Environmental Factors and Health
10 Chemical substances and products
10.1 Human exposure to chemical factors
Chemicals are everywhere
Chemicals are ubiquitous in a modern industrialised society. Many solutions to technical problems and many new products involve the development and use of new chemicals or known chemicals in new applications.
For Denmark it has been estimated that 20,000 substances, 100,000 chemical products and 200,000 goods/industrial products are on the market (DEPA 1996)84. Therefore, sources of exposure to chemicals are a trivial and often unrecognised part of everyday life.
Chemicals, both natural and man-made, may have adverse effects on humans. Some hazardous chemicals may have severe effects such as carcinogenic, reprotoxic or mutagenic effects, other chronic effects or even death. Considerable efforts are therefore directed towards reducing the risk of exposure to these and other chemicals of concern.
Focussing on the areas of concern
The use of chemicals for production of synthetic materials and products, and as components of chemical products for an infinite number of purposes, means that chemicals are everywhere. It is impossible to give a full description of all possible carriers for which exposure of a chemical substance with a potential for human health effects may take place.
The use pattern and composition of a product containing chemicals warrants particular concern where:
| Use involves hazardous chemicals Chemicals with properties dangerous for human health are an obvious risk factor. Uses that involve pesticides or biocides, organic solvents, acrylates and other substances with pronounced biological effects. |
| Use leads to high exposure Use patterns involving a large turnover of a product, in a particular bioavailable form (gaseous or liquid) or an application leading to a high or long-term human exposure. |
| Use involves vulnerable groups Uses involving vulnerable groups are particular in focus. This may e.g. be a product used by pregnant women, for infants, children, sick or elderly. |
A number of products have usage and application patterns that place them in a ’high’ exposure group. These are addressed in the following.
Delimitations
This chapter reports on the environmental factors as they exist in the form of chemicals, their sources, uses and regulations. It also describes the most prominent instruments, actors and strategies of the issue in Denmark.
Focus is directed towards the chemicals and their use pattern, where they are regulated by DEPA. This is particularly the marketed chemical substances and preparations as such, and where these chemicals come in direct contact with non-professional human users, e.g. household chemicals and consumer articles and goods.
Some areas where a considerable direct exposure to chemicals may take place are not regulated by DEPA. This includes the working environment, food and pharmaceuticals. Also, indirect effects associated with the individual’s consumption of tobacco, alcohol, drugs, general lifestyle or living conditions are excluded. A more detailed description of responsibilities of Danish authorities in regulation of health and environmental factors is given in chapter 2 and 3.
10.1.1 Chemical factors
This chapter deals with products, where the chemical substances were put in the original product intentionally or where they appear as contaminants from used raw materials. Therefore, the term ‘environmental factor’ is adjusted to ‘chemical factor’ since the exposure will be directly from the original source with no implication of environmental issues. The exposure from the environment is treated in each of the media chapters.
Distinction is made between chemical substances and preparations in the Danish Law on Chemical Substances and Products:
| Chemical substances are the natural elements and their inorganic or organic compounds as
they are naturally or industrially produced. | |
| Chemical preparations which are mixtures of more substances (such as paints, detergents,
cosmetics, etc.) are sometimes referred to as chemical products. | |
| In addition to chemical preparations as defined in the statutory order, numerous consumer products in the form of articles and goods (a few examples are toys, textiles, building and construction materials) may also lead to exposure covered by the regulation. Typical consumer products will be addressed separately in this chapter. |
It is estimated that in the EU about 30,000 - 50,000 chemical substances are marketed, and approx. 2,700 are so-called ‘High Production Volume Chemicals’ (> 1,000 tons marketed per year) for which a certain amount of data on inherent properties must be available. Data for these substances and also the approximately 20,000 ‘Middle production Volume Substances’ (>10 tons, <1000 tons) are made available in the CD ROM database ‘International Uniform Chemical Information Database’ (IUCLID) developed by the European Chemicals Bureau (ECB 2000, IUCLID, Version 2).
Other substances are marketed in lesser amounts, some in very small quantities. Among these, there are substances of no obvious concern (e.g. common salts, carbohydrates) where the need for information is less crucial but also substances for which a targeted effort is required. For a vast number of the 100,000 chemicals little or close to nothing is known regarding their health and environmental properties.
Chemical properties/ harmful effects
The number of potential contact points between chemicals and humans are enormous. In order to identify the risk of chemical exposure, new and existing chemical substances must be classified with respect to human and environ-mental hazard based on available information on their inherent properties.
Only a limited number of the substances on the market (approx. 5,000) have been officially classified by the authorities and included in the list of dangerous substances (DEPA, 2000). However, producers and importers are obliged to evaluate the chemicals they produce and import. Furthermore to provisionally classify and label the chemicals they should meet the require-ments of the statutory order on classification, packaging, labelling, sale and storage of chemical substances and products.
Table 10.1
Examples of the chemical factors receiving most attention regarding possible
human health effects in the regulation of chemical substances, chemical preparation and
consumer articles and goods (DEPA 1996)85.
Chemical factor |
Origin and characteristics |
Health Impact |
Metals (e.g. lead, chromium, cadmium and mercury) |
Metals are used for a variety of purposes including colouring, stabilisation and as components in batteries. Metals also occur naturally in many products from which they may potentially be released. |
Selected impacts are e.g. Lead and mercury are accumulated in the body. Both organic and inorganic forms are toxic. Cadmium is cumulative and has a chronic adverse effect on renal function. Chromium (in oxidation state 6) has carcinogenic and possibly reproductive effects. |
Nickel |
Widely used in alloys and jewellery. |
Nickel is a common allergen. |
Pesticides |
Used in agriculture and forestry, but also in pest control for domestic purposes. High exposures may occur in non-professional use. |
Many different effects from the range of substances. |
Phthalates |
Plasticisers in PVC and tensides occurring in detergents and cosmetics.
|
Reproductive effects |
VOCs |
Paints and varnishes, glues, degreasers and generally as solvents. |
Acute toxicity |
Fragrances |
Personal hygiene and other odourised products. |
Allergens |
Polyaromatic hydrocarbons (PAH) |
Spillages and deposition of tar products (tar and asphalt production, gasworks, impregnation of ropes and fishing net, wood preservation, tar paper production, etc.) |
Benz(a)pyrene and several other PAHs have been found to be potent carcinogens in animal studies. There are also evidence for carcinogenicity in humans associated with exposure to these substances |
10.1.2 Sources and exposure
Chemical substances
The chemical substances, preparations, articles and goods sold to professionals and laymen, are the source of numerous direct exposures of humans to chemical factors. A great number of different items are in retail and a considerable effort is directed into labelling the chemical substances with risk and safety phrases. The objective is to limit the anticipated exposures by restricting the use or prescribing the use of specific protective equipment.
Single substances like certain solvents, metals, oils, acids and bases (Example 1) are also marketed, sometimes giving rise to high exposures to the individual substance and subsequently their related adverse effects.
Example 1
Sources of chemicals
Chemical substances
|
Articles and goods
|
||||||||||||
Chemical preparations
|
|
Chemical substances released or migrating from articles and goods is another source of exposure, which often requires detailed analysis in order to identify and quantify the actual exposure. This is especially the case when the chemical substances are a result of contamination of the raw material and not intentionally added to the products. Recent examples of such exposures are the findings of traces of tributyl tin in babies' napkins and asbestos in crayons.
Chemical preparations
The number of different chemical products (mixtures of substances e.g. paints, varnishes and detergents) are greater than the number of substances and may individually release health affecting environmental factors during their storage, use and disposal. A strategy for limiting exposure following the same path as for the chemical substances is followed.
Some specific substances have been identified as particularly hazardous: lead, cyanoacrylates, isocyanates, epoxy compounds, active chlorine and cadmium. Additional labelling is required for certain preparations containing these substances.
Substances in consumer articles and goods
Many consumer products are not perceived as the possible source of chemical factors, but the chemicals are used in the production of consumer goods. They serve a purpose, as part of the structural material, the decoration or similar, or reside as a residual concentration or contamination of the produced goods. Examples, such as lead and cadmium in glazing and enamelware, plastisicers in cling film or flame retardants in electronic equipment, may be mentioned.
Indoor living conditions may be greatly affected by the choice of building materials because of the long-term exposure and the huge areas/amount of material. Volatile compounds receive special attention, since evaporation from walls and floors may lead to significant concentration in confined, not ventilated areas with large surfaces. One example is the assigned limit value for evaporation of formaldehyde from chipboards, another issue under much consideration presently is the emission of plastisicers from PVC flooring materials.
Evaporation of chemicals from construction materials is recognised as a major component in determining indoor climate. These materials include e.g. glues, fillers, foams and other polymer materials such as paints, lacquer and varnishes. Most of the exposure is by inhalation and the effects are related to volatile organic compounds used as solvents.
Other chemicals migrating from building and construction materials during use are increasingly in focus as sources of potential health effects. The use of acrylates and related monomers may lead to direct dermal exposure, but the release and subsequent indirect exposure from the environment of e.g. plasticisers migrating from PVC has recently also been addressed.
Cleaning products
Household cleaning products may contain hazardous chemicals such as corrosives, irritants and substances toxic to humans or the environment. The products may fall in many categories. The diversity is exemplified by a few products from the cleaning sector: Dishwashing, car wash, drain cleaner, wax, dry cleaning agents, engine wash, window cleaning, protective coatings, stain remover, speciality metal cleaning products, decalcifiers.
The chemicals used in cleaning products are mainly organic and a number of them are among the undesired substances: alkylphenolethoxylates, phthalates and many are solvents.
Cleaning products are used in large quantities and for some of these products high exposures can also be foreseen, especially substantial dermal exposure. Many cleaning products containing detergents are used in connection with warm water, which increases the risk of skin absorption and inhalation of aerosols and gasses. For a vulnerable group such as children a higher than average exposure must be anticipated from residues on floors and other surfaces, and from inhalation and contact to dust particles.
Cosmetics
Cosmetics are typically used directly on human skin and consequently a large dermal exposure is expected. Some are used near mucous membranes (e.g. eyes and mouth) which may increase absorption of the substances or repetitively on large skin surfaces e.g. lotion for babys/infants. Examples include lipstick, perfume, shampoo, make up, deodorants, sun tan lotion, and toothpaste.
Adverse effects have been attributed to some of the substances traditionally used in cosmetics in low concentration. In high concentrations, these substances show mutagenic, allergenic and reproductive toxicity, e.g. isopropyl alcohol, musk xylenes, DEHP.
Allergy is the most common adverse effect related to cosmetics often caused by preservatives and fragrances. A reason for concern is supported by the fact that the incidence of cosmetic allergy is increasing.
Toys
Infants and small children (< 3 years) represent a vulnerable group by themselves, but also requires special attention because they have high hand-to-mouth ratios and risk exposure via mucous membranes. Much effort is therefore directed towards evaluating migration of substances from the products under conditions mimicking the gastro-intestinal milieu and the substance’s oral toxicity.
The phthalates have been one of the target substances for restrictions recently, since the migration and risk of effects from these substances in flexible PVC were deemed unacceptable by the Danish authorities.
For a number of metals used for colouring in grease paint, paints and crayons limit values have been applied since it is estimated that both oral and dermal exposures can be expected.
Clothes/textiles
In clothes and other textiles a similar attention is given to migration of material components, production chemicals and residues of pesticides or preservatives. The main cause for attention is not the hazard associated with the substances themselves, but rather the fact that a close contact may take place between the material and a large part of skin surface for a relatively long time.
Pesticides
Exposure to pesticides (including biocides) is of particular interest because of the proven biological effects and the products therefore represent a high-risk area in terms of toxicity. Human exposure may take place during mixing of formulations and filling of equipment, and in the application process in the field. Especially the non-professional user may wear insufficient protective equipment or none at all and is therefore not protected against dermal exposure and inhalation (www.agrsci.dk)86.
A higher than average risk level may also be attributed to children playing on pesticide treated lawns. Likewise, the treatment of kindergartens, playing rooms and buildings to combat e.g. insects and other pests may infer higher risk to children.
Biocides occur in many chemical products for preservation purposes, but biocides are also used in building and construction materials, fabrics and other materials to prevent the biological degradation processes.
10.1.3 Risk to human health
Relatively little is known about the actual health effects and the effects of the combined impact from different sources on public health related to the use of chemicals. In some areas, a certain level of knowledge is available. The acute effects of a number of chemicals are relatively well known from workplace and consumer exposure situations, accidents, and poisoning incidents, e.g. from reporting of acute toxicity to emergency sections at hospitals and to the poison control centre. Chemical pneumonia from drinking and aspiration of lamp oil is one such example. In relation to acute effects like toxic, harmful, irritant and corrosive effects, it is usually easy to establish a relation between the exposure to a given chemical and the actual effect, because the response follows immediately after the exposure.
A profound knowledge of the combined pressure of chemicals to the health status in Denmark is not available, and obviously neither of chemicals as they occur in consumer products.
In general, it is difficult to establish firm scientific evidence for long-term effects like cancer, mutagenicity and reproductive toxicity in humans because of numerous confounding factors related to life-style which can be difficult to account for in epidemiological studies. Long-term effects develop over a long period (sometimes many years) and the exposure pattern becomes very complex in epidimiological studies making a causal relationship between exposure and effects very difficult to establish. There are however, some chemicals for which adequate data for this relationship has been generated both in animal experiments and from observations in humans, e.g. carcinogenic substances such as benzene, asbestos, chromium(VI)-compounds, ethyleneoxide, nickel compounds and vinyl chloride.
Pesticides are well investigated compared to other chemicals. Both acute and chronic effects are tested in animals and the results are extrapolated to humans. DEPA does not find it ethical to use tests on humans with regard to pesticide exposure.
In 1995, it was concluded by the Ministry of Environment and Energy that some chemical factors may influence the life expectancy in a negative direction, and perhaps does so in Denmark. A number of specific factors and effects are mentioned: Chemical carcinogens as such, radon in indoor air, ozone depleting substances increasing UV transmittance, lead and dioxins increasing neural dysfunction, and compounds with reproductive effects. A range of allergenic responses, including eczema and asthma, can also be coupled to exposure to chemical substances and products.
When it comes to long term exposure of the population, knowledge is more scarce, as it can be very difficult to relate the use of various chemicals to the incidence of cancer, reproductive toxicity, allergy and other effects. Again knowledge from workplace exposure and related effects provides some indication of a relation between e.g. the development of eczema, allergies and reduced tolerance to chemical substances, and the exposure of the population to chemicals.
Knowledge about the cancer incidences in the population is difficult to relate to the use of chemicals. This is because very little is known about the actual level of exposure to chemicals and also because a number of life style factors may influence the development of cancer. The impact on the state of health of combined exposure to chemicals and chemicals in combination to other environmental factors like e.g. noise is another area characterised by limited knowledge.
Lack of knowledge makes it therefore very important to identify exceptional exposure situations involving hazardous chemicals, high and repeated exposure and exposure of vulnerable groups like children and pregnant women. Realising this, DEPA has intensified its efforts to identify such specific exposure situation which will also form a background for the future protection of vulnerable groups.
This section focuses on the direct exposure to ’containerised’ substances and products in the human sphere. The main concerns for risk of health threatening exposure are the previously mentioned areas with
| Hazardous chemicals | |
| High exposure | |
| Vulnerable groups |
The exposure to chemical factors related to chemical substances and preparations, articles and goods is by far the most direct exposure situation. In this area, however, indirect exposure takes place as well, e.g. in the form of pesticide residues in food or substances migrated from food packaging material.
The main overall distinction in exposure pattern is between the chemical substances, preparations and products used in the industry, incl. agriculture, and those used in the household sector. Typically, more hazardous chemicals are allowed to be used in the industrial sector because the users in the working environment have access to training and protective equipment. Especially, more CMR substances and products may be used (CMR is short for carcinogenic, mutagenic and reproductive harmful substances). However, occupational exposure of professionals is not covered here.
In a number of instances laymen also risk exposure to ‘professional’ products. These include the many uses by ‘do-it-your-self’ people using such products in building and construction, painting and decoration, washing, cleaning and rinsing of surfaces and items. It is therefore the products (the vehicles) rather than the sectors, which are in focus as the source of exposure for the non-professional user of such products. As for the consumer, a great number of items comprise the everyday sources of exposure and they are also coupled to the products – not the sectors. The exposure is therefore addressed via the products of use.
Children, as well as the unborn child, have in some cases appeared to be uniquely vulnerable to chemical substances because of their biological growth and development. Furthermore, children may be more heavily exposed than adults to certain chemicals and pollutants in the environment because children, on a body weight basis, breathe more air, drink more water, and eat more food than adults. Additionally, their behavioural patterns, such as play close to the ground and hand-to-mouth activities, can increase their relative exposure.
10.2 Level of protection
From the beginning, the primary objective of chemical regulation has been to protect humans from the adverse health effects from exposure to chemicals, also illustrated by the previous Danish Poison Act from 1961.
The Poison Act applied to substances, which were not covered by other acts, e.g. the Pesticide Act. According to the poison act, substances were classified and categorised in one of the following categories: ‘strong poison’, ‘poison’, ‘harmful’, 'harmful organic solvent’, and ‘harmful gas’. Substances were not evaluated with regard to long-term effects. Criteria for long-term effects were introduced through an amendment to the European Substance Directive which was first issued in 1967 (EU, 1967). Much later, through the 12th adaptation to this directive, criteria for evaluation of environmental effects for substances were introduced.
The main tools used to define and administer the level of protection are risk assessment procedures and classification criteria for classification of chemical substances and preparations. In general, the classification of chemical substances forms the background for all downstream regulation.
For some specific chemical products, e.g. plant protection products and biocides, the level of protection is primarily built into pre-marketing approval schemes, as these products because of the way they are used may be particularly hazardous to the health. In the process of the approval by the authorities, a detailed risk assessment is performed and the need for specific restrictions attached to the use of the products is evaluated before these are put on the market. In the downstream regulation restrictions in marketing and use are either based on the classification of substances and preparations or on specific risk reduction measures. The latter resulting from risk assessment of the particular substances as part of the risk assessment procure for new and existing substances. For example substances classified as Very toxic or Toxic because of CMR effects cannot be sold to the general public. These measures also contribute to sustain a certain level of protection.
Vulnerable groups like children and pregnant women are often more at risk to exposure from chemical factors than the general public and are therefore also getting increasing attention in relation to regulatory development and the level of protection as such. As a result of this, DEPA is working to increase the level of protection in relation to certain uses of chemicals, e.g. with regard to toys and cosmetics. Another area of concern is exposure to endocrine disruptors, which are currently not addressed directly in the regulations. DEPA therefore finds it very important to improve this area in the regulations.
It is difficult in quantitative terms to define the level of protection in chemical regulation, as protection levels from different kinds of regulations in different areas can hardly be compared. Thus a protection level in more qualitative terms may be reflected in an overall view of which kind of regulations that have been issued.
Denmark has, in order to obtain a high level of protection of human health, prioritised bans and restriction of a number of problematic substances including heavy metals like cadmium, mercury and lead, ozone depleting substances, creosote, brominated flame retardants, phthalates and PVC. These restrictions are motivated by either the hazardous properties, the amount used, the method of use or the exposure of vulnerable groups.
The principles for setting health based quality criteria for specific chemicals in order to prevent health hazards in the human population caused by chemical substances as pollutants are discussed in relation to the various media (air, soil and drinking water) and are outlined in more detail in Appendix 1.
10.2.1 Pre-marketing approval
Plant protection products and a great variety of biocides are subject to pre-marketing approval schemes, which in a very direct manner contributes to the level of protection. In general these chemicals are considered to be particularly hazardous to health and are used in large quantities.
Pre-marketing approval implies that the responsible importer or manufacturer must submit a detailed dossier to the competent authority, DEPA. DEPA will then make an evaluation of the data in the dossier before an approval can be assigned. DEPA can also decide to restrict the approval if the forwarded dossier gives reasons for concern in relation to e.g. the planned use.
10.2.2 Classification and labelling of substances and preparations
The purpose of the classification and labelling system is to consider the hazards arising from normal handling and use of substances and preparations in the form they are placed on the market, i.e. to:
| identify all the inherent physico-chemical and toxicological properties and for
substances also the environmental properties, which may involve a risk during normal
handling and use, - and | |
| inform the general public about the hazards and adequate precautionary measures for handling and disposal in order to prevent accidents and harmful effects among the users. |
Substances are categorised according to the identified properties in one or more categories of danger expressing physico-chemical, toxicological and environmental properties. Only classification with respect to human health is addressed in this section, i.e. classification based on toxicological properties.
The following danger categories are related to health:
| Very toxic | |
| Toxic | |
| Harmful | |
| Corrosive | |
| Irritant | |
| Sensitising | |
| Carcinogenic | |
| Mutagenic | |
| Toxic to reproduction |
The classification into these nine danger categories is based on existing data and reflects the present level of knowledge and possibilities for testing the various endpoints. These criteria are continuously developed as more know-ledge is generated. DEPA has especially worked to promote more adequate criteria for neurotoxicity, immunotoxicity, reproductive toxicity and impact on the endocrine system. This is also to be better prepared for handling the endocrine disrupters in relation to the endpoints for this newly acknowledged effect.
For endocrine disruption87, appropriate test methods have still not been developed for the various endpoints, and this effect is therefore not considered by the classification system. Also for some of the well-known effects like neurotoxicity and respiratory allergy, the test methods have been questioned and are still not fully developed.
Because of the problems with testing for chronic brain damage, it has been difficult to reach agreements in EU about how to evaluate and classify many organic solvents with regard to neurotoxicity in the past. Many organic solvents were only classified with regard to flammability in EU, and Denmark has used the safeguard clause in several situations in order to introduce a classification reflecting the neurotoxic potential as expressed through the risk phrase R48/20 (Danger of serious damage to health by prolonged exposure through inhalation). Recently, two additional risk phrases have been introduced to cover typical solvent-related health hazards, namely R66 (Repeated exposure may cause skin dryness or cracking) and R67 (Vapours may cause drowsiness and dizziness).
The criteria for classification do not focus on sensitive subgroups, e.g. children. However, a number of R- and S-phrases are specifically related to the protection of children and/or pregnant women.
As classification is based on the inherent properties of the substance or preparation in the form it is marketed, it does not take the actual use situation and the exposure into consideration. For example whether the chemicals are intended to be heated during use, diluted, sprayed or processed in any other way.
The classification of a substance / preparation comprises the relevant danger categories and the related risk phrases. Risk phrases express the type and degree of danger from the different routes of exposure. In the classification system there are in addition some risk phrases, which can be assigned to substances classified for other effects. These phrases cover accumulation in the human body and danger to breast-feeding children. The classification system includes currently 65 individual risk phrases of which 33 are directly related to health hazards.
Examples of danger categories and related risk phrases:
Danger category |
Risk phrase |
Toxic |
R23: Toxic by inhalation |
Sensitising |
R43: May cause sensitisation by skin contact |
Corrosive |
R35: Causes severe burns |
Mutagenic (Mut 3) |
R40: Possible risk of irreversible effects |
Carcinogenic (Carc 1 or 2) |
R49: May cause cancer by inhalation |
Labelling serves the purpose of communicating the potential hazard through danger symbols, risk and safety phrases and other required information on the label.
The use situation is considered by labelling the substances and preparations with safety phrases expressing adequate precautionary measures in relation to handling and disposal of the chemicals. Safety phrases, some of which are obligatory, are assigned based on the classification of the substance/preparation and a defined set of criteria. Preparations which are intended to be applied by spraying must be assigned with specific safety phrases warning about the exposure (S2388) and giving advise on protective measures (S3889 or S5190), as the application is expected to give rise to a considerable exposure.
The health related danger symbols and indications of danger are shown in Figure 10.1 in order of their degree of danger.
Figure 10.1
Health related danger symbols
 Very toxic |
 Toxic |
 Corrosive |
 Harmful |
 Irritant |
The labelling rules also includes some requirements for child-resistant fastenings and tactile danger warnings for certain hazardous chemicals aiming to protect vulnerable groups like children and blind people against exposure from the chemicals.
The classification and labelling rules and the ranking of the chemicals in different classes according to their degree of danger form the basis of a large number of administrative rules with regard to restrictions in marketing and use and further priority setting in much of the downstream regulation. Only a small proportion of the substances already on the market are classified in the regulations, a fact which may be a concern in relation to the level of protection. In order to compensate for this problem, DEPA supports the use of group classification of chemical substances and QSAR as methods to evaluate the substances in order to raise the level of protection. DEPA has therefore recently issued a draft guidance list for self-classification of dangerous substances. The list contains 20,624 substances, which have been classified with regard to acute toxicity from ingestion, allergy from skin contact, mutagenicity, carcinogenicity or dangers to the aquatic environment. The substances have been identified among 47,000 substances using QSAR (Quantitative Structure-Activity Relationship) computer modelling. A final list is expected to be issued in 2001.
10.2.3 Risk assessment of chemical substances
Risk assessment of chemical substances forms the basis of the risk reduction strategies and thereby the level of protection. Lack of data is, however, one of the challenges when securing a sufficient level of protection of human health from exposure to chemicals. Only a limited number of the chemicals which are already on the market have been adequately investigated and even fewer have been through the risk assessment procedure.
The risk assessment process, in relation to both human health and the environment, entails a sequence of actions: effect assessment (hazard identification and dose (concentration) - response (effects) assessment), exposure assessment (estimation of the concentrations/doses to which human populations or environmental compartments are or may be exposed), and risk characterisation (estimation of the incidence and severity of the adverse effects likely to occur in a human population or environmental compartment due to actual or predicted exposure to a substance).
The risk characterisation is then carried out separately for three subgroups of the population: workers, consumers, and man exposed indirectly via the environment. In the risk characterisation for consumers and man exposed indirectly via the environment, the interspecies variation is taken into account when considering the margin of safety (the ratio between the No Observed Adverse Effect Level (NOAEL) or Lowest Observed Adverse Effect Level (LOAEL) and the estimated exposure).
The principle of general risk assessment of chemicals is to evaluate the inherent
properties (the hazards) of the chemical and to make a risk
assessment taking account the most probable exposure of man and environment, with special
reference to protect workers, consumers and other persons ex-posed via drinking water,
food, soil or other parts of the environment. Most often exposure data are not available
for all of these groups and therefore standard scenarios reflecting realistic worst case
situations are used and also modelled in computer models. In this respect, the Danish and
other product data from product registers like the Danish can be an important source of
information about the actual use of many chemicals in the workplaces, but in general the
exposure assessments are subject to some uncertainty. For some substances (e.g.
phthalates), children have been considered as a specific subgroup of consumers as they may
be exposed to these substances in a way which is not considered relevant for adults.
Furthermore they may be more susceptible to the exposure of specific chemicals e.g.
endocrine disrupters.
The EU risk assessment procedure for existing substances listed on EINECS91, applies in principle to more than 100,000 substances, but in practice the 141 priority substances has been selected among the 2,700 substances produced in the highest volumes (above 1000 tons). Only very few substances have been processed through the risk assessment procedure until now. It is not the idea to assess all the substances on EINECS, partly because it is impossible to manage, but also because a considerable number of the substances most likely are not on the market today. Other methods to be used for the assessments have been discussed, e.g. the previously mentioned substance groups evaluations and use of Quantitative Structure-Activity Relationships (QSAR).
10.2.4 Restrictions in use
Rules concerning restrictions in use or actual bans of substances hazardous to health or environment can either be based on decisions taken in international fora or they can be based solely on national decisions.
Denmark has given priority to bans and restrictions of a number of substances including heavy metals like cadmium, mercury and lead, ozone depleting substances, creosote, brominated flame retardants, phthalates and PVC. These restrictions are motivated by either the hazardous properties, the amount used, the method of use or the exposure of vulnerable groups.
10.2.5 Precautionary principle
The precautionary principle is a principle that opens up for regulatory action on the basis of suspicions for harmful consequences rather than waiting for cases or a full scientific evidence. In the context of this report, the use of the precautionary principle may be relevant for the aim of protecting vulnerable groups in the population against potential hazardous exposures for which the harmful consequences are not fully understood.
The precautionary principle has been incorporated in a series of international treaties (e.g., the EU Amsterdam Treaty from 1998) and declarations. In many of these, the principle has been connected mainly to hazardous chemical substances, but there are also examples of broader approaches to environ-mental issues.
In Danish legislation the precautionary principle is not explicitly mentioned, but it is often reflected in the introductory comments to various environmental acts, the central ones being the Environmental Protection Act, the Chemical Substance and Product Act, the Marine Environment Act, and the Gene Technology Act.
Both in Denmark and abroad, there has been increasing focus on the precautionary principle in recent years. In line with the increasing complexity of environmental issues, politicians, enterprises, and authorities are expected to allow this doubt to benefit the health or the environment.
However, a uniform and internationally agreed interpretation of the precautionary principle does not exist. Denmark has argued that the principle should be undertaken in cases where potentially adverse effects are of concern, but scientific evidence is unavailable due to lack of data. Finally, the precautionary principle is mentioned in the final conclusions from the Nice summit. Here the Council states that under international law, the Community and the Member States are entitled to establish the level of protection they consider appropriate in risk management. Further, that they may take the appropriate measures under the precautionary principle and that it is not always possible to determine in advance the level of protection appropriate to all situations.
10.2.6 Protection of vulnerable groups
In 1998, the Ministry of Environment and Energy participated in a conference on children and chemicals together with several other ministries. Each ministry presented a status report on how the protection of children and pregnant women is incorporated in its work. The overall impression from the discussions was that children and pregnant women should generally be recognised as special risk groups with respect to effects from exposure to chemical substances. As a consequence, a project was initiated by DEPA titled "Children and the unborn child: exposure and susceptibility to chemical substances – an evaluation". The aim of the project was to elaborate a detailed review and update the knowledge on the exposure and vulnerability of humans to chemical substances during the embryonic, foetal and postnatal periods. The report is intended to form the scientific basis for future regulatory work of the DEPA in the protection of children and the unborn child to environmental chemical substances and it also gives some recommendations for this work.
The regulation of chemical substances is generally based on the present knowledge regarding adverse health effects and risks related to the use of the substances. However, the available data on a specific chemical substance is seldom sufficient to evaluate whether children or the unborn child should be considered more vulnerable than adults. Concerning specific end-points such as fertility and teratogenicity, data exist only for 20 to 30% of the 2,700 high production volume chemicals, and for thousands of chemicals produced in smaller quantities, this percentage is expected to be considerably lower. Furthermore, for most chemical substances, limited data are available concerning exposure of children as well as of adults.
From a regulatory point of view, different approaches can be applied in order to protect children, including the unborn child, from adverse health effects resulting from exposure to environmental chemical substances. One regulatory approach is to improve the risk assessment of chemical substances by including a requirement for data specifically relevant for the protection of children, including the unborn child. Another approach is to pay more attention to children and pregnant women in the risk management process.
10.3 Regulation and strategy
10.3.1 The Danish strategy
Regulation of chemicals is under the auspices of the Ministry of Environment and Energy. In the 1980s and 1990s, Denmark strengthened efforts in the field of chemicals. In 1999, the Danish government presented a new strategy on chemicals, for the period up to 2002, aiming at strengthening international co-operation, making the chemical industry more responsible, strengthening national regulation and enforcement and increasing public access to information (DEPA, 2000).
DEPA has issued strategies and actions plans in e.g. the following areas:
| Brominated flame retardants | |
| Phthalates in flexible PVC | |
| Ozone depleting substances | |
| Pesticides | |
| Heavy metals |
Information about the action plans are available at DEPA's homepage: http://www.mst.dk/activi/01000000.htm.
10.3.2 Objectives and principles
Objectives
The primary objective of the chemicals regulation is to prevent health hazards and environmental damage caused by the use of chemicals and to promote the use of cleaner technology through the administration of the Act on Chemical Substances and Products.
In DEPA's strategy for intensified efforts in the field of chemicals, the main objectives are presented as follows:
| the consumption of problematic chemicals shall be reduced | |
| the control shall be strengthened, manufacturers responsibility increased and the consumers access to information improved | |
| the EU risk assessment procedure shall be made more coherent, simplified, efficient and faster | |
| the Danish efforts to secure a chemicals regulation at a global level should be strengthened. |
Principles
Chemical regulation in Denmark rests in general on the following principles and instruments:
| Notification and approval of chemical substances and preparations | |
| Evaluation, classification and risk assessment of chemicals | |
| Bans or restrictions | |
| Substitution of dangerous chemicals with less dangerous ones | |
| Use of cleaner technology and products | |
| Inspection and penalties | |
| Information and campaigns | |
| Voluntary agreements | |
| Green guidelines for purchasing | |
| Taxes and fees | |
| Subsidies |
10.3.3 Legislation on chemical substances and products
Danish regulation in the chemical area is, as mentioned, strongly related to the European Union legislation, which in this area is mainly based on totally harmonised directives. Classification and labelling, notification of new chemicals, use restrictions, and hazard and risk assessment are all areas where full harmonisation applies. In some of the directives the so-called ’safeguard clause’ exists which makes it possible for the Member States to implement provisional rules, which are stricter than the rules of the EU. Denmark has on some occasions used this clause, e.g. in relation to classification of certain organic solvents.
Denmark has also issued more strict regulations in areas, covered by minimum regulation in the EU. This regards for instance ozone depleting substances.
In addition to the regulation based on EU directives, Denmark has maintained and issued specific national regulation in areas not addressed by EU regulation, e.g. in relation to mercury, lead and restrictions of substances which are allowed in aerosol containers. Another example is the permit system whereby a consumer must obtain permission from the police to buy toxic substances.
10.3.4 Legislative background
Existing Community legislation on environmental and human health aspects of chemicals is based on a three-stage approach. It includes a hazard identification stage, in which a substance’s inherent capacity to cause adverse effects on human health and the environment is identified, on the basis of the intrinsic properties of a substance. The second stage consists of risk assessment, which is based on an assessment of the hazard combined with an assessment of exposure to the chemical substance. The third and final stage is one of risk management, in which strategies for the management of the risks are developed. This approach is reflected in the EU regulation.
The main EU Directives on chemicals governing Danish regulations are shown in Appendix 6.
National legislation and implementation
The Act on Chemical Substances and Products
The main act regulating chemicals is the Act on Chemical Substances and Products. This act was issued in 1979 and has been amended and revised several times, latest in 2000. The act covers all chemical substances and products and is intended to prevent health hazards and environmental damage caused by the use of chemical substances. It also aims at promoting the use of cleaner technology through the administration of the Act. The Act shall ensure that the dangers presented by substances sold in this country are highlighted, and that sale and use of chemical substances and products known or believed to be hazardous to health are regulated.
As a general rule, the Act does not regulate the content of chemicals in common products and goods, however in certain sections of the act, goods are included. Exposure of humans to chemicals diffusely leaching from consumer goods is only covered by specific regulations. Described in a simplified way, one could say that the environmental regulation in particular regulates environmental conditions involved with production, while the chemical regulation contributes with rules aimed at the later stages in the life cycle of the chemicals, e.g. distribution, use and as waste. (DEPA, 1996).
Text box 10.1
Objectives and Scope of the Act on Chemical Substances and Products
|
A number of statutory orders have been issued pursuant to ACSP, banning or restricting the use of a number of substances, e.g. heavy metals and ozone depleting substances.
Classification and labelling
Directive 67/548/EEC on the approximation of laws, regulations and administrative provisions relating to the classification, packaging and labelling of dangerous substances (the Substances Directive), is an important single market directive that seeks to ensure the operation of the European single market in chemicals and to provide high standards of human health and environmental protection.
In Denmark, the Statutory Order on classification, packaging, labelling, sale and storage of chemical substances and preparations (no. 734 of 31 July 2000) implements the relevant EU Directives into national legislation. The Statutory Order provides the regulations for evaluating the inherent properties of a chemical substance or preparation in relation to hazard identification and assessment in line with the requirements in the Substance Directive described in 10.2 and Appendix 5.
Denmark has issued a statutory order on the List of dangerous substances which implements Annex I to the Substance Directive. For white spirit (CAS-no. 8052-41-3) and solvent naphtha (CAS-no. 64742-88-7) Denmark has used the safeguard clause and implemented a more strict national classification including the risk-phrase R48/20 (Danger of serious damage to health by prolonged exposure by inhalation). The latest revision of the list (no. 733 of 31 July 2000) implements the 26th adaptation to the Substance Directive. Another national feature is the "Ae" code assigned to certain substances in the list. Substances with this code must not be used in spray canisters (aerosol containers) unless other legislation lays down special provisions hereon. The Statutory Order no. 1042 of 17 December 1997, on restriction of the sale and use of certain dangerous chemical substances and products for specific purposes includes a provision that spray canisters must not contain substances classified as "very toxic" or "toxic" and these substances are therefore also assigned the "Ae" code in the list.
As described in section 10.2.2, DEPA has recently issued a draft guidance list for self-classification of dangerous substances comprising 20,624 substances, which have been identified using QSAR. The accuracy of the predicted classifications varies from approximately 70 to 85%. The list is not intended to be mandatory, but exclusively to be a guidance list in case no other documentation is available.
Notification of new substances
A new chemical substance is a substance, which is not listed in EINECS as being on the European market in the period 1 January 1971 and 18 September 1981. Substances notified in the period from 18 September 1981 are listed on ELINCS (European List of Notified Chemical Substances), a list of approximately 400 substances.
New chemical substances must be notified with DEPA. Any manufacturer or importer of a new chemical substance shall, prior to sale, submit notification of the substance to the Danish Environmental Protection Agency. The detailed notification file must include, inter alia, a description of the intrinsic physio-chemical, toxicological, and eco-toxicological properties of the substance. The larger the amount of substance placed on the market, the more data must be submitted to the authorities. The notifier may also provide a preliminary risk assessment to support the succeeding risk assessment procedure carried out by DEPA.
The following chemical substances exclusively imported or sold in the following products ready for use are excluded from the notification requirement:
- Medicinal products for human or veterinary uses.
- Foodstuffs.
- Animal feeding stuff.
- Radioactive substances as defined by Directive 80/836/EEC92.
- Mixtures of substances in the form of waste.
- Pesticides to be authorized under Part 7 of the Act.
- Cosmetic products.
Between September 1981 and September 2000 less than 10 substances have been notified in Denmark, where only limited production of basic chemicals takes place.
Restrictions in marketing and use
The ongoing amendments regarding the main EC Restrictions Directive (76/769/EEC) are either implemented through individual orders or through the Danish statutory order no. 1042 of 17 December 1997 on restrictions on the marketing and use of certain dangerous substances and preparations for specific purposes, latest amended 22 April 2000. The order also covers national provisions. Moreover, Denmark has issued strictly national provisions restricting certain other chemical substances and products.
Examples of amendments regarding the main EC Restrictions Directive (76/769/EEC) implemented by individual orders, include:
| Ban against import of certain Nickel-containing products (94/27/EEC) | |
| Ban against sale, import and manufacture of Cadmium containing products (91/338/EEC) | |
| Order on PCB, PCT and substitutes (85/467/EEC) | |
| Order on restriction in sale and use of pentachlorophenol (91/173/EEC) |
Examples of amendments regarding the main EC Restrictions Directive (76/769/EEC) implemented by the Danish statutory order no. 1042 of 17 December 1997, include:
| Benzene: This substance is not permitted in toys or parts of toys as placed on the market where the concentration of benzene in the free state is in excess of 5 mg/kg of the weight of the toy or part of the toy. Benzene must not be used in concentrations equal to or greater than 0.1 % by mass in substances or preparations placed on the market (82/806/EEC). |
| Arsenic: Arsenic compounds may not be used for antifouling, preservation of wood, or for the treatment of industrial waters irrespective of their use (89/677/EEC). |
| Mercury: Mercury compounds may not be used for antifouling, preservation of wood, impregnation of heavy textiles, or for the treatment of industrial waters irrespective of their use (89/677/EEC). |
| Chloroform, tetrachloromethane, 1,1,2-trichloroethane, 1,1,2,2-tetrachloroethane, 1,1,1,2-tetrachloroethane, pentachloroethane, 1,2-dichloroethyleneor 1,1,1-trichloroethane: These substances, or products containing them, in concentration of more than 0.1 % w/w must not be placed on the market for sale to the general public and/or in diffusive applications such as in surface cleaning and cleaning of fabrics. Preparations with a higher content must be labelled "For industrial installations only" (96/55/EEC). |
Denmark has in addition to the substances mentioned here restricted or banned a number of other substances, e.g. ozone depleting substances, and 'Ae'-substances as well as 'very toxic' and 'toxic' substances in consumer products. In some cases, stricter rules are applied compared to the EU requirements, e.g. in relation to asbestos, PCP, cadmium and creosote (DEPA, 1999).
It should be noticed that Denmark considers the main EU directive (76/769/EEC) a 'minimum directive', but this opinion is not shared by the other Member States.
Examples of orders regarding strictly national provisions, include:
| Ban against sale and use of certain products containing lead |
| Ban against certain ozone depleting substances |
| Order on cadmium in fertilisers |
| Order on propellants and solvents in aerosol containers. |
| Limitation of formaldehyde in chipboards, etc. used for furniture, equipment, etc. |
| Ban against sale and export of mercury and mercury-containing products. |
| Labelling and restriction of import, sale and use of surface treatment products. |
| Ban against phthalates in toys for children aged 0-3 years. |
Example 2
Nickel
In two comparable studies carried out in 1985-86 and 1997-98, the focus was among other sensitisers on the frequency of nickel sensitisation in persons of various ages. Sensitisation to nickel was the most common in both studies. In 1985-86, 13,8% of the patients tested had positive reactions to nickel compared to 15% in 1997-98. Nickel allergy was four to five times more frequent among women both in 1985-86 and in 1997-98. Nickel allergy is caused by close repeated skin contact with objects that release nickel. Nickel-plated jewellery especially earrings play a major role in nickel sensitisation. Among children aged 0-18 years, nickel allergy decreased significantly from 24,8% to 9,2% from the first to the second study. No significant changes in nickel allergy were seen in the age groups above 18 years. This decrease is explained as the initial effect of the nickel exposure regulation, while an increase or unchanged prevalence would be expected in the older age groups. Frequency of nickel allergy in studies carried out in 1985-86 and in 1997-98
(Source: British Journal of Dermatology 2000; 142; 490-495) |
Since the 1980s, the consumption of heavy metals has successfully been cut by 99 percent, and on Danish consumption of ozone layer depleting substances, for which consumption has fallen by 50% over the last decade, and on certain organic compounds such as PCP.
Cosmetic products
Cosmetic products have been regulated in Denmark since 1961. The EU directive on cosmetics, which was adopted in 1976, is intended to ensure that cosmetics are not harmful to consumers under normal use. The EU regulations are implemented with the Danish Statutory Order no. 594 of 6 June 2000 on cosmetic products.
The content of chemical substances in cosmetic products are regulated in three different ways:
| The responsibility for the safety of the products rests as a general rule with the manufacturer/importer | |
| Through negative lists of banned substances and products | |
| Through positive lists of allowed substances and products, which may be used as colouring agents, preservatives and UV-filters. These positive lists also include certain restrictions for use, limit values and/or time limits. All other substances for the mentioned purposes are banned. | |
| Through a list limiting or regulating the use of certain other substances, e.g. limitation in the type of products for which the substances are allowed, limit values and/or labelling criteria. |
Denmark has with varying success suggested more stringent rules to be included in the adaptations to the directive. Currently Denmark is working to improve the requirements on ingredient labelling by including of fragrance allergens. Because of the known risk of allergy related to some perfume compositions, Denmark has now urged the Commission to consider a requirement for declaration. As of now, perfume and aromatic compositions are only referred to as 'perfume' and 'flavours'.
Cosmetic products must be registered with DEPA, mainly with information identifying the manufacturer or the person/company responsible for marketing the product and the product. The manufacturer is in addition obligated to submit additional information upon request from the Competent Authority, DEPA.
Toys
The EU directive on toys was issued in 1988 and uses a regulatory approach called ‘the new method’93, which implies that detailed regulation is exempt from discussions in the Council as well as in the Commission. Instead the private European standardisation organisations, CEN/CENELEC, are charged with the task of filling out the general rules in the directive.
The provisions regarding safety of toys have been implemented by the Danish National Consumer Agency through the Regulation on safety requirements for toys and products which due to their outward appearance may be mistaken for food (DNCA, 1995). This regulation is issued in agreement with the Ministry of Housing and Urban Affairs, the Ministry of Environment and Energy, and the Ministry of Health. In Denmark, DEPA administers the part of the control, which relates to the chemical properties of toys.
In particular, for the protection of children's health, bioavailability resulting from the use of toys must not exceed the following levels per day:
Chemical substance |
Maximum bioavailability |
Antimony |
0.2 �g |
Arsenic |
0.1 �g |
Barium |
25.0 �g |
Cadmium |
0.6 �g |
Chromium |
0.3 �g |
Lead |
0.7 �g |
Mercury |
0.5 �g |
Selenium |
5.0 �g |
In addition, toys must not contain dangerous substances or preparations within the meaning of the Substance Directive and the Preparations Directives (67/548/EEC and 88/379/EEC) in amounts, which may harm the health of children using them. At all events, it is strictly forbidden to include, in a toy, dangerous substances or preparations if they are intended to be used as such while the toy is being used.
DEPA has in March 1999, as mentioned in 10.2.6, issued a statutory order prohibiting the use of phthalates in toys for children under three years of age and certain childcare articles.
Pesticides (plant protection products and biocides)
Pesticides are divided in two main groups depending on their application:
One main group is called plant protection products, used particularly in farming, forestry, and horticulture and gardening as herbicides (weed killers), insecticides and fungicides This group also includes microbiological products that consist of living micro-organisms (bacteria or viruses). It also includes plant growth regulators.
The other main group is biocides (non-agricultural pesticides), which among other things, are used to preserve wood, against insects and fungi in buildings and on livestock and pets, and to exterminate rats and mice. Biocides also include repellants and attractants. In the EU, biocides are regulated separately through the new Biocidal Products Directive.
Plant protection and also biocidal products are made up of one or more so-called active ingredients - the substances effective against one or more pests. In addition, such products can consist of various additives, auxiliary products, solvents and water.
In Denmark, the Plant Protection Directive and the Biocidal Products Directive are implemented in Danish law through Act on Chemical Substances and Products, latest amended through Act. no. 256 of 12 April 2000, and the Statutory Order on Pesticides, latest amended on 5 May 2000 through Order no. 313. These amendments have been made in order to implement the Biocidal Products Directive.
The legislation on pesticides is fairly complicated. The main rules concerning approval of products are set out in Chapter 7 of the Chemicals Act, though other rules in the law apply to pesticides too. The most important rule in the law on pesticides is contained in Section 33, subs. 1, which sets out that, prior to their sale, import or use, pesticides must have been approved by the Minister for Environment and Energy. Any infringement of this rule is punishable in accordance with Section 59 of the Act.
In parallel with the Act, the executive order on pesticides is issued, containing a large number of specific directions and stipulations for pesticide manufacturers, importers, dealers and users.
As provided for in the Act, a series of separate executive orders and regulations are also issued, including one on a full or partial ban on the sale and use of certain pesticides, one on the classification, packaging, labelling, sale and storage of chemical substances and products, one on instructing commercial users and one on aerial spraying.
Approval of pesticides
A pesticide may not be imported, sold or used in Denmark unless the product involved has been approved by the Minister for Environment and Energy.
Applications for approval are submitted to DEPA on special forms by the party wishing to import or market a pesticide in Denmark - even where the product involved is identical with one already approved in Denmark (a parallel product). All information needed for an in-depth evaluation of the active ingredient and the product is submitted together with the application forms.
Above all, the approval - which is also called registration - includes a detailed evaluation of the environmental and the health effects of the active ingredient and the product in the area of use specifically being applied for, the choice of crops, the application season, product dosage, application method and so on.
In addition, the product is required to be efficacious for the application specified; it is decided whether using the product poses a risk to bees; treatment time-limits (spraying deadlines) are stipulated so as to eliminate the risk of product residues in foods, animal products (meat, eggs and milk) and feed, and the contents of the label are finalised.
The establishment of limit values for concentration in food forms part of this approval. Where substances are approved for use, a Maximum Residue Limit MRL is laid down, specifying the maximum acceptable residue concentration of the pesticide or its degradation products or metabolites.
The limit value in food products is based partly on a toxicological evaluation of the health risks presented by intake of the pesticide, specifying an Acceptable Daily Intake (ADI), and partly on the residue concentrations in vegetable food of the pesticide found after use of Good Agricultural Practices GAP. GAP is defined as the nationally authorised applications, which under current conditions are required to efficiently combat harmful organisms.When establishing the MRL the health aspect ADI is combined with the use of the pesticide in question GAP. This is done by combining the ADI with the theoretic maximum daily intake MTDI, calculated by means of cost models, assuming that all crops on which the pesticide can be used contain the maximum acceptable concentration of the pesticide. In order to establish the limit value, the TMDI shall not exceed the ADI. In Denmark, the limit values for food are laid down by the National Veterinary and Food Agency.
Approvals are generally operative for up till ten years for plant protection products and biocides, but if a product is classified as "very toxic" or "toxic" to humans, the approval expires after just five years. If it is wished to keep up the approval, an application to renew the approval must be made at least one year before the existing approval expires.
Targets
The reduction targets for agricultural pesticide consumption can be found in the Minister for the Environment's Pesticide Action Plan II from March 2000. This action plan is based on the work performed by The Pesticides Committee (Bichel-committee) who have evaluated the overall consequences of phasing out agricultural pesticide consumption over a period of ten years. The committee's report was published in March 1999.
The committee estimated that the total average load of pesticides from food and drinking water reached approximately 1% of the ADI. They concluded that it was not possible on the basis of epidemiological data to prove that exposure of the general public, e.g. through food, constitutes a health risk.
Based on the evaluation, the committee suggested that a number of investigations was effected, among others related to the potential health impact of pesticides.
Example 3
Contribution margin by different pesticide uses.
The purpose of including the less restrictive scenarios in the
analysis is to clarify the relation between pesticide use, production and economy, during
different treatment intensity, in order to get a better picture of the consequences of
different levels of phasing out the use of pesticides in agriculture. The analyses are
made at operational level only. The agronomical premises were specified on the basis of
both technical/biological research and managerial economy research. As is the case with
the key scenarios, the analyses are based on the operational model (DØP). |
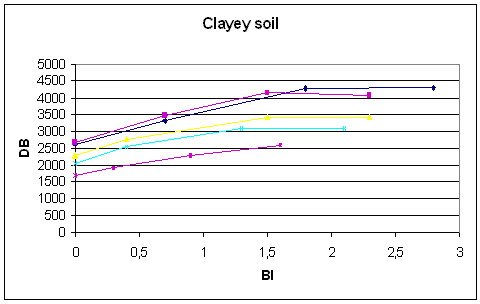
Figure 10.2
The contribution margin by different pesticide uses (treatment frequency) clayey soil.
Source: Ørum (1999)
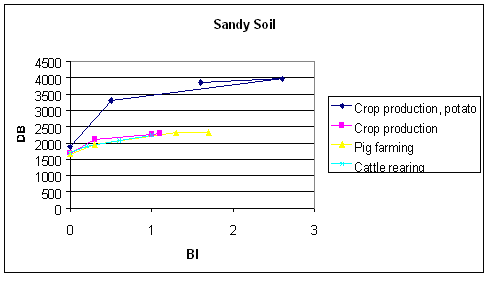
Figure 10.3
Contribution margin by different pesticide uses (treatment frequency) sandy soil94 Source: Ørum (1999)
Biocidal products
As mentioned above, rules pertaining to biocides (disinfectants, preservatives, wood preservatives, rodenticides etc.) have been established through the new Biocidal Products Directive (EC, 1998). The purpose of the directive is to ensure that all chemical and microbiological products intended to kill biological life are approved before use. Product categories like medicinal products, cosmetic products, veterinary medicinal products, and plant protection products, which already fall under the scope of an approval scheme, are exempted from the scope of the Biocidal Products Directive.
Many biocides are not particularly specific, for which reason there is a great risk that they can affect living organisms other than those they are intended to target. Moreover, the use of many biocides is widespread, which means that a large part of the population uses them or is in contact with them daily. Biocides are also directly and indirectly discharged into the environment. On the European market, this relates to products that contain any of a total of about 1,000 active ingredients and which shall be subjected to a restrictive approval procedure. Only a portion of the biocides has been covered by the previous Danish scheme for the approval of pesticides.
The Biocidal Products Directive has now been implemented in Danish law through the Act. no. 256 of 12 April 2000 amending the Act on Chemical Substances and Products and the Statutory Order on Pesticides, latest amended on 5 May 2000 through Order no. 313. A transitory period has been introduced for biocides in the amended legislation. The active substances are divided into 4 main categories covering 23 product types in total. The directive distinguishes between common EU-decisions on active substance level and national decisions on preparations. The substitution principle also apply to biocides and thus an active ingredient can not be included in the annex of approved ingredients if a better alternative exists.
International co-operation
Part of DEPA's strategy aims at strengthening international co-operation in the field of chemicals. Denmark already cooperates on several levels, the Nordic level under the auspices of the Nordic Council of Ministers, the EU level and globally under the auspices of the UN.
Conventions
Transboundary pollution by dangerous chemicals and the increased international trade in chemical substances and products mean that there is a need for an effective effort, to minimise the risk to the environment and health at the global level. Three important conventions in this area are the POP-convention, the PIC-procedure and the Montreal-convention.
POP-convention
In 1997, UNEP Governing Council asked UNEP to establish an International Negotiating Committee (INC) in order to prepare a legally binding instrument with the aim to reduce and/or eliminate the environmental problems caused by Persistent Organic Pollutants (POP). The aim of the POP Convention is to prohibit the production and use (and possibly import and export) of 10 identified POP substances. In addition, the POP Convention aims to prevent, reduce and as far as possible eliminates emissions of 2 identified POP by-products. The 12 substances are all persistent in the environment, they are transported over long distances and far away from the sources and they bioaccumulate in the majority of living organisms.
Denmark has given the POP negotiations high priority. It has been important for Denmark to signal that the agreement of a global POP Convention is necessary in order to minimise and eliminate the environmental effects from the 12 hazardous chemicals. There are numerous important articles in the draft POP Convention as it stands. The prohibition of production and use of POP pesticides as well as the continuing minimisation and ultimate elimination of emissions of dioxins and furans are examples of the most important obligations.
The signature of the POP Convention is expected to take place at a diplomatic conference in Stockholm in May 2001.
PIC-procedure (Prior Informed Concent)
PIC is a procedure that helps participating countries learn more about the characteristics of certain hazardous chemicals before they are shipped to them. The PIC initiates a decision making process on the future import of these chemicals by the importing countries themselves and facilitates the dissemination of this decision to other countries. The aim is to promote a shared responsibility between exporting and importing countries in protecting human health and the environment from the harmful effects of certain hazardous chemicals which are traded internationally.
The PIC-procedure currently includes 24 pesticides and 5 industrial chemicals that are banned or severely restricted in a number of countries and which should not be exported without the consent of importing countries. The aim of the procedure is not to ban or restrict the chemicals on the list, but to alert the importing countries to the dangers of the chemicals. Previously, the PIC procedure was voluntary, but in September 1998 it was turned into a legally binding procedure, i.e. the Rotterdam Convention.
Montreal-convention: Ozone depleting substances
Denmark has actively participated in the international work on substances that deplete the ozone layer. In 1997, the Montreal Protocol on Substances that Deplete the Ozone Layer was adopted. Governments recognised the need for measures to reduce the production and consumption of a number of ozone depleting substances. The Protocol was designed so that the phase-out schedules could be revised, which actually has taken place 5 times since 1987.
The results achieved so far indicate a success story for international ozone initiatives. The used amount of ozone depleting substances has decreased dramatically and agreements have been reached to phase out all ozone depleting substances. The Montreal Protocol is now seen as a model for other international environmental treaties.
On the national arena, Denmark prepared in 1988 an action plan on reduction of the consumption of ozone depleting substance. The plan provided for a combination of instruments, including bans and economic instruments such as taxes and a development programme. Part of the plan was also to phase out the ozone depleting substances as soon as possible and the result has been, that Denmark has stopped the use of the substances quicker than other parties to the Montreal Protocol. These experiences have been used on the international arena under the Protocol in order to strengthen the phase out dates both in the EU and the UN.
TBT-convention (International convention on harmful antifouling systems)
Since 1998 a convention on harmful antifouling systems has been negotiated under the auspices of the United Nations, i.e. in the Environmental Protection Committee (MEPC) under the International Maritime Organisation in London.
The aim of the convention is to reduce or eliminate adverse effects on the marine environment and human health caused by antifouling systems. e.g. chemicals with a biocidal effect. Antifouling systems are used on ships to prevent a wide variety of sea organisms to stick to the ships' hull and thus negatively impacting the ship.
The first antifouling system that will be banned according to the convention are all the organotin compounds, of which TBT is the most well-known. The negotiations in the MEPC are centred on two years: 2003 and 2008. By 2003 is shall no longer be allowed to paint the ship with paint that contains TBT, whereas from 2008 the TBT ban shall be final, i.e. TBT may no longer be part of the ship’s active anti-fouling system, not even if the TBT paint was applied to the ship prior to 2003.
Denmark strongly supports the phasing out of the organotin compounds. However, Denmark is also stressing that adding more systems/substances to the negative list is a very important part of the convention. Should another antifouling system than the TBT-based paints turn out to cause unacceptable adverse effects, the rules and procedures for how to achieve global agreement on the banning or restriction of that antifouling system will already be in place, thus allowing authorities to take action.
The convention is expected to be signed at a diplomatic conference in London in October 2001.
Reporting to the Danish Register of Substances and Products
The product register, which was established as a joint register for DEPA and the National Working Environment Authority (NWEA), contains information on the composition of, and other information on, a number of chemical products that contain dangerous chemical substances, most of which are used commercially. The substances and products which must be registered are those which are used industrially and fall under the concept of danger applied by the NWEA. This concept is somewhat broader than that of DEPA, which only cover classified substances and preparations. NWEA's concept also includes volatile organic solvents, epoxy and isocyanates and substances considered carcinogenic by NWEA. Thus, a number of chemical products are not covered by the rules on reporting to the Register. This applies, for instance, to certain detergents and cleaning products, together with certain paint and varnish products, which are not subject to classification or which are sold only for private use. Cosmetic products shall, however, be reported to DEPA, although such reports do not include information on the component substances of these products.
10.3.5 Instruments
DEPA uses a variety of policy instruments to implement the chemical policy through influencing people’s behaviour. Most of these instruments are elements in DEPA's product-oriented environmental strategy. This strategy focuses on improvement from the standpoint of the health and environment of products (goods), considered over the entire life-cycle of such products and, therefore, on reducing the use of chemical substances in the manufacturing and use of the products.
Integrative instruments
Green guidelines for purchasing
DEPA supports the development of a market for greener products by furthe-ring an increased and more qualified demand for cleaner products. The demand for greener products may come from targeted information to consumers and professional purchasers.
DEPA has issued a number of guidelines for a whole range of products and services including e.g. clothes, furniture, office equipment, buildings and transport. The purpose is primarily to support public purchasers in buying the most environmentally friendly products and services. The documentation, which forms the background for the guidelines, does however also contain an evaluation of the health impact of the products and services.
Regulatory instruments
Action plans
DEPA has issued a number of action plans related to selected chemicals aiming at reducing or phasing out the substances to health and environment both in Denmark and internationally. Such action plans have been prepared for e.g.:
| Brominated flame retardants | |
| Phthalates in flexible PVC | |
| Pesticides | |
| Ozone depleting substances | |
| Heavy metals |
List of undesirable substances
In 2000, DEPA issued an updated "List of Undesirable Substances". The list of about 100 substances, or groups of substances, is a signal to manufacturers, buyers, product developers, etc., that they should consider substitution. The list gives special priority to 26 substances, which the authorities wish to see restricted or completely banned. The special priority assigned to these sub-stances is an advance warning of where DEPA will concentrate its efforts. The List of Undesirable Substances forms the foundation for a number of initiatives over the coming years. These initiatives include prohibition or restriction, phasing out, EU risk assessment, classification, national surveillance programmes and scientific studies.
Today, a sufficient body of knowledge already exists on some of the listed substances to enable the start of restriction initiatives, whereas additional knowledge is needed on other listed substances before any restrictions can be implemented. For the remaining substances, initiatives have been taken in the EU to conduct risk assessments and to implement any consequent restrictions on use.
The new version of the List of Undesirable Substances issued in 2000, was revised against the background of updated information on consumption quantities stored in the Product Register. In the new list, potential candidates from the List of Effects95 are included. Among the health-based selection criteria for the list of undesirable substances are classification for health effects (high acute or chronic toxicity, CMR effects or allergy), specific concern in relation to the use of the substance, and substances targeted for phasing out. Other criteria include environmental effects, also calculated using QSAR modelling, consumption on the Danish market of more than 100 tonnes, and substances which are problematic in the aquatic environment, in waste disposal and in the ground water.
Some of the high priority substances for phase out initiatives or a targeted effort to increase the knowledge base are:
| alkylphenols | |
| alkylphenol ethoxylates (miscellaneous applications) | |
| azo dyes, selected | |
| chromium compounds | |
| dichloromethane | |
| formaldehyde | |
| copper compounds | |
| organotin compounds |
Voluntary agreements
Voluntary agreements between authorities and chemical producers/importers have come into use in recent years, especially in the 1990s. Such agreements have been initiated in relation to e.g. the so-called VOC-chemicals, i.e. volatile chemical hydrocarbons. These hydrocarbons are regularly found as air pollutants evaporated from motor fuels during transportation, relocation or handling. The use of nonylphenol ethoxylates in cleaning agents is another example of a voluntary agreement between DEPA and the Trade.
Organisation for Soap, Perfume and Toiletries entered in 1987. A third example is the agreement regarding nonylphenols in pesticide formulations. This agreement entered into force in 1995 and the phase out was finalised in 2000. More than 100 products containing nonylphenols and nonylphenol ethoxylates have been phased out.
Economic instruments
Taxes
Denmark has introduced taxes on a number of chemicals. These include:
| Pesticides | |
| Lead batteries and sealed nickel-cadmium batteries | |
| Chlorinated solvents | |
| Nitrogen in fertilisers | |
| PVC and phthalates |
These taxes are meant to influence the behaviour of enterprises and of individuals. The purpose is to promote substitution and to reduce the use of chemicals and thereby their occurrence in the different media, soil, water and air and possible exposure on humans.
Subsidies
DEPA has launched a 'Programme for cleaner products' in 1999, which is a subsidised programme with the purpose of strengthening the development and sale of cleaner products. Projects, which are supported must explicitly aim at preventing the environmental impact from all phases in the life cycle of the products or the services. Many of the subsidised projects also focus on the health impact of the products and services.
Information and other instruments
The public sector is under special obligation to lead the way in areas that the government wishes to promote. Increased stable demand for less environmentally degrading goods and services is considered an essential incentive for manufacturers and importers to market cleaner products. Focus in the purchase guidelines is on environmental aspects, but also general health aspects and health in relation to the working environment is taken into consideration in the background information.
Eco-label
Eco-labels are important instruments designed to give the public necessary information for making an environmentally friendly choice. The Nordic swan and the EU-flower are the two official eco-labels for chemical preparations and goods in Denmark. There are no other officially accepted eco-labels in Denmark. The eco-labels are regarded as a tool to minimise the environ-mental impact from products, to be used in connection with the reduction of consumption as part of the Cleaner Products strategy. The Eco-labels do not specifically focus on health aspects, but they do include criteria related to the protection of health. As an example the EU flower can not be assigned to substances or preparations which are classified as very toxic, toxic, carcinogenic, toxic to reproduction, or mutagenic or to goods which have been manufactured by processes, which can cause considerable harm to man or to the consumer in general.
Fact sheets
DEPA has started to prepare easily-understandable information on statutory orders issued under the Chemical Substances and Products Act. This material is entitled "Fakta om kemi" ("Facts on Chemistry", only in Danish) and the target groups are consumers and retailers, together with minor importers and manufacturers. "Facts on Chemistry" are distributed by DEPA and are also available on DEPA's home page (www.mst.dk).
Other information material
In addition to the fact sheets, DEPA publishes different types of information material for consumers, industry and the scientific sphere explaining regulatory requirements, giving advice or adding to the general level of knowledge. This material includes leaflets, brochures, magazines, guidelines and scientific reports.
On the DEPA homepage, a large amount of information about chemicals is available. This includes factual information about chemicals, articles and goods, as well as legislation and scientific reports from DEPA.
It is also possible to contact DEPA and ask specific questions about chemicals using the "chemistry phone" which is administered by The Chemical Inspection Service (CIS).
10.4 Actors
The administrative responsibility for chemicals and chemical pollution rests with several different ministries. These include the Ministry of Environment and Energy, the Ministry of Food, Agriculture and Fisheries, and the Ministry of Health and the Ministry of Labour. Other less directly involved ministries are the Ministry of Housing and Urban Affairs, the Ministry of Social Affairs and the Ministry of Education and Research. Furthermore, the regional councils also play an important role in protecting the environment against the effects of chemical substances and products.
Actors involved directly in the administration of DEPA' s regulation and efforts in the chemical field are listed in the following table.
Table 10.2
Actors, roles and responsibilities concerning chemical regulation
Actors |
Roles and responsibilities concerning chemical regulation |
The Danish Environmental Protection Agency (DEPA) |
The administration of the Chemical Substances and Products Act is centralised in DEPA and very little authority in this area is delegated to the local authorities. The reason for this is that the area demand high specialisation. DEPA administers the act and is responsible for negotiations at the European and international level. |
Chemical Inspection Service |
The Chemical Inspection Service (CIS) of the DEPA carries out supervision to ensure compliance with the rules and rulings in pursuance of legislation on chemicals. This effort focuses particularly on areas of essential importance to health and the environment, areas where requirements differ from the rest of the EU, and imports from non-member countries. Violations of the Chemicals Act and the majority of the statutory orders are punishable by fines, simple detention or up to two years' imprisonment unless more severe punishment is due in accordance with other legislation. |
The Danish Veterinary and Food Administration |
Institute for food safety and toxicology assists DEPA in the area of research, risk assessment and development of health based criteria applied in the regulations. |
The medical officer on health |
The medical officers on health play a very central role locally concerning health aspects of chemicals where they advise counties and municipalities. They are in close contact with the central health and environmental authorities. |
Counties |
- |
Municipalities |
- |
Local Councils |
The local councils help DEPA to enforce retailer compliance with the labelling and storage rules. |
The National Working Environment Authority |
In its capacity of workplace supervisor, the Working Environment Authority reports any violations of the labelling and storage rules to DEPA.. |
10.5 Evaluation
One of the main objectives of chemical regulation in Denmark is to prevent health hazards caused by the use of chemicals in society. A second objective is to promote the use of less health hazardous chemicals through application of cleaner technologies and various instruments restricting the use of certain chemicals or supporting the choice of more safe chemicals for both professional use and for consumers.
Chemical substances form part of daily life in modern societies, also in Denmark. They are used in agricultural and industrial production, and in private households. Knowledge about to what extent the use of chemicals affect public health is relatively sparse, both in relation to individual substances and in relation to the combined pressure from different sources. In some areas, a certain level of knowledge is available, e.g. from reporting of acute toxicity cases to emergency sections at hospitals and the poison control centre. Workplace exposure and related effects is another area contributing to the general knowledge about the possible impact on private users, especially when it comes to acute effects, but also in relation to e.g. development of eczema and other allergic diseases.
Knowledge about the effects of long-term exposure and chronic effects in general is however very limited, as it is extremely difficult to relate the use of various chemicals to the incidence of cancer, reproductive toxicity, allergy and other chronic effects and because little is known about the actual level of exposure to chemicals. A vast number of confounding factors are involved, including factors mainly attributed to the life-style of people like smoking and eating habits, which may add to the same adverse effects.
As the regulation of chemicals is based on the existing level of knowledge, a major concern in relation to industrial chemicals is the large number of substances for which there is insufficient information with regard to hazardous properties. Substances which are not fully investigated and yet on the market may appear as not problematic. Only for chemical substances and preparations, which are covered by an approval procedure before they are marketed, it is ensured that the substances are properly investigated. In addition the regulation on classification on labelling only considers the hazardous properties of the chemicals which means that possible critical uses of the chemical are only regulated and limited when specific use restrictions have been introduced. Another aspect, which needs to be further addressed in the regulations, is the protection of vulnerable groups. In this respect, especially exposure to pregnant women (and thereby exposure to the unborn child) and children should be considered as these groups may either be more susceptible towards chemical exposure or in some cases more exposed. Today, we know that especially the foetus undergoes different periods of development in which susceptibility for adverse effects from exposure to specific chemicals may be increased.
The quantities of chemicals in use are growing violently - solving some problems, but also causing adverse effects relating to health and environment. Consumers and interest groups are making increased demands with regard to the safety of products, which are marketed. At the same time it is expected that the authorities have the market under control and are able to react quickly and efficiently with new regulation in order to guarantee a high level of protection of the public.
A major challenge in chemical regulation is therefore the procurement of information about the many substances, which are not sufficiently investi-gated, and the identification of the problematic substances. In this respect, DEPA supports the work using computerised QSAR-modelling for predicting inherent properties for substances, which have not been adequately tested. Unwanted effects like endocrine disruption also need to be more directly addressed in the regulation, as it is suggested that exposures of vulnerable groups to tiny amounts of endocrine disrupting chemicals may result in serious adverse effects especially in relation to immunotoxic, neurotoxic, reprotoxic or carcinogenic endpoints. In addition there is a need for an effort in relation to exposure assessment in specific fields of application resulting in high exposure of the public or considerable spreading of the substance.
According to the Danish Government there is also a need for a more comprehensive revision of the European chemical regulation, which forms the background for most of the Danish regulation, among other things to speed up the process with regard to risk assessment and identification of problematic substances. Furthermore, the reversion of proof, is an important subject that may promote the protection of the population. Today the chemicals are often considered as harmless unless there are data showing the opposite. In future the goal is to make the industry responsible for presenting data to document the safety of the chemicals.
In order to achieve a higher level of protection of human health, a number of areas have been identified which should be subject to intensified efforts and also generation of more knowledge. The following areas may serve as examples of the goals of such intensified efforts:
| To achieve a higher level of protection of the population through prevention of use situations resulting in high exposure. | |
| To increase the protection of vulnerable groups (e.g. children) through limitation of their exposure to chemical substances and to regulate the use of substances which are particularly problematic to the health. | |
| To reduce to the unintentional spreading of chemicals in products and in the environment in order to avoid diffuse exposure routes. | |
| To protect the health by demanding that the industry documents the safety of chemical substances in use. | |
| To increase the level of protection through a reduction in the number of non-assessed chemical substances using QSAR-evaluation of these chemicals, both for regulatory purposes and screening purposes in relation to further testing. Furthermore to use group evaluation of substances to a greater extent instead of only assessing the individual substances. | |
| To prevent the use and spreading of endocrine disrupters. |
10.6 References
Literature
British Journal of Dermatology (2000).
COM (1999): "Communication from the Commission to the Council and the European Parliament: Community strategy for Endocrine disrupters - a range of substances suspected of interfering with the hormone systems of humans and wildlife". Brussels, 17 December 1999.
Danish National Consumer Agency (DNCA): "Regulation no. 329 of May 23 1995". Regulation on safety requirements for toys and products which due to their outward appearance may be mistaken for food.
DEPA (2000): "More about chemicals", Danish Environmental Protection Agency 27/03/00, DEPA Homepage.
DEPA (2000): "Listen over uønskede stoffer". (List of undesirable substances). Orientering fra Miljøstyrelsen Nr. 9, 2000.
DEPA (1999): "A strategy for intensified efforts in the field of chemicals in Denmark, in the EU and globally." January 1999, DEPA Homepage.
DEPA (1999): "EU, Danmark og miljøreglerne". (EU, Denmark and environmental regulation). Arbejdsrapport nr. 7, 1999.
DEPA (1996): "Chemicals - Status and Perspectives. Excerpts from a discussion paper from DEPA, English Summary and List of Undesirable Substances".
European Chemical Bureau.(2000): IUCLID CD-ROM, Year 2000 Edition, Public data on high volume chemicals. EUR 19559 EN. European Commission Joint Research Centre
http://www/agrsci.dk Exposure model
Ministry of the Environment and Energy (1995): "Miljø- og naturpolitisk redegørelse 1995" .Chapter 10 (Political statement on the Environment and Nature). ISBN 87-601-4898-5.
Ministry of the Environment and Energy, Ministry of Health, Ministry of Food, Agriculture and Fisheries, November 1999: "Protection of children and pregnant women against dangerous substances".
National Institute of Occupational Health (2000): "Kemikalier og produkter i arbejdsmiljøet" - Kap. 2: "Klassificering og mærkning". (Chemicals and products in the working environment. Chapter 2: Classification and labelling).
Nielsen, E. et al, 2000 (draft): "Children and the unborn child: exposure and susceptibility to chemical substances - an evaluation". The Institute for Food
Safety and Toxicology, Danish Veterinary and Food administration.
Danish legislation
Act on Chemical Substances and Products, cf. Consolidation Act no. 21 of 16 January 1996 and subsequent amendments.
Statutory order no. 1065 of 30 November 2000 on classification, packaging, labelling, sale and marketing of chemical substances and products.
Statutory order no. 1012 of 13 November 2000 on prohibition of import and marketing of products containing lead.
Statutory order no. 594 of 6 June 2000 from the Ministry of Environment and Energy on cosmetic products.
Statutory order no. 733 of 31 July 2000 on the list of dangerous substances, with amendments.
Statutory order no. 24 of 14 January 2000 from the Ministry of Environment and Energy on prohibition of import and sale of certain nickel-containing products.
Statutory order no. 151 of 15 March 1999 from the Ministry of Environment and Energy banning phthalates in toys for children aged 0-3 and in certain childcare articles etc.
Statutory order no. 241 of 27 April 1998 from the Ministry of Environment and Energy on pesticides, and subsequent amendments.
Statutory order no. 35 of 19 January 1998 from the Ministry of Environment and Energy on the Community and the Nordic eco-labels.
Statutory order no. 1042 of 17 December 1997 on restrictions on the marketing and use of certain dangerous substances and preparations for specific purposes, and subsequent amendments.
Statutory order no. 1002 of 14 December 1995 from the Ministry of Environment and Energy on notification of new chemical substances.
EU legislation
See Appendix 6.
| 84 | DEPA (1996) Chemicals – Status and Perspectives. Excerpts from a discussion paper from Danish EPA, English Summary and List of Undesirable Substances. |
| 85 | DEPA (1996) Chemicals - Status and Perspectives. Excerpts from a discussion paper from DEPA, English Summary and List of Undesirable Substances. |
| 86 | DEPA (2000) "More about Chemicals", DEPA homepage. |
| 87 | The endocrine system consists of a set of glands and the hormones they produce. Endocrine disrupters are defined as follows according to an international agreement (COM, 1999): a) A potential endocrine disrupter is an exogenous substance or mixture that possesses properties that might be expected to lead to endocrine disruption in an intact organism, or its progeny, or (sub)populations. b) An endocrine disrupter is an exogenous substance or mixture that alters function(s) of the endocrine system and consequently causes adverse health effects in an intact organism, or its progeny, or (sub)populations. |
| 88 | S23: Do not breathe vapours / spray. |
| 89 | S38: In case of insufficient ventilation wear suitable respiratory equipment. |
| 90 | S51: Use only in well-ventilated areas. |
| 91 | EINECS: (European Inventory of Existing Commercial Chemical Substances). EINECS is a static list prepared and published by the Commission and includes 100,116 substances registered on the market in the European Community between 1 January 1971 and 18 September 1981. |
| 92 | Published in OJ 1980 L 246 p.1. |
| 93 | The decision-making process in relation to the ‘new method’ involves very technical discussions in working groups and implementation of the regulations as a Commission Directive. |
| 94 | The unusual course of the crop production and the potato curves indicates that small changes in the composition of the production may cause considerable effects on pesticide treatment. |
| 95 | The List of Effects is the gross list of about 1,000 substances, which the DEPA has identified while drafting the List of Undesirable Substances. The substances on the List of Effects possess properties that are just as undesirable as those of the substances on the List of Undesirable Substances, although they are only used in smaller quantities. |