium chloride solution. The conclusion was that in most instances water was better or as good as any other of the saliva, perspiration or gastric acid simulants for the extraction of organic substances from the plastic matrice. Based on this report the migration examinations could be performed using deionised water as requested in prEN 71-10. However, according to agreement with the Danish Environmental protection Agency and following a severe criticism of the CEN report from the Scientific Committee (SCTEE 2003) the migration examinations were performed using artificial saliva prepared according to Amtliche Sammllung von Untersuchungsverfahren nach § 35 LMBG nr. 82.10 1, copied from DIN no. 53 160. The identified substances were compared to the regulation in the Statutory Order on toys (Bkg. 1116, 2003) concerning regulated substances. Phase 2 The second phase concerns the screening of potential health hazardous effects from substances released (migrated) from the surface treated wooden toys. A literature screening on the identified substances in the qualitative migration analyses is performed. The screening/literature search is based on literature information and has the purpose to ensure that the substances focused on in the quantitative analyses are the most relevant. Some substances are evaluated in the CEN/TG 52 (2003) report and these informations are included in the final assessments. Following the qualitative analyses the appeared results are evaluated. Data on the individual substances such as NOAEL, LOAEL or other relevant data are used when available. Alternatively QSAR is used for substances where no data were found. A comparison is performed to the EU classification criteria. 2 Survey
2.1 IntroductionThe survey of wooden toys is performed in a previous survey report (Survey report no. 33, Ferdinand et al 2003). The most essential information from the report relevant to this project is cited below. The survey has been supplemented with information from Statistics Denmark covering import and export for year 2003. 2.2 SurveyThe Danish market for toys made of solid wood has been estimated to 50 million DKK (approx. 6.7 million Euro) or 2.3% of the total Danish trade turnover of the market of toys. 2.3 ProductionIn Denmark a minor production of toys and especially playing tools of wood takes place. As presented by the National Agency for Enterprise and Housing (Erhvervsfremmestyrelsen 2000), a number of manufacturers of "playing tools" exists. All of these manufacturers have been approached. The majority of what is being manufactured is plastic toys and plastic and wooden tools. The playing tools are parts for playgrounds, as for instance play houses, climbing frames and swings. Only two companies manufacture actual wooden toys for children below the age of 36 months. Economic information about the production of wooden toys and information that there are only a few employees (Erhvervsfremmestyrelsen 2000) indicate that the share of Danish production to the Danish market of wooden natural toys is very small. 2.3.1 Turnover of natural toys of woodAll approached importers except 1 have confirmed that they import toys of wood. The estimation of the turnover of wooden toys is based on economic information from Statistics Denmark, importers and manufacturers and interviews with individual persons. When comparing these information it is estimated that the turnover of natural toys of wood amounts to 50 million DKK in consumer prices. Thus, the wooden toys are estimated to constitute 2.3% or 1/44 of the turnover of toys in Denmark. According to Statistics Denmark, the total turnover of wooden toys (2001) amounts to a minimum of 39.5 million DKK including a profit factor of 2.5 that was confirmed by importers and used for toys. The 39.5 million DKK is estimated as Total turnover = Production + Import – Export, Commodity numbers: CN-9503.30.10, 9503.49.10 and 9503.60.10. Further a contribution comes from the commodity number CN-9503.90.99: "Toys, except when made of plastic, rubber, textile and metal, not mentioned elsewhere". This group alone covers 119.5 million DKK in consumer prices. This means that according to Statistics Denmark the turnover of wooden toys is between 39.5 and 159 million DKK. Table 1 Import and export of wooden toys according to Statistics Denmark 2001 and 2003
CN: Commodity numbers used by Statistics Denmark Import –export in 2001 was in Ferdinand et al. (2003) stated to 39.5 million DKK. The value is observed to be a little adjusted in the table above (table 1). A change in the statistics of the calculation system for external trade has lead to an upgrading of the import and export values for the years 2000, 2001 and 2002 according to News from Statistics Denmark no. 72, 2003. Most importers of wooden toys have passed on information from manufacturers and it is estimated that the major part of natural toys made of wood in Denmark comes from these manufacturers. Information on the individual manufacturers is found in Ferdinand et al. (2003). The wooden toys are primarily being manufactured in Thailand and in Germany. The Thai manufacturers all use softwood or rubber wood from 30 to 40 year old discarded rubber plantations. The German manufacturers all use wood from Central Europe and of Central European wood species, often beech and to a limited degree maple, ash, birch and pine. 2.4 Surface treatment of wooden toysWooden toys can be surface treated with stains, glazes, paint and /or lacquers which may release health hazardous substances. About surface treatment preparations for natural toys made of wood it was found that a large part of surface treated wooden toys was painted, stained or glazed with additional treatment with a lacquer or an oil. For lacquering primarily nitrocellulose lacquers are used. The manufacturers all recommend that stained or glazed products should be additionally treated with a surface treatment that closes the surface, such as a lacquer or a drying oil. None of the pigments in the mentioned products are classified. Toys are regulated by the Statutory Order on toys (Bkg. nr. 1116, 2003). The person who places the toy on the market has the responsibility that it is safe and as such does not harm the health in relation to the use. Products placed on the market as toys must be CE marked. The Statutory Order on toys further mentions that the person who places a toy product on the market has the responsibility that it must not contain dangerous substances or preparations that may harm the health of children using the toys. In most instances the manufacturers have no other information on the surface treatment of the wooden toys than that they are CE authorised/marked. The manufacturers usually forward a copy of a test report on compliance to EN70-3 and refer to the manufacturer of the surface treatment product (paint, lacquer, glaze or stain) (Ferdinand et al. 2003). EN (European Norm) no. 71 is the European standard for testing of toys in compliance with the Statutory Order of Toys. The standard contains currently 7 parts of which part 3: "Migration of certain elements " is especially relevant to this study. In the standard the following requirements (limit values) for the content of 8 elements in mg/kg are stated: Antimony <60, arsenic <25, barium <1000, cadmium <75, chromium <60, lead <90, mercury <60 and selenium <500 mg/kg. According to the Statutory Order on toys the bioavailability of these elements resulting from the use of toys must not, as an objective, exceed the following levels per day: 0.2 μg for antimony, 0.1 μg for arsenic, 25.0 μg for barium, 0.6 μg for cadmium, 0.3 μg for chromium, 0.7 μg for lead, 0.5 μg for mercury, and 5.0 μg for selenium. 2.5 Types of surface treatmentMost wooden toys have been surface treated, which gives the wood an attractive look and makes it more resistant against outer influences. The colouring can either be done by covering the wood with a paint or by adding a stain or glaze, which penetrates the wood and sets off/maintains the structure of the wood. In most cases the toys have had additional treatment with a lacquer or oil. Lacquer and oil add to making the product more glossy and resistant against outer influences such as UV light, bumps, sweat and saliva (Ferdinand et al. 2003). 2.5.1 Information on surface treatmentThe manufacturers of wooden toys have been asked to inform type and chemical composition of the surface treatment, which their toys have had. Most manufacturers have replied only that their toys comply with the requirements in EN71-3, most surface treatments of wooden toys are made with stains or paints with a final lacquering, and as lacquering is mostly used nitrocellulose lacquer (Ferdinand et al. 2003). 2.5.2 Stains and glazesTwo types of surface treatments exist that colour the wood and at the same time maintain the structure of the veins in the wood visible, namely stain and glaze. The stain pigment penetrates the wood and forms a chemical reaction with the lignin of the wood. According to one of the manufacturers, these reactive colouring agents are classified as toxic and very toxic. After the colouring, a risk of release of free reactive colouring agent remains. Glaze colouring agents are, however, often agents that have been approved for food. Instead of reacting directly with the lignin, they are glued on. The agents are not very reactive. Both stain and glaze manufacturers recommend that wooden toys, which have been treated with their products, are additionally treated with a product which closes the surface, i.e. a lacquer, wax or a drying oil, in order to be resistant against sweat and saliva (Ferdinand et al. 2003). 2.6 Sampled toy typesMost sampled toy types are labelled for the use by children 0 to 3 years of age. However two products for a little older children have been included as younger siblings may get hold op them and put them into the mouth. Finally a birthday decoration was included as it was considered tempting to play with for small children. The sampled and analysed products are described below: Table 2. Description of sampled and analysed wooden toys
3 Analysis results
3.1 Analysis methodsA number of 15 products were selected for quantitative analysis of migration of heavy metals and organic chemical substances. A representative area of the product was sampled for the analysis according to the standard prEN 71-10 (CEN standard EN 71-10: Safety of toys - Sample preparation and extraction). The sample was milled off the wooden toys surface to a depth of 3 mm and then further demolished/milled to uniformity. The migration of heavy metals, inorganic and organic substances was measured according to the EN 71-3 standard where the simulant 0.07 M hydrochlorous acid (HCl) simulates artificial saliva or gastric acid. The extraction was performed according to the standard EN 71-3, which prescribes 1 hour during agitation at 37±2°C and an additional 1 hour at standing at 37±2°C (CEN standard EN 71-3: Safety of toys – Migration of certain elements. EN71-3, 2003). 3.1.1 Inorganic substances3.1.1.1 Test preparation0.5 g sample – accurately weighed – in a polyethylene container was added 25 g 0.07 M hydrochloric acid and left under steady shaking at 37 ± 2 °C for 1 hour. Then the extraction container was left standing at 37 ± 2 °C for a further 1 hour after which the extract was filtered. Blind samples were prepared correspondingly. Double preparations were prepared. 3.1.1.2 Quantitative analysisThe extract was analysed for its content of antimony, arsenic, barium, cadmium, chromium, lead, mercury and selenium by inductively coupled plasma atomic emission spectrometry (ICP-AES) with quantitation against external standards in0. 07 M HCl. 3.1.2 Organic substances3.1.2.1 Sample preparationApproximately 2 g sample – accurately weighed – in a 100 ml glass beak was added 20 ml artificial saliva (saliva solution) and extracted at 37 ± 2 °C for 2 hours. The extract was filtered and then extracted using dichloromethane added deuterium labelled internal standards. Blind samples were prepared correspondingly. Double preparations were prepared. 3.1.2.2 AnalysisThe dichloromethane extract was analysed for its content of organic components by capillary gaschromatography with mass spectrometric detection. The quantitation against external standards of selected analytes and the internal standards. 3.2 Migration of inorganic substancesThe migration of inorganic substances (heavy metals) are performed as described in the section on methods (cf. above). 3.2.1 Analysis resultsThe analysis results from the quantitative determinations for inorganic elements in the selected wooden toy products are presented in the table below. Table 3 Measured migrated amount of inorganic substances in saliva extracts
-: below the detection level (DL) Table 3 continued
-: below the detection level (DL) Table 3 continued
-: below the detection level (DL) Barium was the only metal detected in the analyses of the extracts. Barium was detected in 5 out of 15 analysed pieces of toy. The measured concentrations varied from 29 to 90 mg/kg sample of the products' surfaces. A health evaluation of barium is performed in section 4. 3.3 Migration of organic substances3.3.1 Analysis resultsThe analysis results from the quantitative determinations of identified organic substances extracted by saliva from the surface of the selected toy products are presented in the table below. Table 4 Identified organic substances migrated from the surface of 15 toy products (μg/g) 3.4 Selection of chemical substances for evaluationFrom the detected substances and in co-operation with the Danish Environmental Protection Agency, were selected based on classification and detection a number of substances for further evaluation of a potential health risk to the consumers. The consumers are defined in this context as children 0 to 3 years of age. Of the 125 identified substances are 43 classified in the List of dangerous substances (Miljøministeriet 2002), further 16 substances could be self-classified according to the Advisory list for self-classification from the Danish EPA (Miljøstyrelsen 2001). Of the classified substances 2 were classified to be possible carcinogenic (Carc.cat.3), 1 substance to be possible mutagenic (Mut.cat.3), and 9 substances toxic to reproduction (Repr.cat.2-3). Below is mentioned the substances that had the most rigorous classification and selected substances with a less rigorous classification. A possible reason for the selection of the substance for an actual health evaluation is presented. Benzyl alcohol, CAS no. 100-51-6, is classified Xn;R20/22 i.e. Harmful by inhalation and if swallowed. The substance is detected in the extract from 3 products. Based on a low concentration and the classification the substance is not included. Benzyl butylphthalate (BBP), CAS no. 85-68-7, is classified Repr.Cat.2;R61 Repr.Cat.3;R62, i.e. the substance is reproduction toxic (May cause harm to the unborn child, and Possible risk of impaired fertility). However, the substance is detected in the extract from 1 product only and at trace level. The substance is therefore not included for a further evaluation. 1-Butanol, CAS no. 71-36-3, is classified R10 Xn;R 22 Xi;R37/38-41 R67 (i.e. Flammable. Harmful if swallowed. Irritating to respiratory system and skin. Risk of serious damage to eyes. Vapours may cause drowsiness and dizziness). The substance was detected in extracts from 5 products and even if the substance is harmful and an irritant it is evaluated to be at such low concentrations that it is not selected for further evaluation. 2-Butoxyethanol, CAS no. 111-76-2, is classified Xn;R20/21/22 Xi;R36/38 (Harmful by inhalation, in contact with skin and if swallowed. Irritating to eyes and skin). The substance is harmful and an irritant. The substance was detected in extracts from 12 out of 15 tested products and in a few at very high concentrations. The substance is therefore selected for further evaluation. 2-(2-Butoxyethoxy)-ethanol (= butyldiglycol), CAS no. 112-34-5, is classified Xi; R36, i.e. Irritant (Irritating to eyes). The substance was detected in extracts from 5 out of 15 products and even if the concentrations were moderate a further evaluation is decided. 2-Butoxyethylacetate, CAS no. 112-07-2, is classified Xn;R20/21, i.e. Harmful (Harmful by inhalation and in contact with skin). The substance was detected in extracts from 3 products at low concentrations. 1-Butoxy-2-propanol, CAS no. 5131-66-8, is classified Xi;R36/38, i.e. Irritant (Irritating to eyes and skin). The substance was detected in extracts from 2 products. Butylacetate, CAS no. 123-86-4, is classified R10 R66 R67, i.e. Flammable. Repeated exposure may cause skin dryness or cracking. Vapours may cause drowsiness and dizziness. The substance was detected in extracts from 6 products. The classification refers to effects from the pure substance and is therefore considered less relevant in this context. Cyclohexanol, CAS no. 108-93-0, is classified Xn;R20/22 Xi;R37/38, i.e. both harmful and irritating (Harmful by inhalation and if swallowed. Irritating to respiratory system and skin). The substance was detected in the extract from 1 product at trace level. Cyclohexanone, CAS no. 108-94-1, is classified R10 Xn;R20, i.e. Flammable and Harmful by inhalation. The substance was detected in extracts from 2 products at moderate levels. To evaluate the level the substance has been included for a further evaluation. Dibutyl phthalate (DBP), CAS no. 84-74-2, is classified Repr.Cat.2;R61 Repr.Cat.3;R62 N;R50, i.e. May cause harm to the unborn child, Possible risk of impaired fertility and Very toxic to aquatic organisms. The substance is thus classified seriously for a substance that can be extracted from a toy. The substance was detected in extracts from 4 products but at very low concentrations. Diethylhexylphthalate (DEHP), CAS no. 117-81-7, is classified Repr.Cat.2;R60-61, i.e. May impair fertility and May cause harm to the unborn child. The substance was detected in extracts from 1 product at trace level. Diisobutyl phthalate, CAS no. 84-69-5, is not classified in the List of dangerous substances but is found in the Advisory list for self-classification with the classification N;R50/53, i.e. Very toxic to aquatic organisms, may cause long-term adverse effects in the aquatic environment. The substance was detected in the extracts from 8 out of 15 products. All the measured concentrationswere low and the substance therefore not considered a candidate for further evaluation. 2,6-Dimethoxybenzoquinone, CAS no. 530-55-2, is not classified in the List of dangerous substances but is found in the Advisory list for self-classification with the classification Xn;R22 R43, i.e. Harmful if swallowed and May cause sensitization by skin contact. The substance was detected in extracts from 7 out of 15 products. Even if the concentrations are low the substance is selected for further evaluation. 3,5-Dimethoxy-4-hydroxycinnamaldehyde, CAS no. 87345-53-7, is not classified in the List of Dangerous substances but the substance was detected in 11 out of 15 products. The substance was detected at low concentrations. Because the substance probably has its origin from the wood used to make the toy it is not selected for a further evaluation. 3,6-Dimethyl-1,4-dioxan-2,5-dion, CAS no. 4511-42-6, is not classified in the List of dangerous substances but is found in the advisory list for self-classification with the classification Xn;R22, i.e. Harmful if swallowed. The molecular structure of the substance is identical to the following substance: 3,6-Dimethyl-2,5-dioxo-1,4-dioxane (EU: dilactide), CAS no. 95-96-5, which is also self-classified Xn;R22, Harmful if swallowed. The two substances appear to alternate in all products and a further evaluation is therefore performed. N,N-Dimethylformamide, CAS no. 68-12-2, is classified Repr.Cat.2;R61 Xn;R20/21 Xi;R36, i.e. May cause harm to the unborn child. Harmful by inhalation and in contact with skin and Irritating to eyes. A serious classification but the substance was detected only in 1 product and at trace level. 2-Ethoxyethanol, CAS no. 110-80-5, is classified R10 Repr.Cat.2;R60-61 Xn;R20/21/22, i.e. Flammable, May impair fertility, May cause harm to the unborn child, Harmful by inhalation, in contact with skin and if swallowed. The substance was detected in extracts from 2 products but due to the classification it was selected for further evaluation. 2-(2-Ethoxyethoxy)-ethanol (= diethylene glycol ether), CAS no. 111-90-0, is not classified in the List of dangerous substances. The substance was detected in the extracts from 4 products at moderate to high concentrations. The substance is therefore selected for further evaluation. 2-Ethylhexanoic acid, CAS no. 149-57-5, is classified Repr.Cat.3;R63, i.e. Possible risk of harm to the unborn child. The substance was detected in extracts from 1 product at a very low concentration. Formamide, CAS no. 75-12-7, is classified Repr.Cat.2;R61, i.e. May cause harm to the unborn child. The classification is serious and since the substance is detected in the extracts from 2 products at moderate concentrations it is selected for further evaluation. Furfural, CAS no. 98-01-1, is classified Carc.Cat.3;R40 T;R23/25 Xn;R21 Xi;R36/37, i.e. Limited evidence of carcinogenic effects, Toxic by inhalation and if swallowed, Harmful in contact with skin, Irritating to eyes and respiratory system. The substance was detected in extracts from 4 products. Although the concentrations were low the substance was selected for further evaluation due to its classification. Hexanoic acid, CAS no. 142-62-1, is not adopted on the List of dangerous substances. However it is noted that the substance was detected in 13 out of 15 products at concentrations from trace levels to high concentrations. Because the substance not immediate is considered to pose a health concern at the determined concentrations (1.8-120 μg/g) the substance is not selected for further evaluation. 4-Hydroxy-3,5-dimethoxy-benzaldehyde, CAS no. 134-96-3, is not classified in the List of dangerous substances but is found in the Advisory list for self-classification with the classification R43, i.e. May cause sensitization by skin contact. The substance was detected in extracts from 12 out of 15 products at low to moderate concentrations. Therefore, the substance is selected for further evaluation. 2-Methoxyethanol, CAS no. 109-86-4, is classified R10 Repr.Cat.2;R60-61 Xn;R20/21/22, i.e. Flammable, May impair fertility, May cause harm to the unborn child, Harmful by inhalation, in contact with skin and if swallowed. The substance was detected in extracts from 1 product. 1-Methyl-2-pyrrolidone, CAS no. 872-50-4, is classified Xi; R36/38, i.e. Irritant (Irritating to eyes and skin). The substance was detected in extracts from 5 products at moderate to high concentrations. Therefore, the substance is selected for further evaluation. Phenol, CAS no. 108-95-2, is classified Muta.Cat.3;R68 T;R23/24/25 Xn;R48/20/21/22 C;R34, i.e. Possible risk of irreversible effects, Toxic by inhalation, in contact with skin and if swallowed, Harmful: danger of serious damage to health by prolonged exposure through inhalation, in contact with skin and if swallowed and Causes burns. The classification is very serious but the substance was only detected in extracts from 1 product and at trace level. Therefore the substance is not selected. Phthalic acid anhydride, CAS no. 85-44-9, is classified Xn;R22 Xi;R37/38-41 R42/43, i.e. Harmful if swallowed, Irritating to respiratory system and skin, Risk of serious damage to eyes, May cause sensitization by inhalation and skin contact. The substance was detected in extracts from 4 products: at trace level in 3 and at low concentration in 1 product. Toluene, CAS no. 108-88-3, is classified (29th ATP, Directive 2004/73/EC, EC 2004) F;R11 Repr.Cat.3;R63 Xn;R48/20-65 Xi;R38 R67, i.e. Highly flammable, Possible risk of harm to the unborn child, Harmful: danger of serious damage to health by prolonged exposure through inhalation, Harmful: may cause lung damage if swallowed, Irritating to skin and Vapours may cause drowsiness and dizziness. The substance was detected in extracts from 2 products but at low concentrations. 3,5,5-Trimethyl-2-cyclohexen-1-one (=isophorone), CAS no. 78-59-1, is classified Carc.Cat.3;R40 Xn;R21/22 Xi;R36/37, i.e. Limited evidence of carcinogenic effects, Harmful in contact with skin and if swallowed, Irritating to eyes and respiratory system. The substance was detected in extracts from 3 products at moderate concentrations. Due to the strict classification the substance has been included for further evaluation. Vanillin, CAS no. 121-33-5, is not classified in the List of dangerous substances. The substance was detected in extracts from 12 out of 15 products and even if the concentrations were low the substance has been selected for further evaluation. Xylene, CAS no. 95-47-6, is classified R10 Xn;R20/21 Xi;R38, i.e. Flammable, Harmful by inhalation and in contact with skin and Irritating to skin. The substance was detected in extracts from 2 products at trace level. Summary of selected substances For evaluation of individual substances the following substances have been selected in co-operation with the Danish Environmental Protection Agency. Of organic substances: 2-Butoxyethanol 2-(2-Butoxyethoxy)ethanol Cyclohexanone 2,6-Dimethoxybenzoquinone 3,6-Dimethyl-1,4-dioxan-2,5-dione (3,6-Dimethyl-2,5-dioxo-1,4-dioxane) 2-Ethoxyethanol (2-(2-Ethoxyethoxy)ethanol Formamide Furfural 4-Hydroxy-3,5-dimethoxy-benzaldehyde N-Methyl-2-pyrrolidone 3,5,5-Trimethyl-2-cyclohexen-1-one (= Isophorone) Vanillin Of inorganic substances: Barium 4 Health assessment
A screening of potential health hasardous effects from substances released (migrated) from surface treated wooden toys has been performed. A literature screening on the substances identified at the qualitative migration examinations is performed. It has the purpose to make sure that the substances focused on at the qualitative analyses are the most relevant substances. After the quantitative analyses are carried out the appeared results are evaluated. Data on individual substances such as NOAEL, LOAEL or other relevant data are used to the extent they are available. Alternatively, QSAR data are used for substances where no data are found. 4.1 IntroductionTo the assessment of the health risk from the use of wooden toys, a selection of effects on health by the detected substances are evaluated in relation to the relevant exposure duration and exposure route to the consumer of the toy. 4.1.1 Exposure durationThe average potential exposure duration, i.e. the period where the child is awake and not eating is 10 hours/day for children under 2 years of age and 10.7 hours/day for children between 2 and 3 years of age in an American study (Kiss 2001). In a Dutch study, the period was 8 to 9 hours for children between 3 and 36 months of age (Groot et al. 1998, table 5). The oral intake strongly depends on the exposure duration, i.e. how often and for how long the toy is placed in the mouth. That question has been studied especially in connection to the assessment of the exposure to phthalates in plastic toys. Studies on the behaviour of children mouthing subjects including fingers have for instance been performed in several studies. Groot et al. (1998) studied 42 children between 3 and 36 months of age and Juberg et al. (2001) studied 385 children between 0 and 36 months of age. Their studies show that the youngest children mouth subjects more often than older children do (table 5). Table 5 Average period of toys in the mouth (minutes) and exposure duration (hours) per day (Groot et al. 1998)
*: Other than comforter, teething ring etc. In an American observation study of the mouthing behaviour of children 0 to 6 years of age, the behaviour of 491 children was studied by parents and 169 children at the age if 0 to 36 months by trained observers. In conclusion it was observed that for all objects mouthed except pacifiers the average mouthing period was 70 minutes/day for children between 3 months and 1 year, 48 minutes for children between 1 and 2 years, and 37 minutes for children between 2 and 3 years of age (Kiss 2001) In a statistical analysis of the same numbers, Greene (2002) has examined the behaviour in 169 children between 3 and 36 months of age. He found a skewness in exposure as few children mouthed objects for a long period and many children mouthed objects for a short time or not at all. This observation was supported by e.g. a previous American study on the play activity of children. The study finds average times of 46 to 70 minutes and 90 percentiles for play activity of children (1 to 17 years of age) between 120 and 255 minutes per day (US-EPA 1997, US-EPA 2002). In a British study on the mouthing behaviour of children, 236 children at the age of 1 month to5 years were studied (DTI 2002). The study found a peak at the age of 9 months when only observing the period of 0 to 3 years of age (table 6). Table 6 Estimated maximum daily oral exposure (hours : min) (DTI 2002)
In the DTI study 50% of the toys were made of plastic and 6% of wood. However, that distribution depends on the availability of toys and can therefore not be included in the evaluation. The exposure duration for playing with wooden toys may vary considerably but most common is assumed to be several short-term uses that may vary from a few to many minutes. This study especially focuses on the oral exposure, i.e. exposure after mouthing the toy and sucked on it, biting it etc. by children of 0 to 36 months of age. In the CSTEE evaluation on exposure to plasticisers was recommended an exposure period of 3 hours in the assessment of migrated phthalates to children (CSTEE 1998). Based on the above observations/studies which indicate that children at the age of 3 to 36 months mouth toys at an average of 70 minutes and at maximum (90 percentile) of 3 hours and the recommendation of CSTEE, the assessment in this project has selected an exposure duration of 3 hours/day as a reasonable worst case scenario. 4.1.2 Body weightSince the exposure is given in mg/kg body weight it is evaluated which body weight that would be an appropriate average for children. Studies by Bremmer and van Veen (2002) and US-EPA (2002) have been obtained. The results are presented in table 7, below). Table 7 Body weight of children
Whether the differences between Dutch and American children is actual or a result of the selection of children and variation in numbers are unknown. The study by Bremmer and van Veen covers children of "well educated" parents and a small number whereas the American study covers a wider section of children and a far higher number of children. In the CSTEE evaluation of the exposure to plasticisers was recommended a body weight of 8 kg in the assessment of migrated phthalates on children (CSTEE 1998). As the study especially relates to very small children that weight may be a little too low for the target group of this project. The study further is indicated to be based on the Bremmer and van Veen / Groot et al. studies. In regard of realistic worst case, this project therefore in the calculations uses an assumed body weight of 10 kg for children based on the far more extensive American study (US-EPA 2002) and a peak of mouthing behaviour at 9 months of age (DTI 2002). That wooden toys may be purchased or intended for a little older child does not exclude the possibility that younger children may or will get hold of the toys. 4.1.3 Exposure routeDuring the screening it was realised that some the detected substances were volatile substances and an exposure via inhalation possible. However, this exposure route is considered to be less essential in this context. Exposure of the skin (dermal exposure) is considered relevant as wooden toys are specifically intended to "handling". The primary exposure is to the skin of the hands. Intake vi the mouth (oral intake) is assumed potentially to present the largest concern for children under 3 years of age, partly because this age group is known to mouth objects but they may also suck on fingers after having touched the toy and thereby transfer potential contamination from the hands to the mouth. 4.1.4 UptakeUptake via inhalation, oral or dermal exposure will be substance specific and therefore depend on which substances that are detected released from the toy. If no information was available on the specific uptake of the individual substances via inhalation, by dermal contact or via moth / mucous membranes an uptake of 100% is assumed. In the study, 14 specific substances have been selected in co-operation with the Danish Environmental Protection Agency. The selection is based on the classification of the substances, number of detections and the level of the measured concentration in the saliva extracts. The selected substances have been reviewed individually after a presentation of the assessment method (cf. below). Each of the selected substances has been identified by its common name and CAS no. for unambiguously identification. The most common synonyms are stated and furthermore is mentioned:
Finally, an assessment of the amount of detected released substances has been carried out. This has been performed by calculating / estimating the uptake based on the duration of exposure and the body weight of the person (amount/kg body weight/day). If possible, one of the established values for tolerable daily intake (TDI, ADI or RfD) is used for evaluation of the exposure by comparing the values with the obtained analysis results used to estimate the exposure. The used uncertainty factors (safety factors) are mentioned in the text. In case more TDI, ADI or RfD values exist, the lowest value is preferred. If no TDI, ADI, RfD values are available, a comparison to a concentration where no adverse effects are observed (NOAEL: No Observed Adverse Effect Level) from a relevant long-term study is used. The procedure is mentioned at the individual substances. 4.2 Exposure scenarios4.2.1 IntroductionThe exposure to the consumer from wooden toys will vary considerably according to use duration, handling or area of contact and duration of direct contact. To evaluate the exposure in a standardised manner, theoretical exposure scenarios have been derived to illustrate the worst possible but realistic exposures. To evaluate the exposure of consumers, the following scenario has been derived for the exposure via the mouth (oral exposure). Oral exposure is based on measurements of the substance in extractions of artificial saliva. It is assumed that the amount of substance released (migrated) from the toys during an average time of 2 hour extraction x 1.5 = 3 hours corresponds to the potential maximum oral exposure per day. (Note that the extraction is performed for 2 hours according to the standard EN71-3 and recalculated to 3 hours exposure period as the target period). 4.2.2 MethodologyFor the chemical substances detected as migrated to saliva from the wooden toys, an evaluation of which substances appeared to be the most interesting (cf. section 3). Data on the individual substances are retrieved to perform a health hazard evaluation based on known information from previously prepared Danish or foreign monographs, etc. The obtained data for threshold limit values or toxicity are then compared to the concentrations estimated in the used scenario. The methodology used is approximately the same as recommended in connection to risk assessments in the European Union (EU) described in the Technical Guidance Document, TGD (EC 2003). In the TGD the potential risk to humans is estimated as the ratio between the predicted no-effect concentration (no-adverse-effect level, NOAEL) and the predicted exposure concentration in the surrounding environment (Predicted Environmental Concentration, PEC), i.e. NOAEL / PEC or the estimated uptake in the exposed humans. NOAEL is based on mammalian data that is often not humans but typically rats, mice and rabbits. Therefore, a safety factor is introduced to cover differences extrapolating from other animals to humans. This is expressed either by attaching a fixed safety factor (SF) or by expressing the margin of safety (MOS) which represents the distance between NOAEL and the estimated uptake. Typically MOS is preferred to be above 100. The safety factor is interpreted as a margin of safety applied to a NOAEL to produce a value below which exposures are presumed to be without health risk. The safety factor is traditionally composed of a factor 10 for extrapolation between species (animal to human, interspecies variation), a factor 10 to protect the most sensitive individuals of the population (intraspecies variation) such as e.g. children. A third factor is applied depending on the data and may vary. For instance 10 is used if LOAEL (lowest observed adverse effect level) is used instead of NOAEL or using subchronic data instead of chronic data. The total safety factor is a result from multiplication of the three factors. The effect level divided with the safety factor is used to evaluate whether there is reason of concern (concern level) or a further examination of methodology or data is necessary. Thus the assessment may be expressed on basis of concentration divided with the safety factor (such as e.g. ADI, TDI, RfD, RfC) or MOS. In modern society is used many chemical products. It can be difficult for the single consumer to keep track of them all. The handling of the chemical substances is therefore regulated on basis of an extended chemical legislation. In connection with this project no values have been derived for chemical substances already evaluated by national or international experts in the field. The classification authorised in Denmark (Miljøministeriet 2002), which is an implementation of EU classification (28th amendment to EU directive 67/548/EEC), is used in the evaluation. The amendments performed in the 29th amendment and adopted in Directive 2004/73/EC (EC 2004) and not yet implemented in Denmark are included, however, as the implementation may be expected within the near future. For the evaluation of the individual substances is used the threshold limit values mentioned above and explained below. The threshold limit value (TLV) valid for the working environment in Denmark (AT 2002) is generally not used as it is only valid for the working environment and does not cover the consumer at home. The TLV value is presented for information and comparison, if available. Other limit values included in the health evaluation were:
The effect level for each piece of wooden toy is based on evaluations of individual substances. The established Danish threshold limit values are used when they exist. When no Danish threshold limit values exist, foreign threshold limit values are used including their background if available. 4.2.3 Exposure via the mouthBy oral exposure the absorption takes place after release (migration) of the compounds from the wooden toy and mixing with the saliva. Uptake is assumed to takes place over the epithelium in the mouth cavity or in the gastro-intestinal-tract. As basis for the assessment of the oral intake is used the general equations described in relevant references (TGD 2003, OECD 1993, Bremmer and van Veen 2002). The equations are then adjusted to an equation fit for the actual scenario with measurements of chemical substances migrated to artificial saliva. The weight of the surface area of the toy (10 cm2 x sample depth x density) which the child may have in the mouth for a prolonged time is used in the exposure assessment.
where
As basis is assumed that a child plays with the wooden toy one or several times each day. The total oral exposure duration is assumed to be maximum 180 minutes or 3 hour/day. The child's body weight is set to 10 kg. The analysis results represent the amount migrated to the saliva extracts after 2 hours of extraction according to the standard EN 71-3. The amount is recalculated to release per hour and multiplied by 3 (3 hours average). The migration is calculated as release per cm2. According to the standard EN71-10 the sample should be 3 mm deep. Using the density of the wood used to make the wooden toy (based on information on the density of dry wood) the released amount per cm2 is calculated. Table 8 Density of dry wood, 12% moisture (Danish Technological Institute, Wood Technology, Træteknik 2004)
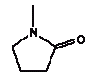
The released amount/cm2 is the measured concentration x sample depth x density of sample: Qoral (μg/cm2) = Fcmigr (μg/g) sample depth (0.3 cm) density (0.7 g/cm3) The calculations are performed as released substance per 10 cm2/hour (based on the analysis extraction over 2 hours) exposure duration/day absorption/kg body weight: Thus oral absorption = Weight of exposed toy μg substance released/hour 1/2 (hours) exposure duration (3 hour/day) (% absorption/100%) / 10 (kg) = μg/kg body weight per day. It is noted that oral intake also may take place by hand-to-mouth, i.e. hands or fingers, which have touched the product, and then are placed in the mouth. This may result in a transfer of substance from the fingers to the mouth. As information in the reference literature (Bremmer and van Veen 2002, Green 2002, Kiss 2001) indicate that hand-to-mouth averages 3 to 10 minutes that part is considered included in the selected exposure period of 3 hour. Absorption After exposure to the mouth cavity the chemical substance has to pass the epithelium before actual absorption may take place. Data for oral absorption have not been available for all of the selected substances. The oral uptake across the epithelium (oral absorption) is therefore estimated or set to 100% for many for the substances (TGD, EC 2003). 4.3 Evaluation of individual substancesFor the evaluation of individual substances the below mentioned chemical substances have been selected in co-operation with the Danish Environmental Protection Agency. Of organic substances: 2-Butoxyethanol 2-(2-Butoxyethoxy)ethanol Cyclohexanone 2,6-Dimethoxybenzoquinone 3,6-Dimethyl-1,4-dioxane-2,5-dione (3,6-Dimethyl-2,5-dioxo-1,4-dioxane) 2-Ethoxyethanol 2-(2-Ethoxyethoxy)ethanol Formamide Furfural 4-Hydroxy-3,5-dimethoxy-benzaldehyde N.Methyl-2-pyrrolidone 3,5,5-Trimethyl-2-cyclohexen-1-one (= Isophorone) Vanillin Of inorganic substances: Barium 4.3.1 2-ButoxyethanolIdentification
The melting point is –74.8°C. The boiling point is 168.4°C (DOW 1990). The vapour pressure is 117 Pa at 25°C (0.88 mmHg, DOW 1990). The water solubility is 1 kg/l at 25°C (miscible, DOW 1990). The partition coefficient is log Kow is experimentally determined to 0.83 (Hansch et al. 1995). Use 2-Butoxyethanol is used as a solvent in surface coatings and in vinyl and acrylic paint (CICAD 1998). Further is mentioned the use as solvent in printing inks and colorants in the EU risk assessment report, draft 2004 (ECB 2004). Classification 2-Butoxyethanol is classified in the List of dangerous substances (Miljøministeriet 2002):
Effects on health 2-Butoxyethanol is moderately acute toxic, irritating to eyes and skin (but not a skin sensitizer). Eye irritation examinations showed that 30 and 70% concentrations of the substances were irritating to the eyes with increasing irritation with corresponding increasing exposure duration. The skin irritation was mild at 4 hours of exposure of rabbit skin, but the irritation increased with increasing exposure duration (CICAD 1998). The effects have mostly been registered as a haemolytic activity of 2-butoxy-ethanol. The effect was dependent on age with older rats as the most sensitive (CICAD 1998). Acute toxicity:
In a subchronic 90 days inhalation study, rats were exposed to 2-butoxyethanol at concentrations of 0, 5, 25, or 77 ppm for 6 hours/day, 5 days/week for 13 weeks. Based on haematotoxic effects, the NOAEL and LOAEL were 25 ppm (121 mg/m3) and 77 ppm (372 mg/m3), respectively (Dodd et al. 1983). In a study on developmental effects, pregnant rats were exposed to 2-butoxyethanol at 0, 25, 50, 100 or 200 ppm (35 rats per group) for 6 hours/day on days 6 to 15 of gestation. Based on haematotoxic effects the NOAEL and LOAEL were 50 ppm (242 mg/m3) and 100 ppm (483 mg/m3), respectively (Tyl et al. 1984). In a 13 weeks study with rats, groups of 10 of each sex were exposed through the drinking water. Based on the water consumption, the male rats were exposed to 0, 69, 129, 281, 367 or 452 mg/kg/day and female rats to 0, 82, 151, 304, 363 or 470 mg/kg/day. Based on effects of the blood parameter and liver, which were observed at even the lowest concentration, LOAEL was 69 mg/kg/d for males and 82 mg/kg/d for females. When water consumption and body weight from the last week of the exposure is used, LOAEL is converted into 55 mg/kg/d for males and 59 mg/kg/d for females. NOAEL could not be determined in the examination (NTP 1993, IRIS 2004). The LOAEL value from the rat study of 55 mg/kg bw/day was recalculated assuming 1.5 l /day drinking water and a body weight of 60 kg to a human equivalent dose, HEC, of 5.1 mg/kg bw/day (IRIS 2004). In the EU risk assessment report, the same value was recalculated assuming 2 l/day of drinking water and a body weight of 70 kg to a LOAEL (HEC) 7.6 mg/kg bw/day (ECB 2004). Because several studies demonstrates that rats are much more sensitive than human to the haemolytical effects of 2-butoxyethanol LOAEL values is accepted as NOAEL (ECB 2004). 2-Butoxyethanol has been evaluated as potential human carcinogen, Group C (IRIS 2004). Threshold limit values The threshold limit value for the working environment is 20 ppm corresponding to 98 mg/m3 with skin notation, i.e. the substance may penetrate the skin (AT 2002). The C-value is 0.04 mg/m3 (B-værdivejledningen, Miljøstyrelsen 2002). The inhalation RfC value is 13 mg/m3. The value is based on a subchronic rat inhalation study (Tyl et al. 1984, cf. above). The value is based on NOAEL 242 mg/m3 and calculated with a safety factor 10, 6/24 in order to convert 6 hours' exposure to 24 hours per day, a conversion from rat to human (inhalation rate for rat 0.16 m3/d and for human 22 m3/d, the body weight of rat 0.215 kg and for human 64 kg) (CICAD 1998). The RfC calculated using the mentioned variables is then: RfC = (242/10) (6/24) [(0.16/0.215)/(22/64)] = 13.1 mg/m3. Oral RfD value is 0.5 mg/kg bw/day. The value is based on a 13-week of subchronic study where haematological effects were found as the most sensitive endpoint with a LOAEL of 55 to 59 mg/kg/day for rats (NTP 1993, cf. above). US-EPA used the PBPK model of Corley (1994) to "back-calculate" human equivalent dose (HEC) of 5.1 mg/kg bw/day for humans. Using a safety factor of 10 for intraspecies variation an orall RFD value of 0.5 kg/kg bw/day was derived (IRIS 2004). Absorption 2-Butoxyethanol is easily absorbed after inhalation, or by oral or dermal exposure (CICAD1998). Consequently, an absorption of 100% has been used. In the ECB (2004), Risk Assessment Report draft is used 61% for absorption via inhalation and 30% dermal absorption. No information on oral absorption is presented. Assessment The assessment of oral exposure is based on exposure for 3 hours to a child of 10 kg body weight (bw). The absorption is set to 100%. Calculation example (lab.no. 31342-4): Migration: 322 0.3 (cm) 0.7 (g/cm3) = 67.6 μg/cm2 Total migration: 67.6 10 / 2 = 338 μg/10 cm2/hour Oral uptake: 338 3 (h) 1 (100%) / 10 = 101 μg/kg bw/day Table 9 Uptake of 2-butoxyethanol by oral exposure
2-Butoxyethanol was detected in 12 wooden toys. The calculated uptake is equal to or more than 5 times below the RfD value of 500 μg/kg lgv/day. Using the NOAEL value 7.6 mg/kg bw/day and the highest estimated uptake the margin of safety (MOS) is equal to or more than: 7.6/0.1 = 76. Conclusion From the above table is observed that none of the amounts taken up by mouthing the wooden toy result in exceeding the RfD value. Thus the substance is assessed not to cause a health risk to the consumer The lowest margin of safety is lower than the 100 usually preferred as the lowest MOS. Thus, the migration of the substance in at least 1 product may cause health concern. 4.3.2 2-(2-Butoxyethoxy)-ethanolIdentification:
The melting point is -68°C. The boiling point is 230°C at 1013 hPa. The water solubility is high (miscible with water). The vapour pressure is 2.7 Pa at 20°C. The octanol/water partition coefficient is measured to log Kow 0.56. All data are from the EU risk assessment of the substance (RAR vol. 2, ECB 2000). Classification 2-(2-Butoxyethoxy)ethanol is classified in the List of dangerous substances (Miljøministeriet 2002):
Use The substance belongs to the group of glycol ethers that is mainly used as solvents. 2-(2-Butoxyethoxy)ethanol is used as solvent in paints, dyes and colorants in both aqueous and non-aqueous systems thereof a large part in surface coatings. The amount in paint is noted to be maximum 5% but typically at 1 to 4%. Besides the substance is used in a series of other industrial processes and products (ECB 2000). Effects on health 2-(2-Butoxyethoxy)ethanol has a low acute toxicity by oral and dermal exposure routes. Some data on acute toxicity have been found (IUCLID 2000). Of these are mentioned:
By acute exposure no mortalities were observed even at exposure to saturated vapours (IUCLID 2000). The substance is evaluated to be eye irritating and may be skin irritating after repeated exposure to high concentrations of 2000 mg/kg bw/day (ECB 2000). Of studies with prolonged exposure was found a 5-week subacute study on rats exposed to 0, 13, 39 and 117 mg/m3 for 6 hours/day, 5 days/week for 5 weeks. Based on effects on the liver NOAEL was 39 mg/m3. Because the same effects were not observed in a 90-day study where rats were exposed to 0, 13, 40 and 94 mg/m3, 6 hours/day, 5 days/week the NOAEL was established at 94 mg/m3 (ECB 2000). In a 13 weeks subchronic dermal study rats were exposed for 13 weeks at 0, 200, 600 and 2000 mg/kg bw/day. No systemic effects were observed at the highest concentration even if concentration dependent skin irritations (erythema) were observed. NOAEL was established at 2000 mg/kg bw/day (ECB 2000). The oral toxicity is based on a one-generation reproduction study by Nolen et al. (1985). The rats were administered the substance via gavage directly into the stomach at 0, 250, 500 and 1000 mg/kg bw/day. Since no effects on fertility were observed NOAEL for maternal toxicity was established at the highest test dosis of 1000 mg/kg bw/day. Based on reduced body weight gain in rat pups a reproduction toxicity NOAEL of 500 mg/kg bw/day was established (ECB 2000). Threshold limit values The threshold limit value for the working environment is 100 mg/m3 (AT 2002). The C-value is 0.02 mg/m3 (Miljøstyrelsen 2002). Absorption The dermal absorption has been studied. In rats 2-(2-butoxyethoxy)-ethanol relatively easy is absorbed through the skin with between 30 and 50% of the applied radioactive labelled substance (Boatman 1993). No values for oral absorption were available but presuming it is easier than via the skin, a 100% absorption by oral exposure is assumed. Assessment The assessment of oral exposure is based on the exposure for 3 hours of a child of 10 kg body weight (bw). The absorption is set to 100%. Table 10. Uptake of 2-(2-Butoxyethoxy)ethanol by oral exposure
2-(2-Butoxyethoxy)ethanol was detected in the extracts from 5 products Because no threshold limit values for effects based on amount taken up after oral exposure NOAEL is used for the calculation of the margin of safety (MOS). Using a NOAEL of 500 mg/kg bw/day MOS is:(500/0.0167 =) at least 30000. Conclusion With a margin of safety of at least 30000, 2-(2-butoxyethoxy)ethanol is assessed not to cause any health concern to the consumers (children mouthing the wooden toy). 4.3.3 CyclohexanoneIdentification
The melting point is -31°C. The boiling point is 155°C (Budavari 1996). The vapour pressure is 577 Pa at 25°C (4.3 mmHg, Daubert and Danner 1985). The water solubility is 25 g/l at 25°C (Yalkowsky and Dannenfelser 1992). The partition coefficient log Kow is experimentally determined to 0.81(Hansch et al. 1995). Use Cyclohexanone is used in the chemical industry for organic synthesis, particularly in the production of adipic acid and caprolactam (ca. 95%), polyvinyl chloride and its copolymers, and methacrylate ester polymers. Classification Cyclohexanone is adopted on the List of dangerous substances (Miljøministeriet 2002) and classified:
Effects on health Acute toxicity:
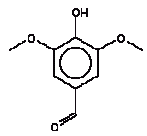
For humans was observed that the threshold for irritation to the nasal mucous membranes was 0.28 mg/l of air (280 mg/m3 or about 70 ppm). The value was seconded by irritation of eye, nasal, and throat at 0.362 mg/l of air (362 mg/m3 or about 90 ppm). A second exposure 2 weeks after the initial series indicated an increase in the sensory irritation threshold. In this series, the only response recovered was throat irritation at 0.547 mg/l of air (547 mg/m3 or about 136 ppm) (OECD 1996). Humans exposed for only 3 to 5 minutes found 50 and 75 ppm (200-301 mg/m3 air) irritating to the eyes, nose and throat while a concentration of 25 ppm was unobjectionable (Nelson et al. 1994). Cyclohexanone exhibits low to slight acute toxicity by the oral and inhalation exposure routes and is moderately toxic by the dermal route. Cyclohexanone is an eye and skin irritant but does not induce skin sensitisation. Upon repeated administration to rats of cyclohexanone in drinking water, the NOAEL was 4700 ppm after 25 weeks and the LOAEL was 3300 ppm after 2 years. Effects at higher concentrations were primarily body weight decreases. The NOAEL in repeated dose inhalation studies was 100-190 ppm. Those values were based on either gray mottling of the lungs or ocular irritation and degenerative changes in the liver and kidney at higher concentrations. However, the NOAEL in those studies was not confirmed in later and better inhalation studies where for reproductive and developmental effects NOAEL values of 650-1000 ppm were observed. In a two-generation reproduction study, decreased fertility was observed in rats exposed via inhalation at 1400 ppm but not at 500 ppm. The effect was found to be reversible following a post-exposure recovery period (IRIS 2004). In a chronic rat oral study, rats in groups of 52 animals per dose were exposed to cyclohexanone in drinking water at 3300, 6500, 13000 and 25000 ppm. Based on mortality and decrease in body weight a LOAEL of 6500 ppm corresponding to 910 mg/kg bw/day was found. NOAEL was 3300 ppm in the drinking water corresponding to 462 mg/kg bw/day (Lijinski and Kovatch 1986). Threshold limit values The threshold limit value (TLV) is 10 ppm equivalent to 40 mg/m3 with skin notation (H), i.e. the substance may penetrate the skin (AT 2002) The C-value is 0.1 mg/m3 (B-værdivejledningen, Miljøstyrelsen 2002). The oral RfD value is 5 mg/kg bw/day. In a chronic oral rat study was found a NOAEL of 462 mg/kg bw/day (cf. Lijinski and Kovatch 1986 above). Applying a safety factor of 100 (10 for inter- and 10 for intraspecies extrapolation) derived an oral RfD value of 5 mg/kg bw/day. TDI (tolerable daily intake) value is 4.6 mg/kg bw/day (Baars et al. 2001). Assessment Cyclohexanone was detected in the saliva extracts from 2 wooden toys. The calculated uptake by oral exposure is summarised in the table below. The assessment of oral exposure is based on the exposure for 3 hours of a child of 10 kg body weight (bw). The absorption is set to 100%. Table 11. Uptake of cyclohexanone by oral exposure
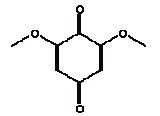
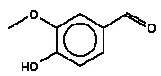
No exceeding of the TDI value was found (the margin was a factor of approx. 500 to the TDI value of 4.6 and the RfD value of 5 mg/kg bw/day). Using a NOAEL value of 462 mg/kg bw/day the margin of safety (MOS) is >54000. Conclusion With a margin of safety of more than 54000, cyclohexanone is assessed not to cause health concern to the consumers (children mouthing the wooden toy). 4.3.4 2,6 DimethoxybenzoquinoneIdentification:
The melting point is 256°C. The boiling point is estimated to 286°C. The water solubility is estimated to 70600 mg/l at 25°C (EPI). The vapour pressure is estimated to 3 mPa at 25°C (2.2 x 10-5 mmHg). The octanol/water partition coefficient is measured to log Kow –0.06 (Hansch et al. 1995). Classification 2,6-Dimethoxybenzoquinone is not classified in the List of dangerous substances (Miljøministeriet 2002). However, the substance is on the Danish EPA's Advisory list for self-classification with the classification:
Use No information on the use of the substance has been available. The few available information on the substance indicates that the substance occurs naturally in several plants. Quinones and derivated substances are used in a series of products among others in the manufacture of dyes (IPCS 1994). Effects on health Only a few informations were found on 2,6-dimethoxybenzoquinone. Based on information on quinones or hydroquinones. They seem to have similar effects. Quinones such as 2,6-dimethoxybenzoquinone are the main reason to contact dermatitis in certain plants and are regarded as common in leaf trichomes and pollen (Lovell 1993, Rasmussen 1986, MacAuley 1997). Quinone (CAS no. 106-51-4) may cause dermatitis (Budavari 1996). Therefore, information has been searched on quinone and hydroquinones, which are analogous structures. It is discovered that hydroquinones can be metabolised to 1,4-benzoquinone. For hydroquinones the acute oral LD50 values vary between 300 and 1300 mg/kg bw. Of prolonged studies a 13 weeks rat study was found with oral administration of hydroquinone. Kidney damages, reduced body weight and adverse effects to the central nervous system were observed. NOAEL was 20 mg/kg bw/day (IPCS 1994). Hydroquinones are sensitising to both humans and animals (IPCS 1994). Quinone appears to be more toxic than hydroquinone. Acute data indicate an acute oral toxicity of 25 to 50 mg/kg. No data on prolonged studies were available (HSDB 2004). Threshold limit values No threshold limit values, ADI values or similar for 2,6-dimethoxy-benzoquinone are available. For p-benzoquinone (quinone, CAS no. 106-51-4) the threshold limit value for the working environment is 0.1 ppm corresponding to 0.4 mg/m3 (AT 2002). For hydroquinones (CAS no. 123-31-9, 1,4-benzenediol) the threshold limit value for the working environment is 2 mg/m3 with notation LK. L means that the threshold limit value is a ceiling value that must not be exceeded at any time. K means that the substance is adopted on the list of substances considered to be carcinogenic (AT 2002). Absorption No values on the absorption of 2,6-dimethoxybenzoquinone are found. Hydroquinones are readily absorbed through the skin where it is metabolised to 1,4-benzoquinone and distributed to all tissues (IPCS 1994). Therefore 100% absorption is assumed. Assessment The assessment of oral exposure is based on the exposure for 3 hours of a child of 10 kg body weight (bw). The absorption is set to 100%. Table 12. Uptake of 2,6-dimethoxybenzoquinone by oral exposure
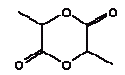
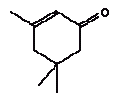
2,6-Dimethoxybenzoquinone was detected in the extracts from 7 products. The estimated uptake by oral exposure is calculated to vary between 1.5 and 10 μg/kg bw/day. Since no threshold limit values for effects from amounts taken up by the body or NOAEL values were available then based on the available information an actual assessment can not be performed The self-classification is based on modelled molecular structure analysis and together with the suspicion that the substance is released from the wood and not from the surface treatment the detection is not considered an immediate cause for concern. However, it should be noted that quinones are recognised allergenic substances that may cause contact dermatitis. The determined concentrations may not result in problems unless the child is particularly sensitive or already sensitive to the substance. 4.3.5 3,6-Dimethyl-1,4-dioxane-2,5-dione3,6-Dimethyl-1,4-dioxane-2,5-dione and 3,6-dimethyl-2,5-dioxo-1,4-dioxane are discussed together. The names indicate that it may be the same substance although they have individual CAS and EINECS numbers. The differences may be only stereoisomeric. Identification:
Identification:
Because no specific data could be found the physico-chemical data are estimated based on the molecular structure. As the molecular structure is identical for the two substances the estimated values are the same. The melting point is estimated to 26°C. The boiling point is estimated to 308°C. The water solubility is estimated to 3200 mg/l at 25°C. The vapour pressure is estimated to 0,17 Pa at 25°C (0,00126 mmHg). The octanol/water partition coefficient is estimated to log Kow 1.6 (EPI). Classification Neither 3,6-dimethyl-1,4-dioxane-2,5-dione nor 3,6-dimethyl-2,5-dioxo-1,4-dioxane is classified in the List of dangerous substances (Miljøministeriet 2002). However, both substances are included in the Danish Environmental Protection Agency's Advisory list for self-classification of dangerous substances with the classification:
Use No information on use is available. Since the chemical compound is detected in almost all extracts a possible explanation could be that the substance/substances are occurring naturally in wood. Effects on health No data on the toxicity of the substance are available. Threshold limit values No threshold limit values, ADI or corresponding values are found. Absorption No values are found and therefore 100% absorption is assumed. Assessment The assessment of oral exposure is based on the exposure for 3 hours of a child of 10 kg body weight (bw). The absorption is set to 100%. Table 13. Uptake of 3,6-Dimethyl-1,4-dioxane-2,5-dione by oral exposure
The substance was detected in the extracts from 14 out of 15 products. The estimated uptake by oral exposure is calculated to vary between 3 and 31 μg/kg bw/day. No threshold limit values for effects from amounts taken up by the body or NOAEL values were available. Thus based on the available information an actual assessment can not be performed 4.3.6 2-EthoxyethanolIdentification:
The melting point is -70°C. The boiling point is 135°C. The water solubility is high (miscible with water, DOW 1990). The vapour pressure is 706 Pa at 25°C (5.3 mmHg, Daubert and Danner 1989). The octanol/water partition coefficient is measured to log Kow -0.32 (Hansch et al. 1995). Classification 2-Ethoxyethanol is adopted on the List of dangerous substances and classified (Miljøministeriet 2002):
Use The substance is used for instance in paint, dye and lacquer industry as solvent for nitrocellulose and lacquers and to increase the stability of emulsions. Effects on health Some data on acute toxicity have been found. Of these are mentioned:
Of studies with prolonged exposure a 13-week inhalation study is found where rats were exposed via inhalation to 0, 25, 103 and 403 ppm corresponding to 0, 92, 380 and 1485 mg/m3 for 6 hours/day, 5 days/week. At the highest concentration was observed a significant decrease in the weight of pituitary in males and decreased leukocyte count in females. NOAEL is therefore set to 103 ppm (380 mg/m3) (Barbee et al. 1984). In a 13 weeks study dogs were exposed by oral administration of the doses 0, 46, 93 and 186 mg/kg bw/day. Based on observation of testicular oedema, a reduction in haemoglobin concentration and haematocrit value a NOAEL was set to 93 mg/kg bw/day (IUCLID 2000). In studies on reproduction toxicity with oral administration severe effects on testicles, spermatid count and semen quality at doses of 300 mg/kg bw/day and above (IUCLID 2000). In a 14 weeks fertility study where mice were exposed for 18 days before mating to the test substance in drinking water at 750-2600 mg/kg bw/day. Mice of both sexes were infertile at 2600 mg/kg lgv/day, the fertility in both sexes was reduced at 1500 mg/kg bw/day and at 750 mg/kg bw/day no effects on fertility was observed (Lamb et al. 1984). In a study with exposure to pregnant female rats via inhalation in the gestation period, increased mortality in the foetuses, deformities and other effects were observed. NOAEL for teratogenic effects was set to 10 ppm corresponding to 36 mg/m3 (IUCLID 2000). Threshold limit values The threshold limit value for the working environment is 5 ppm corresponding to 18.5 mg/m3 with notation H. H indicates that the substance may penetrate the skin (AT 2002). The C-value is 0.2 mg/m3. The value is under revision, a new proposal is 0.01 mg/m3 (Miljøstyrelsen 2002). The inhalation reference concentration, RfC, is set by US-EPA to 0.2 mg/m3. The value is derived from a subchronic rat study with NOAEL 380 mg/m3 (Barbee et al. 1984). The NOAEL was recalculated from 6 hours/day, 5 days/week to 24 hours/day for 7 days/week, i.e. NOAEL adjusted to (380 6/24 hours 5/7 days =) 68 mg/m3. Using a safety factor of 300 (3 for interspecies and 10 for intraspecies variation and 10 for the use of a subchronic study) RfC is derived at 68/300 = 0.2 mg/m3 (IRIS 2004). Absorption The substance can be absorbed via inhalation, the skin and via gastro-intestinal tract. After oral intake of the substance 76 to 80% was excreted in the urine within 96 hours (IPCS 1990). Therefore 100% absorption is assumed. Assessment The assessment of oral exposure is based on the exposure for 3 hours of a child of 10 kg body weight (bw). The absorption is set to 100%. Table 14. Uptake of 2-ethoxyethanol by oral exposure
2-Ethoxyethanol was detected in extracts from 2 products. Because no limit values for effects from amounts taken up in the body are found NOAEL is used to calculate the margin of safety (MOS). Using a NOAEL of 94 mg/kg bw/day MOS is at least: 94/0.0054 = 17400. Conclusion Based on a high MOS value 2-ethoxyethanol is considered not to pose a health risk to the consumer. 4.3.7 2-(2-Ethoxyethoxy)-ethanolIdentification:
The melting point is -76°C. The boiling point is 196°C. The water solubility is high (miscible with water, Riddick et al. 1986). The vapour pressure is 16.8 Pa at 25°C (0.126 mmHg, Daubert and Danner 1989). The octanol/water partition coefficient is measured to log Kow -0.54 (Funasaki et al. 1984). Classification 2-(2-Ethoxyethoxy)-ethanol is not classified in the List of dangerous substances (Miljøministeriet 2002). Use 2-(2-Ethoxyethoxy)-ethanol is used as solvent in paint, dyes and lacquers (Buadavari 1996). Effects on health Some data on acute toxicity have been available. Of these are mentioned:
2-(2-Ethoxyethoxy)-ethanol is a moderate irritant to eyes (Grant 1986). In a dermal study rabbits were applied 0.1, 0.3, 1 and 3 ml/kg/day on the skin 5 times/week for 90 days. Based on observations of kidney damages NOAEL was set to 0.3 ml/kg/day or approx. 300 mg/kg bw/day (IUCLID 2000). In a 30 days oral rat study 2-(2-ethoxyethoxy)-ethanol was administered in the drinking water at doses from 410 to 3200 mg/kg bw/day. Reduced weight gain was observed at 1830 mg/kg bw/day and unspecified micropathological changes at all doses. NOAEL is set to 590 mg/kg bw/day (IUCLID 2000). In a 90 days study, pigs were orally administered the test substance at the doses 0, 167, 500 and 1500 mg/kg. Damages to the kidney and changes to the blood picture at 1500 mg/kg and damages to the liver at 500 mg/kg were observed. NOAEL is set to 167 mg/kg bw/day (Gaunt et al. 1968). Threshold limit values No threshold limit values for the working environment was available. The C-value is 1 mg/m3 (Miljøstyrelsen 2002). Absorption No values on absorption of the substance are found and therefore 100% absorption is assumed. Assessment The assessment of oral exposure is based on the exposure for 3 hours of a child of 10 kg body weight (bw). The absorption is set to 100%. Table 15. Uptake of 2-(2-ethoxyethoxy)-ethanol by oral exposure
2-(2-Ethoxyethoxy)-ethanol was detected in extracts from 4 products. Because no limit values for effects from amounts taken up in the body are found NOAEL is used to calculate the margin of safety (MOS). Using a NOAEL of 167 mg/kg bw/day MOS is more than: 167/0.0189 = 8800. Conclusion Based on a high MOS it is assessed that 2-(2-ethoxyethoxy)-ethanol is not considered to pose a health risk to the consumer of the wooden toy. 4.3.8 FormamideIdentification:
The melting point is 2.25°C. The boiling point is 210°C at 1013 hPa (Budavari 1996, Hsdb). The water solubility is high at 25°C (miscible with water, IUCLID 2000). The vapour pressure is 8.1 Pa at 25°C (0.061 mmHg, Daubert and Danner 1989). The octanol/water partition coefficient is measured to log Kow -0.82 (IUCLID 2000). Classification Formamide is adopted on the List of dangerous substances (Miljøministeriet 2002) and classified:
Use The substance is used in the production of several chemical compounds as solvent and in adhesives (HSDB). Effects on health Some data on acute toxicity have been available. Of these are mentioned:
The substance is moderately irritating by contact to the skin, eyes and mucous membranes (Lewis 1996). Formamide is experimentally demonstrated to be teratogenic following oral or percutaneous use (ILO 1983). However, later studies indicate that the observations were caused by very high concentrations, i.e. undiluted test substance or 50% dilutions (ACGIH 1991). The effect of formamide on reproduction is studied in a reproduction toxicity test in mice where formamide was administered orally in drinking water at the doses of 0, 100, 350 and 750 mg/l corresponding to approx. 24 to 195 mg/kg bw/day. Effects on reproduction such as reduced fertility and litter size were observed at 750 mg/l in the parental generation and in the first litter generation. Therefore NOAEL is set to 350 ppm corresponding to 87 mg/kg bw/day (NTP 1992). In a reproduction study on rats, the rats were administered formamide orally via gavage at the doses of 0, 50, 100 or 200 mg/kg bw/day on gestational days 6 to 19 after the mating. Reduced weight of gravid uterine weight at 200 mg/kg bw/day and reduced foetal body weight at 100 mg/kg bw/day and above was observed, i.e. NOAEL was 50 mg/kg bw/day (NTP 1998). The effect of formamide on foetal development is studied on rabbits where formamide was administered orally via gavage at the doses 0, 35, 70 or 140 mg/kg bw/day in the days 6 to 29 of gestation. At the highest dosis significantly reduced weight of uterus, mean litter size and reduced foetal body weight were observed. NOAEL is therefore set to 70 mg/kg bw/day (NTP 2001). Threshold limit values The threshold limit value for the working environment is 10 ppm corresponding to 18 mg/m3 with notation H. H indicates that the substance may penetrate the skin (AT 2002). The C-value is 0.01 mg/m3 (Miljøstyrelsen 2002). Absorption Formamide is absorbed directly through the skin in guinea pigs (Patty 1963). Following oral administration at 2-4 g/rabbit 39% of the dose was recovered unchanged (Snyder 1990). No further information are found and therefore a 100% absorption is assumed. Assessment The assessment of oral exposure is based on the exposure for 3 hours of a child of 10 kg body weight (bw). The absorption is set to 100%. Table 16. Uptake of formamide by oral exposure
Because no limit values for effects from amounts taken up in the body are found NOAEL is used to calculate the margin of safety (MOS). Using a NOAEL of 50 mg/kg bw/day MOS is more than: 50/0.022 = 2300. Conclusion Based on the calculated MOS value that is calculated from the extract containing the highest concentration of the substance, formamide is not considered to pose an immediate health risk to the consumer. 4.3.9 FurfuralIdentification:
The melting point is –36.5°C. The boiling point is 161.7°C. The water solubility is 77000 mg/l at 25°C (Yalkowsky and Dannenfelser 1992). The vapour pressure is 295 Pa at 25°C (2.21 mmHg, Daubert and Danner 1989). The octanol/water partition coefficient is measured to log Kow 0.41 (Hansch et al. 1995). Classification Furfural is adopted on the List of dangerous substances and classified (Miljøministeriet 2002):
It is noted that the classification is dependent of the concentration:
Furthermore, the classification is proposed included R38: Irritating to skin by the 30th ATP (ECB 2005). Use Furfural is used as solvent and in the manufacture of resins and adhesives (CICAD 2000) and dyes (ECB 2004). Effects on health 2-Furfural is acute toxic by oral intake and by exposure via inhalation. A few data have been found on acute toxicity. Of those are mentioned:
Irritation of the respiratory system and eyes are observed at single as well as by repeated exposure. By exposure to the skin the effects seem moderate or irreversible (CICAD 2000). In 13-week studies inhalation studies, NOAEL values of 80 mg/m3 in hamsters and 208 mg/m3 in rabbits for neoplastic effects were found (CICAD 2000). No reports on irritation to respiratory tract and eyes in humans at exposure to 40 mg/m3 (10 ppm) during 8 hours or at 80 mg/m3 during 4 hours were found (CICAD 2000). In a 13 weeks inhalation study, Syrian golden hamsters were exposed to furfural vapours for 6 hours/day, 5 days/week in 13 weeks at the doses 0, 20, 115 and 552 ppm equivalent to 0, 80, 460 and 2208 mg/m3. Based on observations of harmful effects to the olfactory epithelium NOAEL was 20 ppm equivalent to 77 mg/m3 (JECFA 1999, CICAD 2000). A 13 weeks diet toxicity study on rats is performed. The measured exposure doses were 0, 26, 53, 82 and 160 mg/kg bw/day. At 82 mg/kg changes in the liver in 5 out of 10 male rats were observed while there were no changes at 53 mg/kg bw/day (measured concentration). NOAEL was therefore set to 53 mg/kg bw/day (JECFA 2001, ECB 2004). A 90 days rat study from the American National Toxicology Program (NTP) was found. On basis of reduced body weight gain and histopathology, a LOAEL was set to 11 mg/kg bw/day. As this was the lowest concentration used, no NOAEL could be established (NTP 1981 study referenced in IRIS 2004). In a 2-year study on oral administration malignant tumours were observed in rats at 60 mg/kg bw/day and in mice at 50 mg/kg bw/day (CICAD 2000). In CICAD (2000) is mentioned that furfural was genotoxic in vitro in mammalian cells but the genotoxic potential in vivo could not be finally concluded on. The possibility that the genotoxicity may contribute to the carcinogenic process prevents the establishment of a reliable NOAEL (CICAD 2000). At a later evaluation by the Scientific Committee on Cosmetic and Non-Food Products intended for consumers (SCCNFP) was concluded that furfural was not genotoxic in vivo (SCNNP 2004). IARC has evaluated furfural and concluded that there is limited evidence for the carcinogenic effect in experimental animals and inadequate evidence for carcinogenic effect in humans. This means that furfural by IARC (1995) is not classifiable as to its carcinogenicity to humans (Group 3) while furfural by EU is classified Carc.Cat. 3;R40, i.e. Limited evidence of carcinogenic effects. Furfural is found irritating to skin and mucous epithelium and reports on both eczema and allergic skin sensitizing and photo sensitising are found (SCCNFP 2004). Furfural is under the EU program for risk assessment on existing substances but report not finalised yet (the latest draft on human health part from October 2004, ECB 2004). Threshold limit values The threshold limit value for the working environment is 2 ppm equivalent to 7.9 mg/m3 with notation HK. H means that the substance may penetrate skin. K means that the substance is on the list of substances that is considered carcinogenic (AT 2002). The C-value is 0.002 mg/m3 (Miljøstyrelsen 2002). The ADI 0.5 mg/kg bw/day is set on the basis of NOAEL 53 mg/kg in the 13 weeks rat study and applying a safety factor of 100 (JECFA 2001). The RfD-value is set on the basis of the 90-days rat study by NTP. Based on LOAEL 11 mg/kg/day and applying a safety factor of 3000 (10 for inter- and 10 for intraspecies variation, 10 for extrapolating from subchronic to chronic data and a factor of 3 for using a LOAEL) an oral reference dosis (RfD) of 11/3000 = 0.003 mg/kg bw/day was derived (IRIS 2003). Absorption Following oral administration in rats, 14C-2-furfural was readily absorbed and up to 85% recovered in the urine within 24 hours and approx. 7% in the exhaled air. In humans, absorption of the vapour via skin and the lungs and dermal absorption of the substance in liquids has been demonstrated (CICAD 2000). In the evaluation an absorption of 100% is assumed. Assessment The assessment of oral exposure is based on the exposure for 3 hours of a child of 10 kg body weight (bw). The absorption is set to 100%. Table 17. Uptake of furfural by oral exposure
Furfural was detected in the extracts from 4 products. The RfD value of 3 μg/kg/day is not exceeded. Both JECFA (2001) and the EU risk assessment report (ECB 2004) reach the same conclusion that the NOAEL value of 53 mg/kg bw/day is the preferred value for risk characterisation. Using NOAEL 53 mg/kg bw/day the margin of safety (MOS) is more than: 53/0.00145 = 36000. Conclusion Furfural does not exceed the RfD value even though the calculated highest oral uptake of 1.5 μg/kg bw/day is at the same level as the RfD of 3 μg/kg bw/day. However, the MOS is more than 36000 and the substance is concluded not to pose a health problem to the consumer. Considering that the substance is found a possible carcinogenic the question is whether the presence of the substance is undesirable in this context. 4.3.10 4-Hydroxy-3,5-dimethoxy-benzaldehydeIdentification:
The melting point is 113°C. The boiling point is estimated to 305°C. The water solubility is estimated to 9500 mg/l at 25°C. The vapour pressure is estimated to 8.6 mPa at 25°C (6.510-5 mmHg). The octanol/water partition coefficient is estimated to log Kow 0.88 (EPI). Classification 4-Hydroxy-3,5-dimethoxy-benzaldehyde is not classified in the List of dangerous substances (Miljøministeriet 2002) but included in the Danish EPA's Advisory list for self-classification of dangerous substances (Miljøstyrelsen 2001) with the classification:
Use No informations on the use are available. Effects on health No data on acute toxicity or other relevant data on 4-hydroxy-3,5-dimethoxy-benzaldehyde have been found. Threshold limit values No threshold limit values for the working environment are available. Absorption No values on absorption have been found and therefore a 100% absorption is assumed. Assessment The assessment of oral exposure is based on the exposure for 3 hours of a child of 10 kg body weight (bw). The absorption is set to 100%. Table 18. Uptake of 4-hydroxy-3,5-dimethoxy-benzaldehyde by oral exposure
Since no effect values, threshold limit values or similar are found no specific evaluation of the substance could be performed. For a group of benzaldehyde analogous substances a group-ADI has been set at 5 mg/kg bw/day (WHO 1996). However, the evaluation is based on benzaldehyde, CAS no. 100-52-7, where a reproduction toxicity study has been available with the NOAEL of 5 mg/kg bw/day (IUCLID 2000). Besides for benzaldehyde a RfD value of 0.1 mg/kg bw/day is available (IRIS 2004). Because these values are below the WHO group-ADI they are considered a little safer to use. Because no limit values for effects from amounts taken up in the body are found NOAEL is used to calculate the margin of safety (MOS). Using a NOAEL of 5 mg/kg bw/day MOS to the highest estimated uptake is: 5/0.0063 = 790. Conclusion Based on MOS values at and above 790 and that none of the amounts taken up are close to the oral chronic RfD for benzaldehyde, which is evaluated to be more toxic than 4-hydroxy-3,5-dimethoxy-benzaldehyde, 4-hydroxy-3,5-dimethoxy-benzaldehyde is assessed not to pose a health risk to the consumer. 4.3.11 N-Methyl-2-pyrrolidoneIdentification:
The melting point is -24°C. The boiling point is 202°C. The water solubility is high at 25°C (miscible with water). The vapour pressure is 25.3 Pa at 25°C (Riddick et al. 1986). The octanol/water partition coefficient is measured to log Kow –0.46 (IUCLID 2000). Classification N-Methyl-2-pyrrolidone is adopted on the List of dangerous substances and classified (Miljøministeriet 2002):
Use The substance has several uses within the chemical industry among others in paints, dyes and lacquers. The substance is used as solvent and in the production of pigment and printing inks (IUCLID 2004, CICAD 2001). Effects on health N-Methyl-2-pyrrolidone has a low acute toxicity. Some data on acute toxicity have been found. Of those are mentioned:
Uptake of acute toxic doses by oral intake, dermal exposure or exposure via inhalation causes functional disorders and affects the central nervous system. Irritation of the respiratory system by exposure via inhalation and of the gastro-intestinal tract after oral administration has been observed (CICAD 2001). N-Methyl-2-pyrrolidone has a low potential for skin irritation and a moderate potential for eye irritation in rabbits (IUCLID 2000). In a 13 week study with exposure via inhalation rats were exposed to 0, 500, 1000 and 3000 mg/m3 for 6 hours/day, 5days/week. Nasal irritation and crust formations on nasal edges were observed at 1000 mg/m3. At 3000 mg/m3, decreased body weight and decreased relative weight of testis were observed. The NOAEL was then set to 500 mg/m3 (CICAD 2001). Of studies on prolonged duration of oral exposure, a 28-day study where rats were orally administered (via gavage) the doses 0, 257, 514, 1028 and 2060 mg/kg bw/day was found. A dose-dependent increase in relative liver and kidney weights was observed at 1028 mg/kg bw/day. Thus, the NOAEL was 514 mg/kg bw (CICAD 2001). In a 90-day study, rats were administered the substance in the diet at the doses 0, 3000, 7500, and 18 000 mg/kg diet/day corresponding to 0, 169, 433 and 1057 mg/kg bw/day in males and 0, 217, 565 and 1344 mg/kg bw/day in females. A dose-related decrease in body weight and effects on the central nervous system was observed at 433 and 565 mg/kg bw/day in males and females, respectively. The NOAEL was 169 mg/kg bw/day in males and 217 mg/kg bw/day in females (CICAD 2001). Threshold limit values The threshold limit value for the working environment is 5 ppm corresponding to 20 mg/m3 (AT 2002). The C-value is 0.5 mg/m3 (Miljøstyrelsen 2002). The TDI value is 0.6 mg/kg bw/day. The value is derived from the 90-day oral study with a NOAEL of 169 mg/kg bw/day divided with a safety factor of 300 (10 for interspecies and 10 for intraspecies variation and 3 for adjusting from a 90 day subchronic to chronic) (CICAD 2001). Absorption N-Methyl-2-pyrrolidone is absorbed easily after inhalation, oral or dermal exposure and is distributed to the whole body. Approximately 80% of the administered dosis are excreted via the urine within 24 hours (Åkesson and Paulsson 1997). Therefore, an absorption of 100% is assumed. Assessment The assessment of oral exposure is based on the exposure for 3 hours of a child of 10 kg body weight (bw). The absorption is set to 100%. Table 19. Uptake of N-methyl-2-pyrrolidone by oral exposure
N-Methyl-2-pyrrolidone was detected in extracts from 5 products. The TDI value of 600 μg/kg bw/day is not exceeded. The highest uptake is a factor of 30 below the TDI value. Using the 90-day diet toxicity NOAEL of 169 mg/kg bw/day the margin of safety (MOS) is at least: 169/0.019 = 8900. Conclusion N-Methyl-2-pyrrolidone is assessed not to pose a health risk to the consumer of the toy. 4.3.12 3,5,5-Trimethyl-2-cyclohexen-1-one (= isophorone)Identification:
The melting point is -8,1°C. The boiling point is 215°C. The water solubility is 12000 mg/l at 20°C (IUCLID 2000). The vapour pressure is 58 Pa at 25°C (0.438 mmHg, Daubert and Danner 1989). The octanol/water partition coefficient is measured to log Kow 1.67 (IUCLID 2000). Classification 3,5,5-Trimethyl-2-cyclohexen-1-one is adopted on the List of dangerous substances and classified (Miljøministeriet 2002):
Use 3,5,5-Trimethyl-2-cyclohexen-1-one is used as solvent in synthetic resins, polymers, polyacrylates and cellulose derivatives and has some use in certain paints and printing inks (IPCS 1995). Effects on health 3,5,5-Trimethyl-2-cyclohexen-1-one has a low to moderate acute toxicity. Some data on acute toxicity have been available. Of these are mentioned:
3,5,5-Trimethyl-2-cyclohexen-1-one is evaluated to be irritating to eyes, nose and throat but not to skin (IPCS 1995, OECD 2003). Several studies on prolonged exposure exist. In a 91 days oral repeated dose toxicity study, rats were administered 3,5,5-trimethyl-2-cyclohexen-1-one via gavage at the doses 0, 62.5, 125, 250, 500 and 1000 mg/kg bw/day for 5 days/week. Effects were only observed as one mortality at the highest dose. NOAEL is therefore set at 500 mg/kg bw/day (IUCLID 2000). In a 91 days oral repeated dose toxicity study, mice were administered 3,5,5-trimethyl-2-cyclohexen-1-one via gavage at the doses 0, 62.5, 125, 250, 500 and 1000 mg/kg bw/day for 5 days/week. Effects were only observed as mortality at the highest dose and dosis related decrease in body weight in males at 250 mg/kg bw/day and above. NOAEL is set to 125 mg/kg bw/day (IUCLID 2000). In a 90 days oral study on beagle dogs, which daily for 7 days/week were administered the substance in gelatine capsules at the doses 0, 35, 75 and 150 mg/kg bw/day, no essential substance related effects at any of the used concentrations were observed. NOAEL is set to 150 mg/kg bw/day (IUCLID 2000). In a 2-year oral carcinogenicity study on mice and rats were administered the substance via gavage 5 days/week at the doses 250 and 500 mg/kg bw/day. Some evidence of carcinogenic effects (tumours) was observed by pathological examinations of the kidneys in males but not in females at the lowest dose (NTP 1984). Threshold limit values The threshold limit value for the working environment is 5 ppm corresponding to 25 mg/m3 with notation LK. L means that the threshold limit value is a ceiling value that must not be exceeded at any time. K means that the substance is included on the list of substances that is considered carcinogenic (AT 2002). The C-value is 0.03 mg/m3 (Miljøstyrelsen 2002). The oral RfD value is 0.2 mg/kg bw/day. The value is based on the 90-day study on dogs with NOAEL 150 mg/kg bw/day and the 2-year carcinogenicity studies with a recalculation of the 250 mg/kg bw administered 5 days/week to 7 days/week (250 x 5/7 =) 179 mg/kg bw/day. Using a safety factor of 1000 (10 for inter-, 10 for intraspecies variation and 10 for subchronic to chronic (dog) or 10 for LOAEL to NOEL in the rat study) is derived an oral RfD at 0.2 mg/kg bw/day (IRIS 2004). Absorption By oral administration of radioactive labelled 3,5,5-trimethyl-2-cyclohexen-1-one, 93% was recovered in the urine and exhaled air within 24 hours. The uptake of the substance through the skin is fast indicating a readily dermal absorption (IPCS 1995). Based on this information a 100% absorption is assumed. Assessment The assessment of oral exposure is based on the exposure for 3 hours of a child of 10 kg body weight (bw). The absorption is set to 100%. Table 20. Uptake of 3,5,5-trimethyl-2-cyclohexen-1-one by oral exposure
3,5,5-Trimethyl-2-cyclohexen-1-one does not exceed the RfD value of 200 μg/kg bw/day. The highest calculated oral uptake of 6.6 μg/kg bw/day is a factor 30 below the RfD value. Using the 91-day mice diet toxicity NOAEL of 125 mg/kg bw/day the margin of safety (MOS) is at least: 125/0.0066 = 19000. Conclusion 3,5,5-Trimethyl-2-cyclohexen-1-one is assessed based on the RfD value and the MOS not to pose a health risk to the consumer of the toys. 4.3.13 VanillinIdentification
The melting point is 81.5°C. The boiling point is 284°C (Kirk-Othmer 1991). The vapour pressure is 0.016 Pa at 25°C (0.00012 mmHg, Yaws 1994) or 0.33 Pa (OECD 1996). The water solubility is 11000 mg/l at 25°C (Yalkowsky and Dannenfelser 1992). The partition coefficient log Kow is measured to 1.23 (OECD 1996). Classification Vanillin is not classified (Miljøministeriet 2002). Use Vanillin is present as a natural compound in plants and is identified in plant oils, balsams, resins and wood. The best known natural source is the plant Vanilla planifolia in the Orchid family (Kirk-Othmer 1991, Ullmann 1993). Vanillin is also manufactured synthetically from guaiacol but mainly from lignin, which is the main component in waste from sulphite pulp in the paper industry (Hocking 1997). Vanillin is used in food ("vanilla"), pharmaceuticals and in the cosmetics and perfume industry. Effects on health Vanillin is a phenol aldehyde with reactive aldehyde and hydroxyl moieties in the molecule. Acute toxicity:
Acute toxicity was not observed after inhalation of vapours from saturated solutions. However, irritation was observed after application to the skin and mucous membranes (Kirk-Othmer 1991). A 30 days inhalation test with exposure for 4 hours/day, 5 days a week to saturated vapours at 20°C showed no mortality but reduction in body weight and effects to liver and to the haemoglobin level (ECB 2000). A 4 months inhalation study showed that 15 mg/m3 (LOAEL) affected nerves and cardiovascular systems, liver and blood systems. Therefore, NOAEL is set to 0.5 mg/m3 (ECB 2000). Several diet toxicity tests are performed and the summarised in IUCLID (ECB 2000) and SIDS (OECD 1996). The most essential are presented below: In a 14 weeks study with oral administration via gavage of 300 mg vanillin/kg to rats, twice weekly, no adverse effects were observed. Rats fed diets containing vanillin at levels of 20 mg/kg/day for 18 weeks had no adverse effects while 64 mg/kg/day for 10 weeks caused growth depression and damage to the myocardium, liver, kidney, lung, spleen and stomach. Rats fed diets containing vanillin for 13 weeks exhibited growth depression and enlargement of the liver, kidney and spleen at dosage levels 5% (2500 mg/kg/day*), mild changes at 1% (500 mg/kg/day*), and no changes at 0.3% (150 mg/kg/day*). Four to six week old rats maintained for 91 days on diets containing vanillin exhibited no adverse effects at rates of 3000 ppm (150 mg/kg/day*), mild adverse effects at 10,000 ppm (500 mg/kg/day*) and growth depression and enlargement of the liver, kidney and spleen at 50,000 ppm (2500 mg/kg/day*). Rats fed dietary levels of vanillin of 10,000 ppm (500 mg/kg/day*) for 16 weeks, 1000 ppm (50 mg/kg/day*) for 27-28 weeks, 20,000 or 50,000 ppm (1000 or 2500 mg/kg/day*) for 1 year, or 5000, 10,000, or 20,000 ppm (250, 500, or 1000 mg/kg/day*) for 2 years exhibited no adverse effects on growth or haematology and produced no macroscopic or microscopic changes in tissues (Hagan et al. 1967). Rats fed for 5 weeks on a diet of vanillin at 0.5 g/kg of the diet showed symptoms of intoxication, including decreases in adrenal vitamin C and in liver protein (Kirwin and Galvin 1993). *Based on a food factor of 0.05 for a 0.35 kg rat (US-EPA 1985). The highest NOEL from the 1-year repeated dose toxicity rat study with oral administration was 50000 ppm corresponding to 2500 mg/kg/day. NOEL in the 2 years rat study was 1000 mg/kg bw/day. The latter value from Hagan et al. (1967) is used by WHO to derive an ADI-value. Teratogenicity, genotoxicity and carcinogenicity tests are performed that were all negative, i.e. there was no indications of such effects (OECD 1996). Threshold limit values A threshold limit value for the working environment is not available (AT 2002). An ADI (Acceptable Daily Intake) of 10 mg/kg has been agreed between FAO/WHO and EU (OECD 1996) based on NOEL 1000 mg/kg bw/day from a 2-year rat study (Hagan et al. 1967). JECFA has placed vanillin in structure class I together with other terpenoid substances, i.e. vanillin is applied a group-ADI of 0.05 mg/kg/day. Absorption No data are available on adsorption. Therefore 100% adsorption is used Assessment The assessment of oral exposure is based on the exposure for 3 hours of a child of 10 kg body weight (bw). The absorption is set to 100%. Table 21. Uptake of vanillin by oral exposure
From the table above is seen that none of the amounts taken up by oral exposure results in a dosis above the ADI value of 50 μg/kg bw/day. The WHO recommended ADI of 10 mg/kg bw/day is not exceeded either. Using the NOAEL 1000 mg/kg bw/day from the 2-year rat study the margin of safety is more than: 1000/0.0047 = 200000. Conclusion Vanillin does not pose any health risk to the consumer by oral exposure to the migrated amounts determined by analyses of the selected toy products. 4.3.14 BariumIdentification:
The melting point is 710°C. The boiling point is 1600°C (Budavari 1996). The vapour pressure is assumed to be very low. The water solubility also assumed to be very low. Classification Barium and barium compounds are not classified. Use Some barium compounds among others barium acetate is used in colorants (Budavari 1996). Barium compounds are used in several industries but also in cosmetics, pharmaceuticals and in paints and dyes (CICAD 2001). Effects on health The toxic effects are based on the assumption that the barium-ion is the active substance since the toxicity studies usually are performed with water soluble barium salts and the results then recalculated to barium equivalents. Acute oral LD50 values for rats administered barium chloride, - carbonate, and –sulphide are between 118 and 800 mg/kg bw (IPCS 1990). In a 13 weeks oral rat study, the rats were administered barium chloride in drinking water at daily doses of 0, 125, 500, 1000, 2000 and 4000 mg/l corresponding to 0, 10, 30, 65, 110, and 180 mg/kg/bw. Based on kidney effects NOAEL was set to 65 mg/kg bw/day in a NTP study from 1994 (CICAD 2001). In a chronic oral rat study, rats were dosed via the drinking water for 2 years with 0, 500, 1250 and 2500 mg barium chloride/l corresponding to 0, 15, 45 and 75 mg/kg bw for females and 0, 15, 30 and 60 mg/kg for males. Based on a relative increase of the kidney weight NOAEL was set to 45 mg/kg bw/day (CICAD 2001). The critical effect in humans appears to be increased blood pressure (hypertension) and harmful effects to the kidneys (CICAD 2001). In an oral study on humans, barium chloride was administered in drinking water at gradually increased doses for 4-week periods while the blood picture was examined continuously. NOAEL was set to 10 mg Ba/l/day when the test volunteers were given1.5 l/day. Assuming a mean body weight of 70 kg this corresponds to a NOAEL of 0.21 mg/kg bw/day (Wones et al. 1990). In an epidemiological study of population groups in USA, a NOAEL of 7.3 mg/Ba/l/day was calculated. Assuming an intake of 2 l/day and 70 kg body weight 7.3 mg Ba/l corresponds to 0.21 mg/kg bw/day (Brenniman and Levy 1984). Threshold limit values The threshold limit value for working environment is 0.5 mg/m3 (AT 2002). The C-value is preliminary set to 0.005 mg/m3 (Miljøstyrelsen 2002). RIVM has derived an acceptable daily breathing air concentration (HAC) to 0.001 mg/m3 (Baars et al. 2001). US-EPA has suggested an acceptable daily oral intake dosis (RfD) to 0.070 mg/kg bw/day based on several studies but as principal studies the studies by Wones et al. (1990), Brenniman and Levy (1984), and NTP (1994). Applying a safety factor of 3 they derived an RfD value of (0.21/3 = 0.07 mg/kg bw/day (IRIS 2002). WHO has suggested a TDI of 0.02 mg/kg bw/day based on Wones et al. (1990) applied a safety factor of 10 for insufficient data and a potential difference between children and adults, i.e. (0.21/10 = 0.02 mg/kg bw/day (WHO 1996, CICAD 2001). RIVM has calculated a tolerable daily intake TDI 0.020 mg/kg bw/day based on the same considerations (Baars et al. 2001). According to the Statutory Order on toys the bioavailability of barium as a consequence of the use of the toy must not exceed 25 μg/day (Bkg. 1116, 2003). Absorption The absorption following oral intake is strongly influenced by e.g. food in the gastro-intestinal tract that may bind barium, the duration of the study period and animal species. Absorption values varying from 0.7 to 85% have been found (CICAD 2001). In a study on humans it was observed that the absorption was approx. 10% with 90 to 98% excreted via faeces and 2 to 10% via the urine (Cember et al. 1961, Tipton et al. 1969). In a Dutch evaluation is estimated that the bioavailability by inhalation is 75%. The bioavailability by oral intake is estimated to 10% (Baars et al. 2001). Assessment The assessment of oral exposure is based on the exposure for 3 hours of a child of 10 kg body weight (bw). The absorption is set to 100% and 10%. Table 22. Uptake of barium by oral exposure
The determined concentrations for oral uptake vary between 9 and 28 μg/kg lgv/day when 100% absorption is assumed. Using the Dutch bioavailability value, which states the absorption to be 10%, the calculated uptakes are the values mentioned in right column of the table above. This reduces all calculated values to below the TDI value of 20 μg/kg bw/day. Using the human oral NOAEL of 10 mg/kg bw/day the margin of safety (MOS) is more than: 10/0.0284 = 350 with 100% absorption and 3500 with 10% absorption. According to the Statutory Order on toys the bioavailability of barium must not exceed 25 μg/day. The value 25 μg/day is based on an assumed oral intake of 8 mg of the toy, i.e. the maximum bioavailable concentration of barium in the toy must be max. 3125 mg Ba/kg toy (CSTEE 2004). In the standard EN 71-3 is given the limit value 1000 mg Ba/kg toy (EN 71-3). Assuming a bioavailability of 10% the measured migration of 29-90 μg/g (= mg/kg) thus far below 1000 mg Ba/kg toy. On the other hand it must be noted that the major source to metal exposure to children is via the food. Other sources to exposure are not included in the assessment. If for instance the limit of the contribution from toys is set to 10% of the TDI value of 20 μg/kg bw/day then at least 3 products exceed the value. Conclusion Based on MOS and assuming the 10% absorption is the most realistic it is assessed that barium does not pose a potential health risk to the consumer of wooden toy. 5 ConclusionBelow is summarised the results of the assessment of the individual substances and the detected concentrations. Table 22 Summary of conclusions
From the detected substances and based on classification and occurrence a number of substances were selected for a further assessment of a potential health risk to the consumers. The consumers are in this context defined as children 0 to 3 years of age. In spite of that none of the detected substances could be assessed to pose any immediate health risks to the consumer it should be noted that the classification of some of the identified substances was of a serious character. Of the 125 identified substances 43 were classified in the List of dangerous substances (Annex I to Directive 67/548/EEC) and further 16 could be self-classified according the Advisory list for self-classification of dangerous substances from the Danish Environmental Protection Agency. Of the classified substances 2 were classified carcinogenic (Carc. cat. 3), 1 substance mutagenic (Mut. cat. 3), and 9 substances reproduction toxic (Repr. cat. 2-3). Besides, 5 substances were classified sensitising. The assessments are mostly evaluated by comparison to data from subchronic or even chronic effect data. As the urge to put toys in the mouth is assumed to decline after a few years, the conclusions should be acceptable. The conclusion from this study is, therefore, that none of the examined pieces of wooden toys are considered to pose a health risk to children mouthing the toys when the evaluation is based on individual substances. However, during the use of the toy the child is exposed to several substances released simultaneously from the toy. The effects from the exposure to several chemicals at the same time are unknown. This means that a potential effect can not be entirely be refused. Another problem by evaluating individual substances in single toys may arise when the child plays with more than one toy. This will increase the exposure accordingly. Besides, it should be noted that by an assessment by comparing to threshold limit values, TDI, etc. that the consumer may also be exposed to the same substances from other sources not included in the evaluation. 6 ReferencesACGIH (1991): Documentation of the Threshold Limit Values and Biological Exposure Indices. 6th ed. American Conference of Governmental Industrial Hygienists, Cincinnati, Ohio, USA. AT (2002): Grænseværdier for stoffer og materialer. At-vejledning. C.0.1, Oktober 2002. Arbejdstilsynet, København. Barbee SJ, Terrill JB, DeSousa DJ, Conaway CC (1984): Subchronic inhalation toxicology of ethylene glycol monoethyl ether in the rat and rabbit. Environ. Health Perspect. 57: 157-163. Baars AJ, Theelen RMC, Janssen PJCM, Hesse JM, van Apeldoorn ME, Meijerink MCM, Verdam L, Zeilmaker MJ (2001): Re-evaluation of human-toxicological maximum permissible risk levels. RIVM report 711701025. National Institute of Public Health and the Environment, Bilthoven, The Netherlands. Bkg. 1116 (2003): Bekendtgørelse om sikkerhedskrav til legetøj og produkter, som på grund af deres ydre fremtræden kan forveksles med levnedsmidler. Bekendtgørelse nr. 1116 af 12/12/2003. Sikkerhedsstyrelsen. Boatman RJ (1993): Toxicology of diethylene glycol butyl ether. 2.Disposition studies with 14C- diethylene glycol butyl ether acetate after dermal application to rats. J. Am. Coll. Toxicol. 12: 145-154. Bremmer HJ, van Veen MP (2002): Children's toys fact sheet. To assess the risks for the consumer. RIVM report 612810012/2002. National Institute of Public Health and the Environment, Bilthoven, The Netherlands. Brenniman, GR; Levy, PS. (1984) Epidemiological study of barium in Illinois drinking water supplies. In: Advances in modern toxicology, Calabrese, EJ, ed. Princeton, NJ: Princeton Scientific Publications, pp. 231-249. Budavari S, ed. (1996): The Merck Index – An Encyclopedia of Chemicals, drugs and Biologicals. Merck and Co., Inc. Whitehouse Station, NJ. Cember H, Watson J, Novak M (1996) Bronchiogenic carcinoma from radioactive barium sulfate. Am. Indust. Hyg. Assoc. J. 22: 27-32. CEN/TC 52 (2003): Final report of the work of CEN/TC 52/WG 9, Risk assessment of organic chemicals in toys". CEN/TC 52 Safety of Toys, Doc. CEN/TC 52 N851 Revision 2, January 2003. CICAD (1998): 2-Butoxyethanol. Concise International Chemical Assessment Document No. 10. International Programme on Chemical Safety. WHO, Geneva. CICAD (2000): 2-Furfural. Concise International Chemical Assessment Document no. 21. International Programme on Chemical Safety. World Health Organization. Geneva, Switzerland. CICAD (2001): Barium and barium compounds. Concise International Chemical Assessment Document no. 33. World Health Organization, Geneva. CICAD (2001): N-Methyl-2-pyrrolidone. Concise International Chemical Assessment Document no. 35. World Health Organization, Geneva. Corley RA, Bormett GA, Ghanayem BI (1994): Physiologically-based pharmacokinetics of 2-butoxyethanol and its major metabolite, butoxyacetic acid, in rats and humans. Toxicol. Appl. Pharmacol. 129: 61-79. CSTEE (1998): Phthalate migration from soft PVC toys and child-care articles. EU Scientific Committee on Toxicity, Ecotoxicity and the Environment (CSTEE). Opinion expressed at the CSTEE third plenary meeting. Brussels, 24 April 1998. CSTEE (2003): Opinion of the Scientific Committee on Toxicity, Ecotoxicity and the Environment (CSTEE) on "Assessment of the European Committee for Standardisation (CEN) report on the risk assessment of organic chemicals in toys". Report version: "Final report of the work of CEN/TC 52/WG 9, January 2003". Adopted by the CSTEE 12 November 2003. European Commission, Brussels, C7/VR/csteeop/CEN/12-13112003 D(03) CSTEE (2004): Opinion of the Scientific Committee on Toxicity, Ecotoxicity and the Environment (CSTEE) on "Assessment of the bioavailability of certain elements in toys". Doc.: C7/GF/csteeop/toysinorg/220604 D(04). Adopted by the CSTEE by written procedure on 22nd June 2004. Brussels Daubert TE, Danner RP (1989): Physical and Thermodynamic Properties of Pure Chemicals. Data Compilation. Washington, D.C.: Taylor and Francis. Dodd DE, Snelling WM, Maronpot RR (1983) Ethylene glycol monobutyl ether. Acute, 9-day and 90-day vapor inhalation studies in Fisher 344 rats. Toxicol. Appl. Pharmacol. 68: 405-414. DOW (1990): The Glycol Ethers Handbook. The Dow Chemical Company, Midland, MI. DTI (2002): Research into the mouthing behaviour of children up to 5 years old. Consumer and Competition Policy Directorate. Department of Trade and Industry, London, United Kingdom. (www.dti.gov.uk). EC (2003): Technical Guidance Document on Risk Assessment in support of Commission Directive 93/67/EEC on Risk assessment for new notified substances, Commission Regulation (EC) 1488/94 on Risk assessment for existing substances and Directive 98/8/EC of the European Parliament and of the Council concerning the placing of biocidal products on the market. Parts 1-4. European Commission (EC), Joint Research Centre, EUR 20418 EN, Office for Official Publications of the EC, Luxembourg. EC (2004): Commission Directive 2004/73/EC of 29 April 2004 adapting to technical progress for the 29th time Council Directive 67/548/EEC on the approximation of the laws, regulations and administrative provisions relating to the classification, packaging and labelling of dangerous substances. Official Journal of the European Union, 16.6.2004, L 216; 3-310. ECB (2000): Vanillin, CAS 121-33-5. IUCLID Dataset. European Commission, European Chemicals Bureau, 18 February 2000. ECB (2000): European Risk Assessment Report on 2-(2-butoxyethoxy)ethanol, CAS no.: 112-34-5, EINECS no.: 203-961-6. RAR Vol. 2. European Commission, Joint Research Centre, European Chemicals Bureau. Ispra, Italy. ECB (2004): Risk Assessment Report on 2-Butoxyethanol, CAS No.: 111-76-2, EINECS No: 203-905-0. Draft of August 2004. Prepared by France for the European Commission – Joint Research Centre. Institute for Health and Consumer Protection. European Chemicals Bureau (ECB). ECB (2004): Risk Assessment Report on Furfural, CAS No.: 98-01-1, EINECS No: 202-627-7. Draft of October 2004. Prepared by the Netherlands for the European Commission – Joint Research Centre. Institute for Health and Consumer Protection. European Chemicals Bureau (ECB). ECB (2005): Recommendations of the Technical Committee for Classification and Labelling and Specialised Experts for possible inclusion into the 30th ATP. European Chemicals Bureau (ECB). EN71-1: Safety of toys - Part 1: Mechanical and physical properties. CEN Standard. European Committee for Standardization (CEN). EN71-2: Safety of toys-Part 2: Flammability. CEN Standard. European Committee for Standardization (CEN). EN71-3: Safety of toys-Part 3: Migration of certain elements. elements (DS/EN 71-3/A1 + AC: 2002. Legetøj - Sikkerhedskrav - Del 3: Migration af særlige stoffer. Dansk Standard, 2003-01-08. EN 71-9: Safety of Toys – Organic chemical compounds – Requirements. CEN Standard. European Committee for Standardization (CEN) EN 71-10: Safety of Toys – Organic chemical compounds – Sample preparation and extraction. CEN Standard. European Committee for Standardization (CEN) EN 71-11: Safety of Toys – Organic chemical compounds – Methods of analysis. CEN Standard. European Committee for Standardization (CEN) Erhvervsfremmestyrelsen (2000): Børns leg og læring - En kompetenceklynge i dansk erhvervsliv. Ferdinand J, Kaysen O, Petersen C (2003): Undersøgelse af naturlegetøj af plantefibre, uldfibre og massivt træ. Kortlægning af kemiske stoffer o forbrugerprodukter nr. 33. Miljøstyrelsen, København. Funasaki N, Hada S, Neya S, Machida K (1984): Intramolecular hydrophobic association of two alkyl chains of oligoethylene glycol diethers and diesters in water. J. Phys. Chem. 88: 5786-5790. Gaunt IF, Carpanin FMB, Grasso P(1972) Long-term toxicity of propylene glycol in rats. Food Cosmet Toxicol 10(2):151-162. Grant WM (1986): Toxicology of the eye. 3rd ed. Charles C Thomas Publ. Springfield, IL, USA. Greene MA (2002): Mouthing times for children from the observational study. Division of Hazard Analysis, US Consumer Product Safety Commission, Washington DC (www.cpsc.gov). 14 p. Groot ME, Lekkerkerk MC, Steenbekkers LPA (1998): Mouthing behaviour of young children - An observational study. Wageningen: Agricultural University, Household and Consumer Studies, Wageningen, Netherlands. Gupta G, Mishra A Agarwal D (1991): Inhalation toxicology of furfural vapours: an assessment of biochemical response in rat lungs. J. Appl. Toxicol. 11: 343-347. Hagan EC, Hansen WH, Fitzhugh OG, Jenner PM, Jones WI, Taylor JM, Long EL, Nelson AA, Brouwer JB (1967): Food flavouring and compounds of related structure. II. Subacute and chronic toxicity. Food Cosmet. Toxicol. 5; 141-157. Hansch C, Leo A, Hoekman D (1995): Exploring QSAR – Hydrophobic, Electronic and Steric Constants. American Chemical Society, Washington DC. Hocking MB (1997): Vanillin: Synthetic flavoring from spent sulfite liquor. J. Chem. Educ. 74: 1055. HSDB (2004): Hazardous Substances Databank, a database of the National Library of Medicine’s TOXNET system (http://toxnet.nlm.nih.gov). IARC (1995): Furfural. International Agency for Research on Cancer (IARC) Vol. 63: 409. ILO (1983): Encyclopedia of Occupational Health and Safety. International Labour Office. Geneva, Switzerland. IPCS (1990): Barium. Environmental Health Criteria no. 107. International Programme on Chemical Safety. World Health Organization, Geneva, Switzerland. IPCS (1990): 2-Methoxyethanol, 2-Ethoxyethanol, and their acetates. Environmental Health Criteria no. 115. International Programme on Chemical Safety. World Health Organization, Geneva, Switzerland. IPCS (1994): Hydroquinone. Environmental Health Criteria no. 157. International Programme on Chemical Safety. World Health Organization, Geneva, Switzerland. IPCS (1995): Isophorone. Environmental Health Criteria no. 174. International Programme on Chemical Safety. World Health Organization, Geneva, Switzerland. IRIS (2004): Integrated Risk Information System. US Environmental Protection Agency (www.epa.gov/iris). IUCLID (2000): International Uniform Chemical Information Database. A database on `Existing Chemicals' compiled by the European Chemicals Bureau (ECB), Ispra, Italy. JECFA (1999): Furfural. Safety evaluation of certain food additives and contaminants. WHO Food Additives Series 42. Prepared by the fifty first meeting of the Joint FAO/WHO Expert Committee on Food Additives (JEFCA). International Programme on Chemical Safety, World Health Organization, Geneva. JECFA (2001): Furfural (addendum). WHO Food Additives Series 46. Prepared by the Joint FAO/WHO Expert Committee on Food Additives (JEFCA). International Programme on Chemical Safety, World Health Organization, Geneva. Juberg DR, Alfano K, Coughlin RJ, Thompson KM (2001): An observational study of object mouthing by young children. Pediatrics 107(1): 135-142. Kirk-Othmer (1991): Encyclopedia of Chemical Technology. 4th ed. J.Wiley & Sons New York. Kirwin CJ, Galvin JB (1993): Ethers. In: Clayton GD, Clayton FE, Eds. 1993. Patty's Industrial Hygiene and Toxicology, Vol.2, Part A, 4th ed. New York: John Wiley & Sons, pp. 445-525. Kiss CT (2001): A mouthing observation study of children under 6 years of age. Division of Human Factors, US Consumer Product Safety Commission, Washington DC (www.cpsc.gov). 14 p. Lamb JC, Gulati DK, Russle VS, Hommel L, Sabharwal PS (1984): Reproductive toxicity of ethylene glycol monoethyl ester tested by continuous breeding of CD-1 mice. Environ. Health Perspect. 57: 85-90. Lewis RJ (1996): Sax's Dangerous Properties of Industrial Materials. 9th ed. Van Nostrand Reinhold, New York, NY. Lijinsky W, Kovatch M (1986): A chronic toxicity study of cyclohexanone in rats and mice (NCI study). J. Natl. Cancer Inst. 77(4): 941-949. Lovell CR (1993): Plants and the skin. Blackwell Scientific Publications. Oxford. MacAulay JC (1997): Orchid allergy. Contact Derm. 1997; 112-113 Miljøministeriet (2002): Bekendtgørelse af listen over farlige stoffer. Bekendtgørelse no. 439 af 3. juni 2002. Miljøministeriet. Danmark. ( In Danish: Statutory Order on the List of dangerous substances, implementation of Directive 67/548/EEC, 28th amendment). Miljøstyrelsen (2001): Report on the Advisory list for selfclassification of dangerous substances. Environmental Project no, 636. Can be obtained together with search function and excell file on the Danish EPA homepage, chemicals, www.mst.dk/chemi/01050000.htm Miljøstyrelsen (2002): B-værdivejledningen. Oversigt over B-værdier. Vejledning nr. 2, 2002. Miljøstyrelsen, København. Miljøstyrelsen (2004): List of undesirable substances 2004. Environmental review no. 15 2004. Danish Environmental Protection Agency. Nelson KW et al. (1943): Journal of Industrial Hygiene and Toxicology, 25, 282-285. I OECD/SIDS. Screening Information Data Set (SIDS) of OECD High Production Volume Chemicals Programme, Vol. 3 part II: 1-163, 1994. NTP (1984): NTP Technical Report on the Toxicology and Carcinogenisis Studies of Isophorone in F344/N Rats and B63F1 Mice (Gavage). NIH Publication No. 84-2547, NTP-83-168. Department of Health and Human Services, National Institute of Environmental Health Sciences, National Toxicology Program (http://ntp-server.niehs.nih.gov). NTP (1992): Reproductive toxicity of formamide, CAS no. 75-12-7) in CD-1 Swiss Mice. NTP study no. RACB90005. Department of Health and Human Services, National Institute of Environmental Health Sciences, National Toxicology Program (http://ntp-server.niehs.nih.gov). NTP (1993): Technical report on toxicity studies of ethylene glycol ethers 2-methoxyethanol, 2-ethoxyethanol, 2-butoxyethanol administered in drinking water to F344/N rats and B6C3F1 mice. National Toxicology Program Technical Report No. 26. US Department of Health and Human Services, National Institutes of Health, Research Triangle Park, NC. NTP (1994): Technical report on the toxicology and carcinogenesis studies of barium chloride dihydrate (CAS No. 10326-27-9) in F344/N rats and B6C3F1 mice (Drinking Water Studies). NTP TR 432. National Toxicological Program, Research Triangle Park, NC. NIH Pub. No. 94-3163. NTIS Pub PB94-214178. NTP (1998): Developmental toxicity evaluation of formamide, CAS no. 75-12-7) administered by gavage to Sprague-Dawley rats on gestational days 6 through 19. NTP study no. TER97002. Department of Health and Human Services, National Institute of Environmental Health Sciences, National Toxicology Program (http://ntp-server.niehs.nih.gov). NTP (2001): Developmental toxicity evaluation of formamide, CAS no. 75-12-7) administered by gavage to New Zealand white rabbits on gestational days 6 through 29. NTP study no. TER98002. Department of Health and Human Services, National Institute of Environmental Health Sciences, National Toxicology program (http://ntp-server.niehs.nih.gov). OECD (1993): Occupational and consumer exposure assessment. Environment Monograph no. 70. Organisation for Economic Co-operation and Development. Paris. OECD (1994): Cyclohexanone. OECD/SIDS. Screening Information Data Set (SIDS) of OECD High Production Volume Chemicals Programme, Vol. 3 part 2: 1-163. OECD (1996): Vanillin. SIDS Initial Assessment Profile. OECD Screening Information Data Sets (SIDS) Vol. 5, part 2. Organisation of Economic and Co-operation and Development. OECD (2003): 3,5,5-Trimethylcyclohex-2-enon. SIDS Initial Assessment Report. OECD Screening Information Data Sets (SIDS) Draft 11.09.03. Patty F, ed. (1963): Toxicology. Industrial Hygiene and Toxicology. Vol 2, 2nd ed. Interscience Publishers. new York. Rasmussen JE (1986): Contact dermatitis from orchids. Clin. Dermatol. 4: 31-35. Riddick JA, Bunger WB, Sakano TK (1985): Organic Solvents. Techniques of Chemistry. 4th ed. Vol 2. John Wiley and Sons, New York, NY. Snyder R, ed. (1990): Ethyl Browning's Toxicity and Metabolism of Industrial Solvents. 2.ed. Elsevier Publ. Amsterdam-New-York-Oxford. Tipton IH, Stewart PL, Dickson J (1969): Patterns of elemental excretion in long term balance studies. Health Physics 16: 455-462. Tyl TR, Millicovsky G, Dodd DE, Pritts IM, France KA, Fisher LC (1984): Teratologic evaluation of ethylene glycol monobutyl ether in Fisher 344 rats and New Zealand white rabbits following inhalation exposure. Env. Health. Perspectives 57:47-68 Ullmann (1993): Ullmann's Encyclopedia of Industrial Chemistry. 5th Ed. VCH Verlagsgesellschaft. US-EPA (1985): Reference values for risk assessment. Environment Criteria Assessment Office. US Environmental Protection Agency, Cincinatti, OH. US-EPA (1997): Exposure factors handbook. Update to exposure factors handbook 1989. EPA/600/P-95/002Fa. U.S. Environmental Protection Agency, Office of Research and Development, National Center for Environmental Assessment. Washington DC. US-EPA (2002): Child-Specific Exposure Factors Handbook. National Center for Environmental Assessment. EPA/600/P-00/002B. US Environmental Protection Agency, Washington DC. WHO (1996): Guidelines for drinking water quality. 2nd ed. Vol 2. health criteria and other supporting information. World Health Organization, Geneva. WHO (1996): Benzyl derivatives. Safety evaluation of certain food additives and contaminants, WHO Food Additives Series no. 48. World Health Organization, Geneva. Wones, RG; Stadler, BL; Frohman, LA. (1990) Lack of effect of drinking water barium on cardiovascular risk factor. Environ. Health Perspect. 85: 355-359. Yalkowsky SH, Dannenfelser RM (1992): The Aquasol Database of Aqueous Solubility. Fifth Ed, Tucson, AZ: Univ Az, College of Pharmacy. Yaws CL (1994): Handbook of Vapour Pressure. Vol. 3: C8-C28 Compounds. Houston, Texas, Gulf Publ. Co. Åkesson og Paulsson (1994): N-Methyl-2-pyrrolidone (NMP). Nordic Expert Group for Criteria Documentation of Health Risks from Chemicals. Arbete och Hälsa 40: 1-24. Arbetsmiljöinstitutet, Solna, Sweden
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||