|
[Front page] [Contents] [Previous] [Next] |
Toxicological Evaluation and Limit Values for Nonylphenol, Nonylphenol Ethoxylates, Tricresyl, Phosphates and Benzoic Acid
2. Toxicokinetics
2. Toxicokinetics
2.1 Absorption, distribution
2.2 Elimination
2.3 Toxicological mechanisms
2.1 Absorption, distribution
o-TCP absorption has been studied in a variety of species using oral or dermal administration, and p-TCP absorption has been studied after oral dosing in rats. No information is available on absorption following inhalation.
Oral intake
After a single oral dose of 770 mg 32P-o-TCP/kg b.w. to chickens, the total radioactivity in the liver increased consistently throughout 72 hours. The levels of radioactivity in the plasma were consistently lower than those in the liver. At 24 hours the plasma levels were 5% of those in the liver. The radioactivity was predominantly associated with o-TCP metabolites in the liver but with unmetabolised o-TCP in blood. (Sharma & Watanabe 1974 - quoted from EHC 1990).
Incomplete absorption of p-TCP in the intestine of rats was demonstrated after a single oral dose of methyl-14C-p-TCP (7.8 or 89.6 mg/kg b.w.) in 1.5 ml dimethylsulfoxide. Much of the radioactivity was recovered in the faeces, predominantly in the form of unchanged p-TCP (Kurebayashi et al. 1985 - quoted from EHC 1990).
Groups of three rats were given 2, 20, or 200 mg/kg b.w. of 14C-o-TCP, 14C -m-TCP, or 14C -p-TCP. Excretion studies (see later) showed that urinary excretion was of importance (60-70% of a dose excreted via urine) (Perry et al. 1983). These data indicate that extensive absorption of all three substances from the gastrointestinal tract occurs.
Dermal contact
Poor absorption of 32P-labelled o-TCP was demonstrated in a dog after a single dermal dose of 200 mg/kg b.w.. In humans, the rate of transfer (dose applied 2-4 mg 32P-o-TCP/kg b.w.) through intact human palm skin appeared to be about 100 times faster than that through the abdominal skin of the dog. This was based on urinary excretion and surface area considerations. (Hodge & Sterner 1943 - quoted from EHC 1990).
After a single dose of 200 mg 32P-o-TCP/kg b.w. to the abdominal skin of the dog, the radioactivity in the blood during the first 24 hours was equivalent to an average o-TCP concentration of 80 mg/l during the period. The radioactivity was distributed throughout the visceral organs, muscle, brain, and bone. The levels of radioactivity in the tissues were in decreasing order of concentration: liver > blood > kidney > lung > muscle or spinal cord > brain or sciatic nerve. (Hodge & Sterner 1943 - quoted from EHC 1990).
Another species, the cat, showed great dermal absorption. When 32P-o-TCP (50 mg/kg b.w.) was dermally applied to adult male cats, the disappearance of radioactivity from the application site was bi-exponential. In the first phase, 73% of the o-TCP disappeared within 12 hours, while in the second phase the half-life was 2 days. (Nomeir & Abou-Donia 1984, 1986b - quoted from EHC 1990).
In cats given a single dermal dose of 50 mg 14C-o-TCP/kg b.w., the chemical was absorbed from the skin and subsequently distributed throughout the body. o-TCP reached its highest concentration in plasma at 12 hours, and its metabolites attained their maximum concentration between 24-48 hours. The relative residence values of unmetabolised o-TCP in various tissues, relative to plasma, were: brain 0.09, spinal cord 0.18, sciatic nerve 2.1, liver 0.44, kidney 0.55, lung 1.27. Parent o-TCP was the predominant compound in the brain, spinal cord, and the sciatic nerve, while the metabolites o-hydroxybenzoic acid and di-o-cresyl phosphate were predominant in the liver, kidney, and lung (Nomeir & Abou-Donia 1984 - quoted from EHC 1990). In contrast, when measuring total radioactivity in samples 1-10 days post exposure, highest levels were found in the bile, gall bladder, urinary bladder, kidney, and liver, with only low levels in the spinal cord and brain (Nomeir & Abou-Donia 1986b - quoted from EHC 1990).
2.2 Elimination
Metabolism
The metabolisms of o-tri- and p-tricresyl phosphate (figures 1 and 2) are essentially the same, starting with oxidation of one of the methyl groups followed by hydrolysis of the ester bond yielding hydroxybenzyl alcohol, which is further oxidised to hydroxybenzoic acid. For o-TCP there is a "bypass" as a ring structure can be formed after the first cresyl moiety has been split off, this results in the formation of saligenin cyclic o-tolyl phosphate. The further metabolism of the substances follows the same general picture i.e. hydroxylation of the cresol moiety, which is then split off and oxidised. No studies on the metabolism of m-TCP have been published. (NTP 1994).
Excretion
Groups of three rats were given 2, 20, or 200 mg/kg b.w. of 14C-radiolabelled o-TCP, m-TCP, or p-TCP by gavage. For all isomers 90-100% of the dose was excreted in urine and faeces within 3 days. Rats receiving 2 and 20 mg/kg b.w. o-TCP eliminated 90% of the dose (60-70% urine, 20-25% faeces), however, at the 200 mg/kg b.w. dose only 60% of the dose had been eliminated (45% urine, 16% faeces) by 24 hours. For m-TCP equal amounts of the 20 and 200 mg/kg b.w. dose appeared in urine and faeces (24% and 12 %, respectively) by 24 hours. By three days the major route of excretion for the 20 and 200 mg/kg b.w. doses was via the faeces, while there was an equal elimination by both routes for 2 mg/kg b.w.. The excretion route and rate of p-TCP also showed dose dependence, with 59, 36, and 17% of the dose (2, 20, and 200 mg/kg b.w., respectively) appearing in the urine and 25, 46, and 47% appearing in the faeces by 24 hours. The major route of p-TCP excretion changed from 2 mg/kg b.w. (urine) to 200 mg/kg b.w. (faecal). (Perry et al. 1983).
Figure 1. Metabolism of o-TCP (From NTP 1994). Look here
Figure 2. The metabolism of p-TCP (From NTP 1994). Look here
2.3 Toxicological mechanisms
It is known that o-TCP and TCP isomers with minimum one o-cresyl group (see below for potency) exerts their organophosphorous induced delayed neurotoxicity (OPIDN) through the metabolite saligenin cyclic o-tolyl phosphate, which is at least five times more neurotoxic than o-TCP after oral administration to chickens (Bleiberg & Johnson 1965 - quoted from EHC 1990). The formula of saligenin cyclic o-tolyl phosphate is given below.
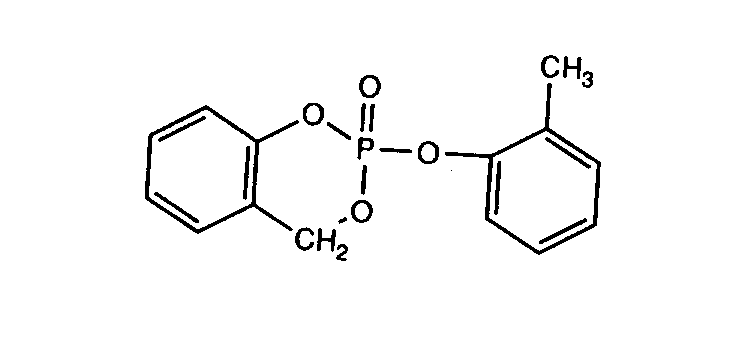
Figure 3. Structure of the neurotoxic metabolite saligenin cyclic o-tolyl phosphate.
Henschler (1959) studied the relative neurotoxicity of the 10 different isomers of tricresyl phosphates after single oral doses in hens. The most potent isomers for inducing OPIDN were the three isomers with only one ortho-cresyl group, these were equally potent. When the OPIDN toxicity of these was set to 100%, the two isomers with two ortho-cresyl groups had a relative toxicity of 50%, and the tri-ortho-cresyl phosphate had a relative toxicity of 10%. The four tricresyl phosphates without the ortho-isomer did not induce OPIDN.
Whether the different m- and p-cresyl phosphate isomers are neurotoxic through another mechanism is not clear. The neurotoxicity seen in the NTP (1994) studies (see later) might be explained by small amounts of tricresyl phosphate with one or more o-cresyl rings being present, as it is only stated that the TCP used contained less than 0.1% o-TCP.
|
[Front page] [Contents] [Previous] [Next] [Top] |