Environmental Project No. 1341, 2010.
Identification and assessment of alternatives to selected phthalates
Contents
2 Application of DEHP, DBP and BBP in products and articles
3 Identified alternatives to DEHP, DBP and BBP
- 3.1 Functional mode of external plasticisers
- 3.2 Introduction to plasticiser substance families
- 3.3 Alternative plasticisers and polymers used in toys and childcare articles
- 3.4 Alternatives recommended by plasticiser producers
- 3.5 Alternative plasticisers applied with processing adjustments
- 3.6 Alternatives plasticisers selected for further assessment
- 3.7 Alternative flexible polymers
4 Human health and environmental assessment of alternative plasticisers
- 4.1 ASE (alkylsulphonic phenylester)
- 4.2 ATBC (acetyl, tri-N-butyl citrate)
- 4.3 COMGHA
- 4.4 DEGD (diethylene glycol dibenzoate)
- 4.5 DGD (dipropylene glycol dibenzoate)
- 4.6 DEHT (di-ethylhexyl-terephthalate)
- 4.7 DINA (diisononyl adipate)
- 4.8 DINCH (di-isononyl- cyclohexane-1,2-dicarboxylate)
- 4.9 GTA (glycerol triacetate)
- 4.10 TXIB (trimethyl pentanyl diisobutyrate)
- 4.11 Summary of human health and environmental assessment of alternative plasticisers
5 Technical and economical assessment of alternative plasticisers
- 5.1 ASE (Alkylsulfonic phenylester)
- 5.2 ATBC (Acetyl tri-n-butyl Citrate)
- 5.3 "COMGHA1" (12-(Acetoxy)-stearic acid, 2,3-bis(acetoxy)propyl ester)
- 5.4 DEGD (diethylene glycol dibenzoate)
- 5.5 DGD (dipropylene glycol dibenzoate)
- 5.6 DEHT (di-ethyl-hexyl-terephthalate)
- 5.7 DINA (diisononyl adipate)
- 5.8 DINCH (di-isononyl-cyclohexane-1,2dicarboxylate)
- 5.9 GTA (glycerol triacetate)
- 5.10 TXIB (trimethyl pentanyl diisobutyrate)
- 5.11 Summary and discussion of technical and economical assessment of alternative plasticisers
6 Assessment of alternative flexible polymers
Annex 1 Companies and organisations contacted
Annex 2 Introduction to plasticiser families
Annex 3 Summary of technical performance parameters
Annex 4 Background data for the environmental and health assessment
Preface
Since spring 2007, three phthalates, DEHP, DBP and BBP, have been prohibited for use in toys and childcare articles. Further three phthalates, DINP, DIDP and DNOP, are prohibited in toys and childcare articles which can be placed in the mouth by children.
On the basis of their classification as toxic to reproduction, DEHP, DBP and BBP have been identified as presenting properties of very high concern, and have been placed on the candidate list for consideration for inclusion in Annex XIV of the REACH Regulation.
Substances included in Annex XIV are subject to authorisation. Authorisation should according to the REACH Regulation be granted where it is demonstrated that the risks to human health and the environment arising from the use of the substance are adequately controlled. Otherwise, uses may still be authorised if it can be shown that the socio-economic benefits from the use of the substance outweigh the risks connected with its use and there are no suitable alternative substances or technologies that are economically and technically viable.
As consequence of the prohibition of the three classified phthalates in toys and childcare articles, alternative substances and alternative materials have been introduced for such products.
In order to establish an overview of the risks to human health and the environment arising from the use of the alternative substances and to evaluate to what extent alternative substances or technologies are economically and technically viable as substitutes for DEHP, DBP and BBP in the different applications, the Danish Environmental Protection Agency has initiated this study.
The results of the study are intended to feed in to the discussion regarding the inclusion of the three phthalates in Annex XIV and the conditions for the authorisation if the substances are included in the Annex.
The objectives of the study are:
- To survey possible alternatives to DEHP, BBP and DBP with particular emphasis on alternatives to restricted phthalates used in toys and childcare articles,
- To assess health and environmental properties of the alternatives;
- To assess the technical and economic feasibility of the use of the alternatives for the main applications of DEHP, BBP and DBP;
- To disseminate information on alternatives and the feasibility of substitution to relevant stakeholders in Denmark.
The study has been guided by a Steering Group consisting of Shima Dobel and Lotte Kau Andersen, Danish Environmental Protection Agency, and Carsten Lassen and Sonja Hagen Mikkelsen, COWI A/S.
This report has been prepared by Carsten Lassen (Project Manager), Ulla Kristine Brandt, Jakob Maag, Jesper Kjølholt, Lise Molander and Sonja Hagen Mikkelsen, COWI A/S, Denmark.
Summary
In this study a number of alternative plasticisers to the phthalates DEHP, DBP and BBP are described and assessed. Suitable alternative plasticisers have been identified for most applications of the phthalates, and 10 of these have been assessed in detail. Some of the assessed alternative plasticisers have a broad application scope, others are more specialised. Plasticiser alternatives to DEHP, first of all the plasticisers DINA, DINCH, DEHT, ATBC and ASE are marketed at somewhat higher to significantly higher prices than the price of DEHP. The results of the assessment further indicate that alternatives to DBP and BBP are available for the major applications of the substances, at prices quite similar to the prices of the phthalates.
All 10 assessed substances are expected to have low acute toxicity based on animal studies. For three of the assessed alternatives data exists demonstrating that they cannot be considered CMR substances (carcinogenic, mutagenic, reprotoxic); for the other alternatives data for at least one critical parameter are missing. The toxicological data for DEGD and DGD, two of the available alternatives to DBP and BBP in polymeric applications (plastics), indicate that the substances might be some effect on reproduction, but the results are not statistically significant and more data are necessary for a clear conclusion. With regard to the environmental properties, none of the 10 studied alternatives meet the criteria for being PBT substances (persistent, bioaccumulative and toxic in the aquatic environment) or vPvB substances (very persistent and very bioaccumulative), although all substances except GTA show one or two of these properties.
Ortho-phthalates are a group of substances which have together proven to be widely applicable for plasticising purposes. Especially the general plasticisers DEHP, DINP and DIDP have had high importance due to their very wide applicability in PVC. DBP and BBP are specialty plasticisers which in polymer applications typically have been used together with other plasticisers in order to obtain specific processing conditions or material properties. The wide range of applications combined with comparatively low prices have made the ortho-phthalates the preferred choice of plasticiser for the PVC industry for many years and phthalates account today for about 90% of the total plasticiser use for PVC in Europe. This percentage has been quite stable for the last ten years only the distribution between the different phthalates has changed. The consumption of DBP and BBP has decreased markedly the last 15 years and the manufactured volume of each of the substances is today approximately 1% of the total manufactured phthalates.
DINP and DIDP have become dominating alternatives to DEHP due to their closeness in performance to DEHP and only moderately higher costs. DINP or DIDP can replace DEHP for practically all applications and the price is approximately 10% higher than the price of the DEHP. DINP and DIDP have not been further assessed in this study because they are already well described and finished EU risk assessments are available for these substances.
Information on alternatives to DEHP, DBP and BBP, actually applied today, has been collected from the following data sources:
- Danish manufacturers and importers of toys and childcare articles;
- Surveys of plasticisers in toys and childcare articles marketed in the Netherlands, Germany, Austria and Switzerland;
- Direct contact to major manufacturers of plasticisers.
These data sources have been supplemented with information from the literature and manufacturers' web sites.
As consequence of the prohibition of DEHP, DBP and BBP in toys and childcare articles and the prohibition of DINP, DIDP and DNOP in toys and childcare articles which can be placed in the mouth by children, experience exist in substitution of the phthalates for these products. Dutch surveys of plasticisers in toys demonstrate that DEHP and DBP a few years ago could be found in a significant percentage of all samples. In accordance with the fact that the major uses of BBP are in flooring and non-polymer applications (e.g. adhesives) BBP was only found in a few of the toy samples and the surveys of plasticisers used in toys would not give any indication of which plasticisers are applied as alternatives to BBP.
Table 0.1 lists plasticisers used in toys and childcare products. The substances reported by Danish manufacturers have specifically been used by the manufacturers as substitutes for phthalates, whereas some of the plasticisers reported in the surveys of plasticisers in toys and childcare products from other EU countries may in fact not have substituted a former use of phthalates.
Three of non-phthalate plasticisers were found in a significant percentage of the samples in both surveys and are reported by all responding Danish manufacturers of toys as used as alternatives to phthalates: DINCH, DEHT and ATBC. All three are marketed as general plasticiser alternatives to DEHP. Among the non-phthalate plasticisers there seems not to be one substance that can make a one-to-one substitution for all applications of each phthalate. Which substitutes are suitable depends on the actual processing conditions and the desired properties of the final product. Finding the right plasticiser for a given application is often a complex process, as many technical criteria have to be met simultaneously. Comprehensive testing of the performance of the polymer/plasticiser system is often required. By way of example one Danish manufacturer reports that the development led to the use of a mixture of ATBC, DINCH and DEHT, which could be blended in a variety of combinations to achieve softened PVC that performed to the required standards with the existing production setup.
On request a number of manufacturers of plasticisers have provided information on their market experience with possible alternatives to DEHP, BBP and DBP. Unfortunately the manufacturers of DINCH and DEHT, widely used in toys, did not provide detailed information, and the assessment of these substances has mainly been based on information from the manufacturers' websites and the literature. Both substances are marketed as general plasticisers for PVC.
Based on information on the plasticisers found in toys and childcare articles and initial information from manufacturers, a gross list of 25 potential non-phthalate alternatives was compiled and from this list 10 plasticisers were selected for further assessment.
Benzoates (represented in Table 0.2 by DGD and "Mix of DGD, DEGD, TGD") have been the main alternative to BBP, because they provide both suitable processing conditions and properties of the final products and have quite similar price. BBP is mentioned as a critical component in seals for insulating double glazing and it cannot be ruled out that niche applications of BBP and DBP exist where alternatives are not suitable.
Click here to see: Table 0.1 Plasticisers found in toys and childcare articles
Experience in the use of alternative plasticiser exists for all major applications of DEHP, DBP, and BBP. Experience for 6 of the selected plasticisers, for which detailed information was obtained is shown in Table 0.2. Note that the alternatives typically do not substitute for the phthalates 1:1; often are more substances used in combination.
| Application | Market experience (1 to 4) *1 | |||||
|---|---|---|---|---|---|---|
| ASE | GTA | DGD | Mix of DGD, DEGD, TGD | ATBC | COMGHA | |
| As substitute for DEHP | ||||||
| Polymer applications: | ||||||
| Calendering of film, sheet and coated products | 2 | 2 | 4 | 4 | 3 | 3 |
| Calendering of flooring, roofing, wall covering | 4 | 2 | 3 | 3 | 3 | |
| Extrusion of hose and profile | 2 | 2 | 3 | 3 | 3 | 3 |
| Extrusion of wire and cable | 2 | 2 | 3 | 3 | 3 | |
| Extrusion of miscellaneous products | 2 | 2 | 2 | 2 | 2 | 3 |
| Injection moulding | ? | 2 | 2 | 2 | 3 | |
| Spread coating of flooring | 2 | 2 | 2 | 2 | 2 | |
| Spread coating | 2 | 2 | 2 | 2 | 3 | |
| Car undercoating | 2 | 3 | 3 | |||
| PVC medical articles | 2 | 2 | ||||
| Toy and childcare articles | 2 | 1 | ||||
| Non polymer applications: | 0 | |||||
| Adhesives/sealant, rubber | 2 | 2 | 1 | 1 | 2 | 4 |
| Lacquers and paint | 2 | 2 | 2 | 2 | 4 | |
| Printing ink | 2 | 2 | 2 | 2 | 2 | 3 |
| Production of ceramics | ||||||
| As substitute for DBP | ||||||
| Plasticiser in PVC | 2 | 1 | 1 | 2 | 2 | |
| Plasticiser in other polymers | 2 | 2 | ||||
| Adhesives | 2 | 2 | 1 | 3 | 4 | |
| Printing inks | 2 | 3 | 2 | 3 | ||
| Miscellaneous: | ||||||
| Sealants | 2 | 3 | 4 | |||
| PU foam sealants | 2 | 4 | ||||
| Nitrocellulose paints | 2 | 3 | 2 | 2 | 2 | |
| Film coatings | 3 | 3 | ||||
| Glass fibre production | 4 | |||||
| Cosmetics | 2 | |||||
| As substitute for BBP | ||||||
| Polymer applications: | ||||||
| General PVC (e.g. for moulded plastic parts) | 2 | 4 | ||||
| Plastisol coating, for flooring | 2 | 1 | 1 | 3 | ||
| Extrusion or spread coating | 2 | 2 | 2 | |||
| Films, calendering | 2 | 4 | 4 | 3 | ||
| Non polymer applications: | ||||||
| Sealants | 2 | 1 | 1 | |||
| Coatings and inks) | 2 | 1 | 3 | |||
| Adhesives | 2 | 1 | ||||
| Nail polish | 1 | |||||
*1: Market experience categories interpretation: 1) Main alternative on market. 2) Significant market experience. 3) Examples of full scale experience. 4) Pilot/lab scale experience
The non-phthalate alternatives to DBP and DEHP have until now primarily been used for particular applications. DBP is in polymers typically used in relatively low concentration in combination with DEHP and the whole plasticiser system is replaced by other plasticiser systems.
A summary of the technical suitability of the 10 alternatives and their prices relative to DEHP, DBP and BBP is provided in Table 0.3.
| Substance | Overall technical assessment | Price relative to DEHP *1 |
|---|---|---|
| ASE | ASE is a general plasticiser alternative to DEHP. The producer has indicated significant market experience for most traditional DEHP, DBP and BBP uses. | + |
| ATBC | The performance of ATBC on some parameters seems similar to DEHP, indicating technical suitability for substitution of DEHP for some applications. The higher extractability in aqueous solutions and the higher volatility may reduce the performance of ATBC as a plasticiser in PVC. The data available does not allow a closer assessment of ATBC's technical suitability as alternative to DEHP, DBP and BBP | ++ |
| Benxoflex 2088 (with DEGD) | The producer has indicated significant market experience in several of the traditional DBP and BBP specialty plasticiser applications and certain DEHP applications, notably in the non-polymer (adhesives, sealants, etc.) and PVC spread coating (plastisol) application fields. According to the producer, Benzoflex 2088 (with DEGD) has become the main non-phthalate alternative to DBP or BBP in vinyl flooring production in Europe. The higher extractability in water may limit its use for some applications. | ≈ |
| COMGHA | According to the producer, COMGHA still has relative moderate market experience, albeit with many examples of full scale usage and pilot/lab scale tests, and significant market experience in some plastisol application and cosmetics. The producer found good performance on key technical parameters indicating a potential for substituting for DEHP and perhaps for DBP and BBP in some traditional uses of these substances. | ++ |
| DEHT | DEHT is a general plasticiser alternative to DEHP. Today, terephthalates like DEHT are more commonly used in the USA than elsewhere. | ≈ |
| DINA | DINA has mostly been used for low temperature PVC applications and in PVC film/wrapping . The data available for this study does not allow clear-cut conclusions as regards DINA's suitability as alternative to DEHP | + |
| DINCH | The producer’s sales appraisal indicates a relatively wide usage of DINCH for general plasticiser purposes. DINCH was the most frequently found plasticiser in two European surveys of plasticisers in toys and childcare articles. The data available does not allow a closer assessment of DINCH's technical suitability as alternative to DEHP, DBP and BBP. | + |
| DGD | The fact that DGD for many years has been a well known and much used competitor to BBP, especially in PVC flooring and in PVA adhesives, indicates a clear potential for substituting DGD for BBP, from a technical point of view. DGD may probably also substitute for some traditional uses of DEHP and DBP. | ≈ |
| GTA | According to a producer, GTA can substitute for DBP and BBP in adhesives, inks and coatings. The data available does not allow a closer assessment of GTA's technical suitability as alternative to DEHP, DBP and BBP. | + |
| TXIB | TXIB was found in more than 10% of the samples in surveys of plasticisers in toys and childcare articles. However, the producer does not consider TXIB an alternative to DEHP, DBP or BBP, and the usage of TXIB in vinyl flooring has declined in the 1990’s due to high emissions from end products. Consequently, TXIB seems not to be a suitable alternative to DEHP, DBP or BBP. | NA |
Notes: *1: Based on comparison with DEHP, but DBP and BBP are reported to have similar price and the notation therefore serves as indicating price relative to DBP and BBP as well. "≈" means similar price or slightly lower or higher than DEHP; "+" means somewhat higher price (10-50% higher) than DEHP and "++" means significantly higher price than DEHP. The report provides actual price examples.
Price of alternatives
As shown in the table above, the price of the non-phthalate alternatives DEHT, DGD and Benzoflex 2088 were in the same price range as the price of DEHP, DBP and BBP, (as well as the ortho-phthalate alternatives DINP and DIDP), whereas ASE, DINA , DINCH and GTA were somewhat more expensive and ATBC and COMGHA were considerably more expensive. The content of DEHP in plasticised PVC is typically 30% of the plastics and an increase in the price of the plasticiser of e.g. 30% will result in a material price increase of 10% for the plastic material.
Prices of chemicals (and other industrial products) tend to decrease as production capacity and competition is increased. Different chemicals are however based on different raw materials and more or less complex and resource demanding chemical synthesis technologies. This of course sets limits to the minimum prices attainable even in a mature market, and some of the alternative plasticisers described may likely remain at higher price levels. It should be noted that the prices of DEHP have dropped significantly over the last ten year or more; probably due to the reduced demand driven by the regulation.
Besides the price of the plasticisers, the substitution of the phthalates may imply some costs of research and development and process changes which have not been assessed in this study.
Assessment of alternative flexible polymers
A number of flexible polymers are available which can substitute for many traditional uses of flexible PVC. Polyethylene (PE), polyolefin elastomers, different polyurethane (PU) qualities, ethylene vinyl acetate (EVA) and different rubber types are examples among others. For many flexible PVC uses, also other substitute materials than flexible polymers exist. The existing LCA-based, application-focused assessments are few, and often the studies could not make clear-cut conclusions. But many materials exist with seemingly equal or better environmental, health and safety, performance and cost profiles. The assessment made here does not allow for a more detailed analysis of possibilities and limitations in the use of alternative flexible polymers.
Environmental and health assessment
A summary of the inherent properties for the investigated alternative plasticisers is shown in Table 0.4 using key parameters: acute and local effects, sensitisation, carcinogenicity, mutagenicity, reproductive toxicity, persistence, bioaccumulation and aquatic toxicity.
From the overview it can be seen that all ten substances are expected to have low acute toxicity based on animal studies. With regard to local effects most substances are non-irritating to skin and eyes or only produce slight irritation which would not lead to classification. None of the tested substances are sensitising.
Effects from repeated dose toxicity studies mainly include reduced body weight gain, increased organ weights (liver and/or kidney) and for some substance also changes in clinical chemistry or clinical pathology parameters. However, more serious pathological effects were not observed.
Studies to evaluate the potential for reproductive/developmental toxicity primarily show toxic effects on parents and offspring. For TXIB statistically significant reproductive and developmental toxicity is observed.
Click here to see: Table 0.4 Overview of main toxicological and ecotoxicological properties
Carcinogenicity has only been evaluated for three substances in combined studies. For all three substances the outcome was negative (no carcinogenicity effect). However, the studies cannot be considered sufficient to exclude possible carcinogenic effects.
The assessment in this study of the toxic properties of ATCB, COMGHA, DINCH and DEHT is in line with the recent assessment from the Scientific Committee on Emerging and Newly-Identified Health Risks (SCENIHR).
All substances have been tested for acute toxicity for at least one exposure route, sensitisation (except ASE), subchronic toxicity and mutagenicity. All substances except ASE, COMGHA and DINA have been tested for both reproductive and developmental toxicity.
With regard to carcinogenicity only ATBC, DEHT and DINCH have been tested in combined chronic toxicity and carcinogenicity studies. For DEGD, DGD and DEHT estrogenic activity has been tested in a uterotrophic assay without positive response.
Most data used for the evaluation are considered of good quality, i.e. studies following accepted guidelines (OECD or US EPA) or studies considered acceptable at the time they were carried out. For some of the studies little information is available to evaluate the quality. However, key information is obtained from IUCLID data sheets, USEPA or OECD HPV robust summaries.).
With regard to environmental properties, none of the 10 studied alternatives meet the criteria for being a PBT or vPvB substance, although all substances except GTA show one or two of these properties. GTA (triacetin) appears to be easily biodegradable, it does not bioaccumulate and has very moderate toxicity in the aquatic environment.
DEGD, DGD and DINA also come out rather favourable, while ATBC and COMGHA come out negatively despite their degradability because of their aquatic toxicities and bioaccumulative properties. ASE and DINCH both have low acute toxicities to aquatic organisms, but are not easily degradable and have high log KOW values. DEHT is also not easily biodegradable and is bioaccumulative but its aquatic toxicity cannot be fully evaluated based on the data available.
Useful fate data regarding biodegradability (in water) and bioaccumulative properties (either as BCF or log KOW) are available for all alternatives while other fate data are incomplete for some substances. With regard to ecotoxicological effect data, results from short-term tests with the base-set of organisms - fish, crustaceans and algae - exist for all 10 substances although the duration of some studies deviate from the current OECD standard.
Overall, the data indentified are of good quality i.e. they are mostly based on studies performed according to accepted guideline procedures, and the studies have been evaluated to be reliable without restrictions or reliable with restrictions (e.g. in the USEPA HPV robust summaries).
Dansk sammenfatning
I denne undersøgelse er en række blødgørere, som kan anvendes som alternativer til ftalaterne DEHP, DBP and BBP, blevet beskrevet og vurderet. Til de fleste anvendelser af ftalaterne er der blevet fundet brugbare alternative blødgørere og 10 af disse er blevet vurderet i detaljer. Nogle af de vurderede alternativer har et bredt anvendelsesområde mens andre er mere specialiserede. Alternative blødgørere, der kan erstatte DEHP, først og fremmest blødgørerne DINA, DINCH, DEHT, ATBC og ASE markedsføres til priser der varierer fra lidt højere til væsentligt højere end prisen på DEHP. Resultatet af vurderingen viser endvidere at alternativer til DBP og BBP er til rådighed for de fleste af anvendelserne af stofferne til priser, som er meget lig prisen på ftalaterne.
Alle 10 vurderede stoffer forventes, på basis af undersøgelser på dyr, at have en lav akut toksicitet. For tre af de vurderede alternativer er der data, som viser at stofferne ikke kan betragtes at være CMR stoffer (at kunne fremkalde kræft, skader på arveanlæg, eller skader på fostre eller forplantningsevnen). For de øvrige alternativer mangler der data for mindst én af de kritiske parametre. De toksikologiske data for DEGD og DGD, to af de tilgængelige alternativer til DBP og BBP anvendt i polymerer (plastmaterialer) indikerer, at stofferne måske kan have en effekt på forplantningsevnen, men resultaterne er ikke statistisk signifikante, og der er brug for flere data for at kunne lave en klar konklusion. Med hensyn til miljøegenskaberne er der ingen af de 10 undersøgte alternativer der opfylder kriterierne for at være PBT stoffer (persistente, bioakkumulerbare og giftige i vandmiljøet) eller vPvB stoffer (meget persistente og meget bioakkumulerbare), selv om alle stofferne på nær GTA udviser én eller to af disse egenskaber.
Ortho-ftalater er en gruppe af stoffer som sammen har vist sig at have en bred anvendelse som blødgørere. Især har almen-blødgørerne DEHP, DINP and DIDP har haft stor betydning på grund af deres brede anvendelighed i PVC. DBP og BBP er special-blødgørere som i polymeranvendelser typisk har været brugt sammen med andre blødgørere for at opnå bestemte procesbetingelser eller materialeegenskaber. Det brede anvendelsesområde kombineret med forholdsvis lave priser har gjort ftalaterne til det foretrukne valg af blødgørere i PVC industrien, og ftalaterne udgør i dag 90% af det totale forbrug af blødgørere til PVC in Europa. Denne andel har været ret stabil i de eneste 10 år, hvor kun fordelingen mellem de enkelte ftalater har forandret sig. Forbruget af DBP og BBP har faldet markant de seneste 15 år og den producerede mængde udgør i dag - for hvert af stofferne - kun omkring 1% af den totale mængde ftalater, som produceres
DINP og DIDP er blevet de dominerende alternativer til DEHP på grund af deres lighed med DEHP og kun lidt højere pris. DINP og DIDP kan erstatte DEHP til stort set alle anvendelser og prisen er omkring 10% højere end prisen på DEHP. DINP og DIDP er ikke blevet nærmere vurderet i denne undersøgelse, fordi de allerede er velbeskrevne og der er færdiggjorte EU risikovurderinger omhandlende disse stoffer.
Information om alternativer til DEHP, DBP and BBP, som faktisk anvendes i dag, er blevet indsamlet fra følgende datakilder:
- Danske producenter og importører af legetøj og småbørnsartikler;
- Kortlægninger af blødgørere i legetøj og småbørnsartikler markedsført i Holland, Tyskland, Østrig og Schweiz;
- Direkte kontakt til større producenter af blødgørere.
Disse datakilder er blevet suppleret med information fra litteraturen og producenternes internetsteder.
Som følge af forbuddet mod DEHP, DBP og BBP i legetøj og småbørnsartikler og forbuddet mod DINP, DIDP and DNOP i legetøj og småbørnsartikler, der kan puttes i munden, er der erfaring med at erstatte brugen af ftalaterne i disse produkter. Hollandske kortlægninger af blødgørere i legetøj viser, at DEHP og DBP for få år siden kunne findes i en væsentlig del af alle prøver. I overensstemmelse med det faktum at de væsentligste anvendelser af BBP er i gulvbelægninger og ikke-polymer anvendelser (f.eks. lime) blev der kun fundet BBP i få af legetøjsprøverne, og kortlægninger af blødgørere i legetøj kan derfor ikke bruges til at vise, hvilke blødgørere der anvendes som alternativer til BBP.
Tabel 0.1 oplister blødgørere, som anvendes i legetøj og småbørnsartikler. De stoffer, som er oplyst af danske producenter, er specifikt anvendt af producenterne som erstatning for ftalater, hvorimod nogle af de blødgørere, der er blevet fundet i kortlægningerne af blødgørere i legetøj og småbørnsartikler i andre EU lande, måske ikke har erstattet en tidligere anvendelse af ftalater.
Tre af ikke-ftalat blødgørerne blev fundet i en væsentlig del af alle prøver i begge undersøgelser og alle de danske legetøjsproducenter, der har svaret, har oplyst at disse bliver brugt som alternativer til ftalater: DINCH, DEHT og ATBC. All tre markedsføres som almen-blødgører alternativer til DEHP. Blandt ikke-ftalat alternativer synes der ikke at være ét stof, der kan lave en en-til-en erstatning for alle anvendelser af hver af ftalaterne. Det afhænger af procesbetingelser og de ønskede egenskaber af det færdige produkt, hvilket alternativ der er brugbart. At finde den rette blødgører til en given anvendelse er ofte en kompleks proces da der er mange tekniske kriterier, der skal opfyldes på samme tid. Der kræves ofte omfattende tests af de tekniske egenskaber af polymer/blødgører systemet. Eksempelvis oplyser en dansk producent at udviklingen ledte til en kombination af ATBC, DINCH and DEHT, som kunne blandes i forskellige blandingsforhold, for at opnå en blødgjort PVC der levede op til de krævede standarder med brug af det eksisterende produktionsapparat.
På anmodning har et antal producenter af blødgørere leveret information om deres markedserfaringer med mulige alternativer til DEHP, BBP og DBP. Desværre har producenterne af DINCH og DEHP, som har en stor anvendelse i legetøj, ikke leveret detaljeret information, og vurderingen af disse stoffer er derfor overvejende baseret på information fra producenternes Internetsteder. Begge stoffer markedsføres som almen-blødgørere til PVC.
Baseret på informationen om blødgørere fundet i legetøj og småbørnsartikler og indledende oplysninger fra producenter, blev der opstillet en bruttoliste på 25 potentielle ikke-ftalat alternativer, og fra denne liste blev der udvalgt 10 blødgørere til den videre vurdering.
Benzoater (repræsenteret i tabel 0.2 af DGD og "Blanding af DGD, DEGD, TGD") har været de vigtigste alternativer til BBP, fordi de giver tilfredsstillende procesbetingelser og egenskaber af det færdige produkt og har næsten samme pris som BBP. BBP er omtalt som en kritisk komponent i forseglinger til termoglasruder, og det kan ikke udelukkes, at der findes nogle niche-anvendelser, hvor alternativerne ikke er anvendelige.
Klik her for at se: Tabel 0.1 Blødgørere fundet i legetøj og småbørnsartikler
Der findes erfaring i brug af alternativer til alle større anvendelser af DEHP, DBP, og BBP. Erfaring for 6 af de udvalgte blødgørere, for hvilke detaljeret information blev tilvejebragt, er vist i tabel 0.2. Bemærk at alternativerne typisk ikke kan erstatte ftalaterne 1:1, men at de ofte bruges i en kombination af flere stoffer.
| Anvendelse | Markedserfaringer (1 til 4) *1 | |||||
|---|---|---|---|---|---|---|
| ASE | GTA | DGD | Blanding af DGD, DEGD, TGD | ATBC | COMGHA | |
| Som alternativ til DEHP | ||||||
| Anvendelser i polymerer: | ||||||
| Kalendrering af film, plader og overfladebehandlede produkter | 2 | 2 | 4 | 4 | 3 | 3 |
| Kalendrering af gulvbelægning, loft- og vægbeklædnin g | 4 | 2 | 3 | 3 | 3 | |
| Extrudering af slanger og profiler | 2 | 2 | 3 | 3 | 3 | 3 |
| Extrudering af ledninger og kabler | 2 | 2 | 3 | 3 | 3 | |
| Extrudering af diverse produkter | 2 | 2 | 2 | 2 | 2 | 3 |
| Sprøjtestøbning | ? | 2 | 2 | 2 | 3 | |
| Plastisol belægning af gulvbelægning | 2 | 2 | 2 | 2 | 2 | |
| Anden plastisol belægning | 2 | 2 | 2 | 2 | 3 | |
| Undervognsbehandling | 2 | 3 | 3 | |||
| Medicinske artikler af PVC | 2 | 2 | ||||
| Legetøj og småbørnsartikler | 2 | 1 | ||||
| Anvendelser i ikke-polymerer: | 0 | |||||
| Lime/fugemasser, gummi | 2 | 2 | 1 | 1 | 2 | 4 |
| Lak og maling | 2 | 2 | 2 | 2 | 4 | |
| Trykfarver | 2 | 2 | 2 | 2 | 2 | 3 |
| Produktion af keramik | ||||||
| Som alternativ til DBP | ||||||
| Anvendelser i polymerer: | ||||||
| Blødgører i PVC | 2 | 1 | 1 | 2 | 2 | |
| Blødgører i andre polymerer | 2 | 2 | ||||
| Anvendelser i ikke-polymerer: | ||||||
| Lime | 2 | 2 | 1 | 3 | 4 | |
| Trykfarver | 2 | 3 | 2 | 3 | ||
| Fugemasser | 2 | 3 | 4 | |||
| Fugemasser af PU skum | 2 | 4 | ||||
| Nitrocellulose maling | 2 | 3 | 2 | 2 | 2 | |
| Overfladefilm | 3 | 3 | ||||
| Glasfiber produktion | 4 | |||||
| Kosmetik | 2 | |||||
| Som alternativ til BBP | ||||||
| Anvendelser i polymerer: | ||||||
| Generel PVC (f.eks. støbte plastikdele) | 2 | 4 | ||||
| Plastisol overfladebelægning af gulvbelægninger | 2 | 1 | 1 | 3 | ||
| Extrudering eller plastisol overfladebelægning | 2 | 2 | 2 | |||
| Kalendrering af film | 2 | 4 | 4 | 3 | ||
| Anvendelser i ikke-polymerer: | ||||||
| Fugemasser | 2 | 1 | 1 | |||
| Overfladebelægninger og trykfarver | 2 | 1 | 3 | |||
| Lime | 2 | 1 | ||||
| Neglelak | 1 | |||||
Note til tabel på
*1: Forklaring af kategorierne for markedserfaring: 1) Vigtigste alternativ på markedet 2) Væsentlig markedserfaring. 3) Eksempler på erfaring i fuldskala. 4) Erfaring på pilot- eller laboratorieskala.
Ikke-ftalat alternativer til DBP og DEHP har indtil nu primært været brugt til særlige anvendelser. DBP er i polymerer typisk blevet brugt i relativt lav koncentration i kombination med DEHP og hele blødgørersystemet erstattes af andre blødgørersystemer. En sammenfatning af de 10 alternativers tekniske egnethed og deres pris sammenlignet med DEHP, DBP and BBP er vist i Tabel 0.3.
| Stof | Overordnet teknisk vurdering | Pris sammenlignet med DEHP *1 |
|---|---|---|
| ASE | ASE er en almen-blødgører, som kan anvendes som alternativ til DEHP. Producenten har indikeret en væsentlig erfaring i forhold til de fleste traditionelle anvendelser af DEHP, DBP og BBP. | + |
| ATBC | ATBC opfører sig i forhold til nogle parametre ligesom DEHP, hvilket indikerer at stoffet teknisk set egner sig til at erstatte DEHP til nogle anvendelser. Den højere ekstraherbarhed i vandige opløsninger og stoffets større flygtighed begrænser muligvis ATBCs egnethed som blødgører i PVC. De data, der har været til rådighed, tillader ikke en nærmere vurdering af ATBCs tekniske egnethed som alternativ til DEHP, DBP og BBP. | ++ |
| Benxoflex 2088 (med DEGD) | Producenten har indikeret væsentlig markedserfaring inden for flere af de traditionelle anvendelser af DBP og BBP som special-blødgørere og visse anvendelser af DEHP, navnlig i ikke-polymer anvendelser (lime, fugemasser og andet) og PVC plastisol overfladebehandling. I følge producenten er Benzoflex 2088 (med DEGD) det væsentligste ikke-ftalat alternativ til DBP og BBP i produktionen af vinylgulve i Europa. Den højere ekstraherbarhed i vand begrænser muligvis stoffets brug til nogle anvendelser. | ≈ |
| COMGHA | I følge producenten har COMGHA stadig en relativ lille markedserfaring, omend der er mange eksempler på fuldskala-brug og pilot- eller laboratorieskala forsøg og en væsentlig markedserfaring i nogle plastisol anvendelser og i kosmetik. Producenten fandt at stoffet havde gode egenskaber i relation til en række væsentlige tekniske parametre, hvilket indikerer et potentiale for at kunne erstatte DEHP og eventuelt DBP og BBP til nogle af disse blødgøreres traditionelle anvendelser. | ++ |
| DEHT | DEHT er en almen-blødgører, som kan anvendes som alternativ til DEHP. DEHT er for øjeblikket mere almindeligt brugt i USA end andre steder. | ≈ |
| DINA | DINA har mest været brugt til lav-temperatur anvendelser i PVC og i PVC film/indpakning. De data, som har været til rådighed for denne undersøgelse, tillader ikke en klar vurdering af DINAs egnethed som alternativ til DEHP. | + |
| DINCH | Producentens salgsmateriale indikerer en relativt udbredt brug af DINCH som almen-blødgører. DINCH var den mest hyppigt fundne blødgører i to europæiske undersøgelser af blødgørere i legetøj og småbørnsartikler. De data, der har været til rådighed, tillader ikke en nærmere vurdering af DINCHs tekniske egnethed som alternativ til DEHP, DBP og BBP. | + |
| DGD | Det faktum at DGD i mange år har været en velkendt og meget anvendt konkurrent til BBP, især i PVC gulvbelægning of PVA lime, indikerer, at DGD fra en teknisk synsvinkel har et klart potentiale for at kunne erstatte BBP. DGD kan formentlig også erstatte DEHP og DBP til nogle af disse blødgøreres traditionelle anvendelser. | ≈ |
| GTA | I følge en producent kan GTA erstatte DBP og BBP i lime, trykfarver og overfladebehandlinger. De data, der har været til rådighed, tillader ikke en nærmere vurdering af GTAs tekniske egnethed som alternativ til DEHP, DBP og BBP. | + |
| TXIB | TXIB blev fundet i mere end 10% af prøverne i kortlægninger af blødgørere i legetøj og småbørnsartikler. Producenten betragter dog ikke TXIB som alternativ til DEHP, DBP og BBP, og brugen af TXIB i vinylgulvbelægninger har været faldende i 1990-erne på grund af høje emissionsrater fra produkterne. TXIB synes derfor ikke at være et egnet alternativ til DEHP, DBP og BBP. | NA |
Noter: *1: Baseret på en sammenligning med DEHP, men DBP og BBP er oplyst at have den samme pris og notationen kan derfor også bruges til at indikere prisen relativt til DBP og BBP.. "≈" betyder samme pris eller en smule højere eller lavere end DEHP; "+" betyder en del højere pris (10-50% højere)end DEHP og "++" betyder betydeligt højere pris end DEHP. Rapporten giver eksempler på de faktiske priser.
Alternativernes pris
Som det er vist i tabellen ovenfor var prisen på alternativerne DEHT, DGD og Benzoflex 2088 på det samme niveau som prisen på DEHP, DBP and BBP (såvel som på ortho-ftalat alternativerne), hvorimod ASE, DINA, DINCH og GTA var en del dyrere og ATBC og COMGHA var væsentligt dyrere. Indholdet af DEHP i blødgjort PVC er typisk 30% af plastikmaterialet, og en stigning i prisen på eksempelvis 30% vil resultere i en prisstigning på 10% på plastikmaterialet.
Prisen på kemikalier (og andre industriprodukter) har en tendens til at falde når produktionskapaciteten og konkurrencen stiger. Forskellige kemikalier er dog baseret på forskellige råmaterialer og mere eller mindre komplekse og ressourcekrævende kemiske synteseteknologier. Dette sætter naturligvis en grænse for den laveste pris, som kan opnås selv i et modent marked, og nogle af de beskrevne alternative blødgørere vil formentlig forblive på et højere prisniveau end ftalaterne. Det skal bemærkes, at prisen på DEHP er faldet væsentligt over de seneste ti år, formentlig på grund af en mindsket efterspørgsel, som konsekvens af reguleringen.
Udover prisen på blødgørerne, kan erstatningen af ftalaterne indebære nogle omkostninger til forskning og udvikling og til procesændringer, som ikke er blevet vurderet i denne undersøgelse.
Vurdering af alternative fleksible polymerer
Der er en række fleksible polymerer til rådighed, som kan erstatte blødgjort PVC til mange anvendelser. Polyethylen (PE), polyolefin elastomerer, forskellige typer af polyuretan (PU), ethylen vinyl acetat (EVA) and forskellige gummityper er eksempler.
For mange anvendelser af blødgjort PVC eksisterer der også andre erstatningsmaterialer end fleksible polymerer. Der er kun få eksisterende LCA-baserede, anvendelsesfokuserede vurderinger, og oftest har der ikke kunnet laves entydige konklusioner i undersøgelserne. Men der findes mange materialer, som synes at have lige så gode eller bedre profiler med hensyn til miljø, sundhed og sikkerhed. Vurderingerne, som er lavet i denne undersøgelse, tillader ikke en mere detaljeret analyse af muligheder og begrænsninger i brugen af alternative fleksible polymerer.
Miljø- og sundhedsvurdering
I tabel 4 er de iboende egenskaber af de undersøgte alternativer sammenfattet ved at angive en række nøgleparametre: akutte og lokale effekter, sensibilisering, kræftfremkaldende effekter, skader på arveanlæg, skader på fostre eller forplantningsevnen, persistens, bioakkumulering, og giftighed i vandmiljøet.
Af sammenfatningen ses, at alle ti stoffer - baseret på undersøgelser på dyr -forventes at have en lav akut toksicitet. Med hensyn til lokale effekter virker de fleste af stofferne ikke irriterende på hud og øjne eller giver kun anledning til en svag irritation, som ikke vil medføre en klassificering af stofferne. Ingen af de testede stoffer er sensibiliserende.
Effekter fra toksicitetsstudier med gentagne doser omfatter overvejende reduceret kropsvægt, forøget organvægt (lever og/eller nyre) og for nogle stoffer også ændringer i kliniske kemiske eller patologiske parametre. Der er dog ikke observeret væsentlige patologiske effekter.
Studier med henblik på at evaluere stoffernes potentiale for at indvirke på forplantningsevnen eller afkommets udvikling viser toksiske effekter på forældre og afkom. Der er blevet observeret en statistisk signifikant toksicitet af TXIB på forplantning og udvikling.
Muligheden for kræftfremkaldende effekter er kun blevet undersøgt for tre af stofferne i kombinerede undersøgelser. For alle tre stoffer var udfaldet negativt dvs. der sås ikke nogen kræftfremkaldende effekt. Undersøgelserne kan dog ikke anses for tilstrækkelige til at udelukke eventuelle kræftfremkaldende effekter.
Vurderingen af de toksiske egenskaber af ATCB, COMGHA, DINCH og DEHT i denne undersøgelse er i overensstemmelse med en vurdering som for nyligt er lavet af de samme stoffer af EUs videnskabelige komité for nye og nyligt identificerede sundhedsricici (SCENIHR).
Alle stofferne er blevet testet for akut toksicitet for mindst én eksponeringsvej, sensibilisering (pånær ASE), subkronisk toksicitet og skader på arveanlæg. Alle stofferne, på nær ASE, COMGHA og DINA, er testet for både skader på arveanlæg og skader på fostre eller forplantningsevnen.
Med hensyn til kræftfremkaldende effekt er kun ATBC, DEHT og DINCH blevet testet i undersøgelser for kombineret kronisk toksicitet og kræft. For DEGD, DGD og DEHT er den østrogene aktivitet blevet testet i en uterustest uden positiv respons
De fleste af dataene brugt til evalueringen anses for at være af god kvalitet dvs. undersøgelserne følger accepterede guidelines (OECD og US EPA) eller undersøgelserne betragtes som acceptable på det tidspunkt de blev udført. For nogle af studierne er det vanskeligt at vurdere kvaliteten da der kun er begrænset information til rådighed. Nøgleinformation er dog hentet fra IUCLID data ark, USEPA eller OECD ”HPV robust study summaries”.
Med hensyn til miljøegenskaber, er der ingen af de 10 undersøgte alternativer, som opfylder kriterierne for at være PBT stoffer (persistente, bioakkumulerbare eller toksiske i vandmiljøet) eller vPvB stoffer (meget persistente og meget bioakkumulerbare), selv om alle stofferne bortset fra GTA udviste én eller to af disse egenskaber. GTA (triacetin) synes at være let bionedbrydeligt, det bioakkumuleres ikke og det har en meget begrænset giftighed i vandmiljøet.
DEGD, DGD og DINA kommer også ud ganske gunstigt, hvorimod ATBC og COMGHA kommer negativt ud - på trods af deres nedbrydelighed - på grund af deres giftighed i vandmiljøet og bioakkumulerbarhed. Både ASE og DINCH har lav akut giftighed i vandmiljøet, men er ikke let nedbrydeligt og har en høj log KOW værdi. DEHT nedbrydes heller ikke let og er bioakkumulerbart, men dets giftighed i vandmiljøet kan på basis af de tilgængelige data ikke blive fuldt ud vurderet.
Anvendelige data vedrørende bionedbrydelighed (enten som BCF eller log KOW) er tilgængelige for alle alternativer, mens andre data vedrørende stoffernes skæbne i miljøet er mere ukomplette for nogle af stofferne. Med hensyn til data vedrørende økotoksikologiske effekter findes der for alle stoffer resultater fra kort-tids tests med basis-sættet af organismer: fisk, krebsdyr, og alger. Varigheden af nogle af undersøgelserne afviger dog fra den nuværende OECD standard.
Alt i alt er de fundne data af god kvalitet, dvs. de er hovedsageligt baseret på undersøgelser, som er udført i overensstemmelse med accepterede guideline- procedurer og undersøgelserne er blevet evalueret til at være pålidelige med forbehold (f.eks. i USEPA ”HPV robust summaries”).
Abbreviations and acronyms
| ASE | Sulfonic acids, C10 – C18-alkane, phenylesters |
| ASTM | American Society for Testing and Materials |
| ATBC | Acetyl tributyl citrate |
| BBP | Butyl benzyl phthalate |
| BCF | Bioconcentration factor |
| BOA | Benzyl octyl adipate |
| CEPE | European Council of producers and importers of paints, printing inks and artists’ colours |
| COMGHA | Mixture of COMGHA 1 and COMGHA 2 |
| COMGHA 1 | 12-(Acetoxy)-stearic acid, 2,3-bis(acetoxy)propyl ester |
| COMGHA 2 | Octadecanoic acid, 2,3-(bis(acetoxy)propyl ester. |
| CMR | Carcinogenic, mutagenic, reprotoxic |
| CSTEE | Scientific Committee on Toxicity, Ecotoxicity and the Environment |
| DBA | Dibutyl adipate |
| DBP | Di-n-butyl phthalate |
| DBT | Di-butyl terephthalate |
| DEGD | Diethylene glycol dibenzoate |
| DEHA | Bis(2-ethylhexyl) adipate |
| DEHP | Di(2-ethylhexyl) phthalate (also designates DOP) |
| DEHS | Dioctyl sebacate |
| DEHT | Di (2-ethyl-hexyl) terephthalate (same as DOTP) |
| DGD | Dipropylene glycol dibenzoate |
| DPHP | Di- (2-Propyl Heptyl) Phthalate |
| DiBA | Diisobutyl adipate |
| DIBP | Diisobutyl phthalate |
| DIDP | Diisodecyl phthalate |
| DINA | Diisononyl adipate |
| DINCH | Di-isononyl-cyclohexane-1,2dicarboxylate |
| DINP | Diisononyl phthalate |
| DNA | Deoxyribo Nucleic Acid |
| DNOP | Di-n-octyl phthalate |
| DOP | Di-octyl phthalate (same as DEHP) |
| DOTP | Di(2-ethylhexyl) terephthalate (same as DEHT) |
| DPHP | Di-(2-propyl heptyl) phthalate |
| DTDP | Ditridecyl Phthalate |
| ECHA | European Chemicals Agency |
| ECOSAR | Ecological Structure Activity Relationships |
| ECPI | European Council for Plasticisers and Intermediates |
| EPA | Environmental Protection Agency (here used for US EPA) |
| ESBO | Epoxidized soy bean oil |
| EU | European Union |
| GLP | Good Laboratory Practice |
| GTA | Glycerol Triacetate |
| HPV | High Production Volume |
| IC50 | Median Inhibitory Concentration. The concentration of a substance which reduces the effect by 50% |
| LC50 | Median Lethal Concentration. The concentration of a substance which causes the death of 50% of a group of test animals |
| LD50 | Median Lethal Dose 50. The dose of a substance which causes the death of 50% of a group of test animals |
| LG-flex BET | Hexanoic acid, 2-ethyl, mixed triesters with benzoic acid and trimethylopropane |
| LOAEL | Lowest Observed Adverse Effect Level |
| Log Kow | The logarithm to the octanol/water partition coefficient |
| Koc | Organic carbon normalised distribution coefficient |
| Kow | Octanol/water partition coefficient |
| NPG-EHA-BA | 2,2-Dimethyl-1,3-propanediol -benzoate 2-ethylhexanoate |
| OECD | Organisation for Economic Cooperation and Development |
| PA | Polyadipate |
| PBT | Persistent, bioaccumulative, toxic |
| PVA | Polyvinyl acetate |
| PVC | Polyvinyl chloride |
| P/vP | Persistent/very persistent |
| RAR | Risk Assessment Report |
| SCENIHR | EU Scientific Committee on Emerging and Newly Identified Health Risks |
| TBAC | Tert-butyl acetate |
| TBG-EHA-BA | Mixed triesters 1,1,1-trimethylol-propane-benzoate/2-ethylhexanoate |
| TEHTM | Tri-(2-ethylhexyl)-trimellitate (same as TOTM) |
| TGD | Triethylene glycol dibenzoate |
| ThOD | Theoritical Oxygen Demand |
| TOTM | Tris- (2-ethyhexyl) trimellitate (same as TEHTM) |
| TXIB | Trimethyl pentanyl diisobutyrate |
| UK | United Kingdom |
| USEPA | United States Environmental Protection Agency |
1 Introduction
1.1 Data collection
Besides collection of information from the literature basic information has been collected from three major groups of data sources:
- Manufacturers and suppliers of toys and childcare articles
In cooperation with the Nordic Association of Toy Manufacturers, manufacturers of toys and childcare articles in Denmark was identified and contacted by e-mail and telephone in order to collect information on applied plasticisers. This survey was supplemented with enquiries to major importers of toys in Denmark identified via the Internet and business databases.
- Manufacturers of alternative plasticisers and materials
Manufacturers of alternative plasticisers and materials were indentified via the Internet and the literature. Information on technical properties and the substances’ ability to substitute for the phthalates concerned, the price in comparison to the phthalates and the human and environmental properties were collected by direct queries to the manufacturers, by use of email or/and telephone interview.
- Databases
A number of databases were consulted in order to collect information on physico-chemical properties and human health and environmental effects of selected substances. Data has been searched for in among others the European Chemical Substances Information System database (ESIS), OECD's e-ChemPortal, CHEM ID, IPCS InChem database, SRC Chemfate, HSDB, IRIS, CCRIS, TOXLINE, NTP – National Toxicology Program, Chemical Health & Safety Data, ECOTOX and RTECS.
Contacted companies and organisations are listed in Annex 1
2 Application of DEHP, DBP and BBP in products and articles
2.1 The phthalates
The term "phthalates" is here (and commonly) used for esters of ortho-phthalic acid and are the most commonly used plasticisers in the world.
The general structural formula of phthalates is shown in Figure 2.1. The functional groups (indicated as R) can either be linear aryl groups or alkyl groups with an aromatic ring. The properties of the phthalates are determined by the functional groups.
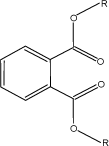
Figure 2.1 General structural formula of ortho-phthalates. R can be linear alkyl groups or aryl groups with an aromatic ring.
Examples of two of the phthalates are shown below, DEHP with symmetric linear functional groups and BBP with asymmetric aryl and alkyl functional groups.
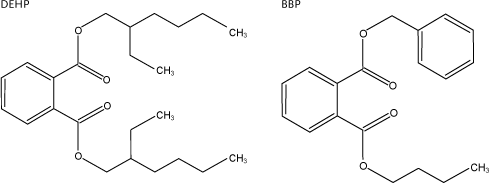
Figure 2.2 Examples of structural formula of phthalates
The softening effects of the phthalates decrease with increasing length of the functional groups (Hoffmann, 1996): DBP > BBP > DEHP > DINP > DIDP > DTDP. Consequently higher loading are necessary to obtain a certain level of softness for the phthalates with longer functional groups.
One property of importance for the application of the phthalates concerned in this study is the gelling ability of the plasticiser i.e. the ability of the plasticiser to mix with the resin by processing. The ability decrease in the following order: BBP > DBP > DIHP > DEHP > DINP > DIDP > DTDP (Hoffman, 1996).
The most common phthalates are di-isononyl phthalate (DINP); di-isodecyl phthalate (DIDP); and di-2-ethyl hexyl phthalate (DEHP).
Phthalates are widely used as general-purpose plasticisers because they offer the advantage of lower costs and increased production efficiency. When added to plastics, phthalates allow the long polyvinyl molecules to slide against one another. The phthalates show low water solubility, high oil solubility, and low volatility.
This is achieved through improved melt viscosity and production speeds of heated PVC with phthalates added. Increased flow characteristics give better workability and reduced out-of service breaks in equipment.
| Chemical name | Abb. | CAS No | Classification cf. 1272/2008 (GHS) |
Use restrictions |
|---|---|---|---|---|
| Di(2-ethylhexyl) phthalate | DEHP, DOP | 117-81-7 | Repr. 1B (Repr. Cat. 2; R60-61) |
Shall not be used as substances or as constituents of preparations, at concentrations of greater than 0,1% by mass of the plasticised material, in toys and childcare articles (Regulation No 552/2009) Not permitted for use in cosmetics (Directive 2004/93/EC) |
| Di-n-butyl phthalate | DBP | 84-74-2 | Repr. 1B Aquatic Acute 1 (Repr. Cat. 2;R61 Repr. Cat. 3; R62 N; R50) |
|
| Butyl benzyl phthalate | BBP | 85-68-7 | Repr. 1B Aquatic Acute 1 Aquatic Chronic 1 (Repr. Cat. 2; R61 Repr. Cat. 3; R62 N; R50-53) |
|
| Diisononyl phthalate | DINP | 28553-12-0 68515-48-0 |
No harmonised classification | Shall not be used as substances or as constituents of preparations, at concentrations of greater than 0,1% by mass of the plasticised material, in toys and childcare articles which can be placed in the mouth by children (Regulation No 552/2009) |
| Diisodecyl phthalate | DIDP | 26761-40-0 271-091-4 |
No harmonised classification | |
| Di-n-octyl phthalate | DNOP | 117-84-0 | No harmonised classification | |
| Diisobutyl phthalate | DIBP | 84-69-5 | Repr. 1B (Repr. Cat. 2; R61 Repr. Cat. 3; R62 ) |
Not permitted for use in cosmetics (Directive 2004/93/EC) |
| Chemical name | Abb. | Use and technical properties |
|---|---|---|
| Di(2-ethylhexyl) phthalate | DEHP | Offers a good all-round performance and is therefore used for a great many cost-effective, general purpose products including building material such as flooring, cables, profiles and roofs, as well as medical products such as blood bags and dialysis equipment The content of DEHP in flexible polymer materials varies, but is often around 30% (w/w). |
| Di-n-butyl phthalate | DBP | Is a specialist plasticiser often used in combination with other high molecular weight phthalates. It is a fast fusing plasticiser which by itself it is too volatile for PVC applications. It is frequently used as a gelling aid in combination with other plasticisers. DBP is used extensively in the adhesives industry to plasticise polyvinyl acetate (PVA) emulsions and used solvent for many oil-soluble dyes, insecticides, peroxides and other organic compounds. It is used as an antifoam agent and as a fibre lubricant in textile manufacturing. |
| Butyl benzyl phthalate | BBP | Butyl benzyl phthalate is an unusual plasticiser because of its chemical asymmetry which results in unique performance properties. One of the manufacturing benefits of using BBP is that it allows PVC transformers to operate with less energy input than with many similar plasticisers. It is used widely by the flooring industry because it adds surface properties to flooring materials that minimise maintenance and give it a prolonged life. More than 90% of BBP is used for plasticising PVC or other polymers. Butyl benzyl phthalate is also used in seals for insulating double glazing |
| Diisononyl phthalate | DINP | General, all-purpose plasticiser. Wide range of indoor and outdoor applications. 95 per cent of DINP is used as a plasticiser for flexible PVC used for construction and industrial applications, and durable goods (wire and cable, film and sheet, flooring, industrial hoses and tubing, footwear, toys, food contact plastics). Heat resistance, low temperature resistance and volatility resistance are some of its main properties that make it suitable for a wide range of applications (ECPI, 2009) |
| Diisodecyl phthalate | DIDP | Diisodecyl phthalate (DIDP) is a common phthalate plasticiser, used primarily to soften Polyvinyl chloride (PVC). The typical content of DIDP in flexible PVC products is between 25 and 50% (w/w). It has properties of volatility resistance, heat stability and electric insulation and is typically used as a plasticiser for heat-resistant electrical cords, leather for car interiors, and PVC flooring (ECPI, 2009). |
| Di-n-octyl phthalate | DNOP | The principal use of DNOP is as a plasticiser in the production of PVC resins and other polymers, such as cellulose esters and polystyrene resins, as a dye carrier in plastic production (primarily PVC), and as a chemical intermediate in the manufacture of adhesives, plastisols, and nitrocellulose lacquer coatings (ATSDR, 2005) DNOP may represent 5 to 60% of the total weight of the plastics and resins (ATSDR, 2005) No actual confirmation of the use of DNOP in the EU today has been identified |
| Diisobutyl phthalate | DIBP | Diisobutyl phthalate (DIBP) is a specialist plasticiser often used in combination with other high molecular weight phthalates. It is a fast fusing plasticiser which by itself it is too volatile for PVC applications. It is frequently used as a gelling aid in combination with other plasticisers. A plasticiser for nitrocellulose, cellulose ether, and polyacrylate and polyacetate dispersions. Has very similar application properties to DBP and may therefore be used to substitute for DBP in most, if not all, of its applications. |
The main alternatives to DEHP have been the two phthalates di-isononyl phthalate (DINP) and di-isodecyl phthalate (DIDP). As illustrated in Figure 2.3 in Sweden the shift from DEHP to first of all DINP took mainly place in the period 1999 to 2002.
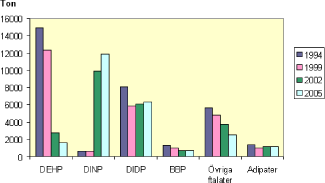
Figure 2.3 Use of plasticisers for PVC in Sweden. KemI (2008) quoting the Swedish Productregister as source. 2005 figures are indicated as "preliminary". "Övrige ftaalter" = "Other phthalates", ”Adipater” = ”Adipates”.
The evolution in the EU use of plasticisers shows that the consumption of DEHP deceased from 1999 to 2005 from 42% of the market to 21% (Figure 2.4). The DEHP was replaced by DINP/DIDP, while the non-phthalate plasticisers remained a market share of around 7-8%.
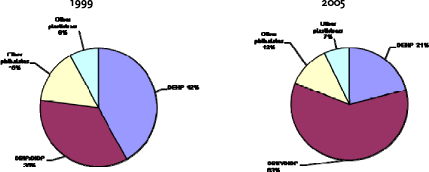
Figure 2.4 Evolution of PVC plasticisers’ sales between 1999 and 2005 (based on Cadogan, 2006)
Technical key parameters of some ortho-phthalates
For comparison with other plasticisers described in this report, some technical key parameters for selected phthalates are shown in Table 2.3. Note that for these related substances, the larger the molecule, the lower is the volatility, the extractability and the softness (efficiency), whereas the gelling temperature increases with higher molecule weight.
2.2 Application of DEHP
The following information is, if nothing else is mentioned, extracted from an assessment of the manufacturing and use of di(2-ethylhexyl) phthalate (DEHP) recently published by the European Chemicals Agency (COWI, 2009a).
DEHP is widely used as a plasticiser in polymer products, mainly PVC. It offers a good all-round performance and is therefore used for a many general purpose products. The content of DEHP in flexible polymer materials varies, but is often around 30% (w/w).
The main end-product uses of DEHP are as follows:
- Flooring:
- PVC flooring (with PVC surface);
- Carpets with PVC back-coating;
- Cork with PVC top-coating or back-coating ;
- Wall covering;
- Roofing;
- Film/sheet and coated products:
- Curtains, blinds, table linen, etc.;
- Packaging;
- Tape and self-adhesive foils;
- Office supplies (ring binders, files, slip cases, etc.);
- Medical bag/sheet devices;
- Bottom sheets for hospitals.
- Wires and cables;
- Hoses and profiles;
- Garden hoses and tubes;
- Hoses and tubes in industry;
- Profiles of windows and electrical products;
- Medical tubing.
- Coated fabric;
- Upholstery and car seats (synthetic leather);
- Luggage;
- Rainwear;
- Tarpaulins;
- Water beds.
- Moulded product;
- Footwear;
- Adult toys; (DEHP is not permitted in toys for children)
- Car undercoating;
Non-polymer applications:
- Adhesives;
- Lacquers and paints;
- Printing inks (see comment below);
- Sealants (glass insulation, construction);
- Ceramics.
The main part of DEHP is added to PVC in different formulation and processing steps including calendering, extrusion, spread coating, and moulding.
At EU level in total 341,000 tonnes was produced in 2007. A part of this was exported and around 291,000 tonnes DEHP was used for manufacturing processes in the EU in 2007. DEHP accounted for around 18% of all plasticiser usage in Western Europe (data for the entire EU plasticiser use is not available). The consumption of DEHP has decreased markedly the last decade. Of the 341,000 tonnes produced in the EU in 2007, 187,000 tonnes were produced in Western Europe corresponding to 31% of the 1997 level of 595,000 t/y. In the early 1990s, DEHP represented about 51% of the total phthalate plasticiser market in Western Europe.
Investigations of the historic use of phthalates in Denmark in different products shows that DEHP represented a major part of the phthalate use in the 1990's and was to a large extent replaced by DINP around 2000. Today DEHP seems to have been replaced by DINP or non-phthalate plasticisers in Danish produced PVC products (Christensen et al., 2007; Brandt et al., 2009).
Quantitative information is available on the approximate split between the different processes. In combination with information on which products are produced by different processes it is possible to indicate the main and uses of DEHP as illustrated in Figure 2.5.
DEHP is to some extent imported and exported with finished products. Table 2.4 shows the estimated extra-EU import and export for some of the main product groups, for which information can be estimated on the basis of the trade statistics (please see COWI, 2009a for details regarding the calculation method). For most product groups the net import is relatively small compared to the production within the EU, and the end-use distribution follows in broad outline the distribution of EU production.
| End-product use area | Tonnage, t/y | % of | |||
|---|---|---|---|---|---|
| EU Manufacture | Import | Export | End-product use | total use | |
| Polymeric applications: | |||||
| Flooring | 33,000 | 2,000 | 4,800 | 30,200 | 10.6 |
| Wall covering | 11,000 | 700 | 1,600 | 10,100 | 3.5 |
| Roofing material | 3,600 | n.d. | n.d. | 3,600 | 1.3 |
| Film/sheet and coated products made by calendering | 44,000 | 13,600 | 16,400 | 41,200 | 14.5 |
| Wires and cables | 64,100 | 6,200 | 5,600 | 64,700 | 22.3 |
| Hoses and profiles | 34,700 | 1,600 | 3,000 | 33,300 | 12.7 |
| Coated fabric and other products from plastisol | 43,800 | 2,200 | 1,400 | 44,600 | 15.1 |
| Shoe soles | 19,400 | n.d. | n.d. | 19,400 | 6.8 |
| Other moulded products | 3,000 | 2,700 | 700 | 5,000 | 1.8 |
| Car undercoating | 4000 | n.d. | n.d. | 4000 | 1.4 |
| Other polymer applications | 12,300 | 10,900 | 3,100 | 20,100 | 7.1 |
| Non polymer applications: | 0.0 | ||||
| Adhesives and sealant | 4,000 | n.d. | n.d. | 4,000 | 1.4 |
| Lacquers and paints | 900 | n.d. | n.d. | 900 | 0.3 |
| Adhesives and sealant | 3300 | n.d. | n.d. | 3300 | 1.2 |
| Printing ink | 1,000 | n.d. | n.d. | 1,000 | 0.4 |
| Other non-polymeric | 20 | n.d. | n.d. | 20 | 0.0 |
| Total end-product use (round) | 282,000 | 40,000 | 37,000 | 285,000 | 100 |
2.3 Application of DBP
The following information is, if nothing else is mentioned, extracted from an assessment of the manufacturing and use of dibutyl phthalate (DBP) recently published by European Chemicals Agency (COWI, 2009b).
DBP is a specialist, fast fusing plasticiser. By itself it is too volatile for PVC applications and it is used in PVC as a gelling aid in combination with other high molecular weight plasticisers. The gelling agent is the agent which reacts fastest with the PVC.
The total manufactured tonnage in 2007 in the EU is confidential, but it was less than 10,000 tonnes. A significant part of the manufactured tonnage is exported to countries outside the EU. DBP seems to represent less than 1% of the production of phthalates in Europe. As DBP is used in relatively small concentrations it may however be present in higher share of products. Dutch surveys of phthalates and other plasticisers in toys and childcare products demonstrate that 30% of 24 analysed products in 2004 contained DBP (FCPSA, 2008a). The share had decreased to 13% of the products in 2007 and 1% in 2008 (FCPSA, 2008b).
The market for DBP has been decreasing over recent decades: In 1994 the production volume of DBP in the EU was 49,000 tonnes and in 1998 it was 26,000 tonnes, with an export of 8,000 tonnes.
Current uses of DBP, according to actual information obtained from industry or product registers, are listed below:
- Gelling aid in combination with other plasticisers in plastics. In general, limited information is available on the actual uses of DBP in polymers. DBP is used in PVC (manufacturer information). It has not been possible to obtain very specific information on the uses, but the following applications are mentioned by different sources: floor coverings, automotive uses (manufacturer information) and garden hoses. The European Plastic Converters (EuPC), has in a survey by their members not indentified any use of DBP, and assume that DBP today is used by relatively few companies for different niche purposes. The results of the Dutch surveys show that DBP was formerly used at a relatively high frequency in many different types of toys (FCPSA, 2008a).
- Rubbers (manufacturer information). The Risk Assessment for DBP (ECB, 2004) specifies that DBP is used in some polychloroprene rubber and nitrile rubber, but not in all polychloroprene (neoprene) or nitrile rubbers. New information on actual uses is not available.
- DBP is used extensively in the adhesives industry to plasticise polyvinyl acetate (PVA) emulsions. The low viscosity and compatibility of DBP make it suited for PVA-based adhesives for bonding cellulosic materials. According to the Risk Assessment for DBP (ECB, 2004) the most important uses of the adhesives are for paper and packaging, wood building and automobile industry.
- Epoxy resins. Probably same application that in the Risk Assessment for DBP (ECB, 2004) is mentioned as "solvent in the production of fiber glass". More specific information on this application has not been available.
- In the coatings industry as a primary plasticiser-solvent for nitrocellulose lacquers.
- Grouting agents, used to reduce water leakages in tunnels, sewer systems, buildings etc. DBP contents as high as 30-60% were found in polyurethane foams used in grouting applications for water control in tunnels, sewer systems, buildings etc. No actual confirmation of this application has been obtained.
- Other applications:
- Solvent for many oil-soluble dyes, insecticides, peroxides and other organic compounds;
- Antifoam agent and as a fibre lubricant in textile manufacturing;
- Used in compounding flavours;
- Printing inks, polishing agents, corrosion inhibitor materials;
- Use in PP (polypropylene) catalytic systems;
- One application described in the confidential part of the ECHA report.
According to CEPE (European Council of producers and importers of paints, printing inks and artists’ colours), DEHP, BBP and DBP are no longer used in printing inks by CEPE/EuPIA (European Printing Ink Association) members following its classification as reprotoxic.
The flow chart below illustrates the "best estimate scenario" for the flow of DBP through the different process and the resulting end-products. It was for the ECHA report (COWI, 2009b) not possible to obtain comprehensive quantitative updated information on the use of DBP for the different uses from manufacturers and suppliers and the available information did not allow real estimates of the distribution between the different use areas to be made. It is, however, deemed that the distribution between applications most likely is different from the 1997 distribution used in the RAR, but the updated distribution is highly uncertain. The figures are for confidentiality reasons rounded and somewhat higher than the actual figures.
DBP may be exported and imported in preparations and articles, but no statistical information allowing an estimate of this import/export is available.
Use in Denmark
No detailed assessment of the use of DBP in Denmark exists. The use of DBP in chemical products (preparations) registered in the Danish product register is shown in Table 2.5. The total consumption of DBP in declared preparations has decreased from 320 tonnes/year in 2000 to 71 tonnes/year in 2004. In the period from 2004 to 2006 the registered consumption was stable at a level of 70-80 tonnes. The main use category was paint, lacquers and varnishes and in total 56 different products within this use category in was registered in 2006. Other preparations representing significant uses of DBP were binding agents (may be used as synonymous for adhesives) and corrosion inhibitors/under seal materials.
The declaration of the substances to the product register is mandatory for preparations containing one or more substances listed in Annex 1 of Directive 67/548/EEC (now the Annex VI to Regulation 1272/2008). All preparations containing DBP should consequently be declared. However, there is no systematic updating of quantities of products. The companies are obliged to send in any new information regarding their products whenever changes occur. If companies fail to fulfil their obligations, a result might be that products that have been discontinued still remain on the lists. The relatively high consumption of DBP in paint, lacquers and varnishes may reflect that DBP is used for production of some specific paints by Danish paint manufacturers.
The data shows a steep decrease i the use of DBP in "flooring materials", "casting materials", " printing inks", "adhesives" and "other".
| Product group | Registered consumption, tonnes DBP/year | ||||||
|---|---|---|---|---|---|---|---|
| 2006 | 2005 | 2004 | 2003 | 2002 | 2001 | 2000 | |
| Paint, lacquers and varnishes | 64.7 | 69.4 | 67.0 | 29.0 | 27.8 | 27.9 | 63.4 |
| Binding agents | 9.1 | 1.4 | 0.8 | 21.1 | 13.3 | 21.4 | 21.7 |
| Filling materials | 0.2 | 0.2 | 0.2 | 2.7 | 51.1 | 49.1 | 50.2 |
| Hardeners, curing agents | 1.7 | 2.0 | 2.0 | 5.9 | 9.8 | 10.3 | 28.6 |
| Flooring materials (joint-less floors) | 0.0 | 0.1 | 0.1 | 18.3 | 18.9 | 19.0 | 19.0 |
| Corrosion inhibitors, anticorrosive paints, underseal materials, incl. cavity seals | 3.9 | 0.1 | 0.1 | 0.2 | 0.3 | 0.3 | 0.7 |
| Casting materials | 0.5 | 0.1 | 0.1 | 3.3 | 4.2 | 4.3 | 6.1 |
| Surface treatment for non-metals | 0.0 | 0.1 | 0.1 | 1.7 | 1.7 | 1.7 | 1.7 |
| Polishing agents | 0.2 | 0.2 | 0.2 | 0.6 | 0.0 | 0.0 | 1.2 |
| Printing inks | 0.7 | 0.9 | 0.1 | 0.7 | 0.3 | 0.3 | 102.0 |
| Adhesives | 0.0 | 0.0 | 0.0 | 4.4 | 0.2 | 0.2 | 16.8 |
| Other | 0.0 | 0.0 | 0.0 | 8.7 | 22.4 | 29.5 | 8.7 |
| Total | 81 | 75 | 71 | 97 | 150 | 164 | 320 |
2.4 Application of BBP
The following information is, if nothing else is mentioned, extracted from an assessment of the manufacturing and use of butyl benzyl phthalate (BBP) recently published by European Chemicals Agency (COWI, 2009c).
The total manufactured tonnage in 2007 in the EU was below 18,000 tonnes. A significant part of the manufactured tonnage is exported to countries outside the EU. BBP seems to represent less than 1% of the production. The market for BBP has been decreasing over the last decade. In the period 1994-1997, the total reported Western European manufacture of BBP was 45,000 tonnes/year whereas for 2004 a production volume of 19,500 tonnes/year was reported.
More than 70% of the BBP was in 2007 used as a plasticiser in polymer products, mainly PVC for flooring. BBP is, according to industry, an unusual plasticiser because of its chemical asymmetry which results in unique performance properties.
BBP is typically used together with other plasticisers e.g. DEHP or DINP. It has not been possible to identify information on typical concentration of BBP in the final materials, but examples of formulations indicate that the concentration may be in the range of 5-20%. As an example Eastman (2001) compares two different BBP/DINP plastisol formulations with other formulation. One of the formulations has 12 parts BBP and 28 parts DINP (together with other additives) to 100 parts PVC whereas the other has 24 parts BBP and 56 parts DINP to 100 parts PVC.
BBP is used widely by the flooring industry because it speeds up production and adds surface properties to flooring materials that minimise maintenance and give it a prolonged life.
The end-product uses of BBP are as follows:
- Flooring (both calendered and spread coated flooring);
- Wall covering;
- Coating of leather and textiles (upholstery, shoe uppers, wallets/bags, luggage);
- Packaging films;
- Sealants (polysulphide based, polyurethane based or acrylic-based) for insulating double glazing and other applications;
- Paints for car care and construction (acrylic lacquers and other);
- Inks for paper and board;
- Adhesives (polyvinyl acetate and other);
- Miscellaneous (hard PVC, nitrile rubber and other).
The main part of BBP is added to PVC in different formulation and processing steps including calendering and plastisol spread coating. The following flow diagram illustrates the relationship between the different processes and the end-product uses. The major application area is flooring accounting for about 50% of the total while the second larges application area, polysulphide sealants account for about 19% of the total.
BBP may be exported and imported in preparations and articles, but no statistical information allowing an estimate of this import/export is available.
Use in Denmark
No detailed assessment of the use of DBP in Denmark exists. The use of DBP in chemical products (preparations) registered in the Danish product register is shown in Table 2.6. The total consumption of DBP in declared preparations has decreased from 233 tonnes/year in 2000 to 75 tonnes/year in 2004. In the period from 2004 to 2006 the registered consumption was fairly stable at a level of 60-75 tonnes.
The main use categories were filling materials (may be used synonymous with sealants), paint, lacquers and varnishes and adhesives. The increase in the use of DBP from a level below 1 tonnes/year in 2000-2005 to 11 tonnes in 2006 may be due to an error in the registration.
The declaration of the substances to the product register is mandatory for preparations containing one or more substances listed in Annex I to Directive 67/548/EEC (now Annex VI to Regulation 1272/2008) . All preparations containing BBP should consequently be declared. However, there is no systematic updating of quantities of products. The companies are obliged to send in any new information regarding their products whenever changes occur. If companies fail to fulfil their obligations, a result might be that products that have been discontinued still remain on the lists. This may explain the relatively high registered consumption of DBP in preparations in Denmark compared to the EU consumption for non-polymeric applications as shown in Table 2.5.
| Product group | Registered consumption, tonnes BBP/year | ||||||
|---|---|---|---|---|---|---|---|
| 2006 | 2005 | 2004 | 2003 | 2002 | 2001 | 2000 | |
| Filling materials | 47.8 | 46.7 | 46.7 | 65.0 | 19.2 | 8.4 | 79.3 |
| Paint, lacquers and varnishes | 14.9 | 8.7 | 25.5 | 18.4 | 9.3 | 14.2 | 45.8 |
| Adhesives | 10.9 | 0.1 | 0.2 | 0.5 | 0.1 | 0.1 | 0.3 |
| Casting materials | 0.2 | 2.7 | 2.7 | 0.0 | 7.0 | 7.0 | 0.0 |
| Binding agents - for binding together the individual constituents in the product | 0.4 | 0.0 | 0.0 | 1.1 | 2.9 | 2.9 | 3.9 |
| Padding materials | 0.0 | 0.0 | 0.0 | 0.0 | 40.1 | 40.1 | 0.0 |
| Tightening materials (putty) | 0.0 | 0.0 | 0.0 | 0.0 | 28.5 | 28.6 | 0.0 |
| Other and unknown function | 0.3 | 0.3 | 0.3 | 0.5 | 164.8 | 110.9 | 103.3 |
| Total | 75 | 59 | 75 | 86 | 272 | 212 | 233 |
3 Identified alternatives to DEHP, DBP and BBP
- 3.1 Functional mode of external plasticisers
- 3.2 Introduction to plasticiser substance families
- 3.3 Alternative plasticisers and polymers used in toys and childcare articles
- 3.4 Alternatives recommended by plasticiser producers
- 3.5 Alternative plasticisers applied with processing adjustments
- 3.6 Alternatives plasticisers selected for further assessment
- 3.7 Alternative flexible polymers
Information on alternatives to DEHP, DBP and BBP, actually applied today, has been collected from the following data sources:
- Danish manufacturers and importers of toys and childcare articles;
- Surveys of plasticisers in marketed toys and childcare articles;
- Direct contact to major suppliers of plasticisers.
These data sources have been supplemented with information from the literature and manufacturers' web sites.
The first section gives a general introduction to the way plasticisers work and to the different available plasticiser substance families, followed by a description of information collected directly from the above mentioned data sources.
3.1 Functional mode of external plasticisers
To get an impression of the many possibilities for plasticising polymers, we give an introduction to the basic functions of plasticisers, the variability in these functions, and which properties govern the functions. The description is used as basis for the later discussion of the technical feasibility of replacing phthalate plasticisers with alternatives.
We describe here the basics of external plasticisation of PVC, the major use of plasticisers. The word "external" denotes plasticisers that are not bound chemically in the polymer matrix, and can therefore migrate out of the polymer at certain conditions. Polymers can also be plasticised "internally" by incorporation of functional groups into the polymer itself, which imparts flexibility. Phthalates are external plasticisers, as are their direct substitutes, and external plasticisation is described in this section.
PVC consists of long chains of the basic vinyl building block. The polymer is bound together in three dimensions by two overall types of forces. In some points the polymer is crystallised into a fixed geometric pattern with strong chemical bonds. In the rest of the polymer matrix, the polymer chains are somewhat more randomly organised and bound together by weaker forces based on attraction between polar parts of the polymer chain with different polarity. The ideal plasticiser works in these less strictly organised parts of the polymer.
In the hard polymer, the chains are packed closely together, also in the randomly organised parts, and the weak attraction forces bind the polymer together to a rigid structure with no flexibility. The (external) plasticiser has solvent capabilities and penetrates the less strongly bound parts of the polymer in the so-called swelling, where plasticiser and polymer resin is mixed. In the polymer, the plasticiser acts as a kind of sophisticated lubricant, as it creates distance between the freely organised polymer chain parts, and shields the attraction forces between polar parts of the chain, and thereby weakens the attraction between the chain parts. This allows for more free movement amongst the weakly bound chain parts, which means that the material becomes flexible.
The properties of the plasticiser have immense influence of how well it plasticises the polymer, and on the performance characteristics of the plasticised material. It is however important to understand that the plasticiser (with a few exceptions) does not form specific chemical bonds with the polymer, and there is therefore in principle a flexibility in which type and configuration of plasticisers that actually can be used to obtain the desired plasticising performance characteristics.
External plasticisers may be separated from the PVC matrix due to extraction by solvents, oils, water, surface rubbing, volatility, migration into adjacent media, or degradation mechanisms.
Structure of some plasticiser families
As mentioned, many families of plasticisers are available. Most of them have however certain chemical functionalities in common with the phthalates family. This can be seen in Figure 3.1, which shows representatives of some different plasticiser families. They are typically branched, quite "voluminous" molecules, with many oxygen bonds (= carbonyl groups). Many have benzyl rings or the hydrogenated counterpart, cyclohexane.
Even so, many similar plasticisers have distinctly different impacts on health and environment, and are therefore relevant alternatives to phthalates. This is probably primarily due to the fact that many types of interactions with biological systems are substance specific, and even shape-specific (structurally specific), meaning that substances with identical chemical composition may work differently, if just a part of the molecule has shifted position from one place to another.
| Phthalate (DEHP, DOP; di 2-ethylhexylphthalat) | 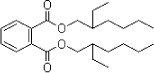 |
| Terephthalate, (DEHT, DOT DOTP; di 2-ethylhexyl terephthalate) | 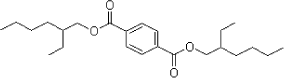 |
| Trimellitates (TOTM; tri (2-ethylhexyl) trimellitate) | 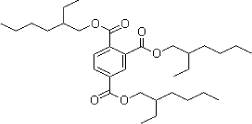 |
| Aliphatic dibasic esters, adipates (DEHA, DOA = di (2-ethylhexyl) adipate ) |  |
| (DBS = Dibutyl sebacate) |  |
| Benzoates (DGD; dipropylene glycol dibenzoate) | 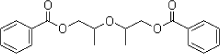 |
| Citrates (ATBC; acetyl tributyl citrate) | 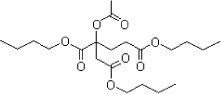 |
| Phosphates (tri(2-ethylhexyl) phosphate) | 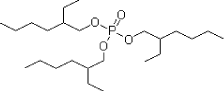 |
| Glyceryl triacetate (GTA, Triacetin) |  |
The substance family of the plasticiser influences its performance significantly, but some functional groups in the molecules also influence the performance across families, and plasticisers can thus to a certain extend be tailor-made to suit different performance needs. In addition, plasticisers can be mixed to achieve desired properties. Some of the important performance parameters of plasticisers are shown in Table 3.1 with some general chemical features influencing a plasticiser's performance for each parameter. All these parameters influence the performance of a plasticiser, and they are not independent, because one change in the plasticiser molecule may affect most or all of the parameters. Finding the good plasticiser is therefore not a distinct theoretical science, but rather an empiric process supported by a large number of measuring methods designed for this purpose.
| Parameter | Plasticiser characteristics influencing parameter |
|---|---|
| Solvency in polymer resin (also called compatibility or miscibility) | The good plasticiser for PVC has, within its molecule, a suitable mix of polar and apolar functional groups. Oxygen (carbonyl groups) and aromatic rings impart higher solvency. Plain hydrocarbon chain parts add apolarity, and thus lower solvency. Good solubility is required for a plasticiser, but too high solubility dissolves the crystalline parts of the polymer and thereby breaks the polymer apart. Lower molecular weight phthalates such as BBP, and other high aromaticity substances such as benzoate esters and tri(cresyl) phosphate, or more polar structures such as sulfonates, are examples of high solvency plasticisers. |
| Efficiency (defined as flexibility in polymer compared to DEHP) |
Branched hydrocarbon chains in the plasticiser increase flexibility amongst the polymer chains, and thus the overall flexibility of the resulting material. Too many hydrocarbon branches decrease solubility and resistance to hydrolysis (degradation in contact with water). |
| Volatility | Smaller, lower weight molecules tend to have higher volatility than larger, heavier molecules. For example DBP is deemed to be too volatile for many polymer applications, while a large molecule like ditridecyl phthalate has low volatility and can therefore be used in polymers exposed to elevated temperatures. Large molecules like trimellitates and polyesters have typically even lower volatility. In some cases high volatility is desired. BBP is an example of as plasticiser which can contribute to volatile fuming during processing and volatilization in end use applications, and thus give a hardened, stain resistant surface, due to volatilization. The same is the case for certain benzoates. Volatile losses of plasticiser are influenced by vapour pressure, solvency strength for the polymer and oxidative degradation. |
| Diffusivity | Movements of the plasticisers within the polymer matrix are ruled by diffusion. Low diffusivity is contributed by high molecular weight and highly branched isomeric structures. For example, DIDP and the polyester family impart improved resistance to diffusion-controlled plasticiser losses. Plasticiser losses due to extraction by oily media (in which plasticisers are highly soluble) are controlled by diffusivity rates. |
| Low temperature performance | Higher share of linear hydrocarbons (versus branched hydrocarbons) in the plasticiser increase flexibility at low temperatures. The entire family of aliphatic dibasic esters contributes exceptional low temperature properties. Di-2-ethylhexyl adipate (DEHA, DOA) is the standard and most widely used plasticiser in this class. Di-2-ethylhexyl azelate (DOZ), di-2-ethylhexyl sebacate (DOS), and diisononyl adipate (DINA) are used for low temperature applications requiring lower plasticiser volatility. |
Another important factor for plasticiser selection is the ease of processing of the resin-plasticiser system in the various steps involved in flexible polymer manufacture. Both the polymer and the plasticiser characteristics influence the processability. For a given polymer resin, the choice of plasticiser influences the temperatures needed for gelling (absorption of the plasticiser in the resin) and fusing (settling of the mixture in its final state), and the viscosity of the hot PVC melt or the plastisol blend, etc. Solvency for the PVC resin plays a role, and strong solvating plasticisers may be mixed into the general plasticiser to enhance processability. The volatility of the plasticiser, on the other hand, is typically the limiting factor on levels of strong solvating plasticisers used. Higher molecular weight plasticisers typically decrease volatility, but also viscosity, etc., with resulting constraints in processability (Krauskopf and Godwin, 2005). BBP is an example of a plasticiser which can reduce the operating temperatures in PVC processing.
3.2 Introduction to plasticiser substance families
This section focuses on the alternative plasticisers. The phthalates are described in more detail in Chapter 2.
Hundreds of substances have plasticising properties in PVC and other polymers. According to Krauskopf and Goodwin (2005), about 70 different plasticisers are available today, even though the consumption has so far been dominated by phthalates. DEHP, DINP and DIDP together comprise around 80% of global plasticiser consumption.
The main families of available plasticisers are shown in a generalised overview in Table 3.2 along with an indication of their performance characteristics. It shall be noted, however, that a number of plasticisers on the market are not member of any of the listed substance families. Each substance family has many members with different performance characteristics, and the table may not include all performance options for all families. The table also shows a traditional grouping of plasticisers in "general purpose" plasticisers with a vide application field and currently low prices (such as DEHP), "performance plasticisers" which provide special performance possibilities and are currently more expensive (such as DBP and BBP), and last "speciality plasticisers" which also provide other functionalities than flexibility and have generally currently even higher prices (Krauskopf and Goodwin, 2005).
A general description of the plasticiser substance families is given in Annex 2. Technical aspects for selected plasticisers are described in more detail in Chapter 6.
| Substance family | General purpose | Performance plasticisers | Specialty plasticisers | ||||
|---|---|---|---|---|---|---|---|
| Strong solvent | Low temperature | Low volatility | Low diffusion | Thermal & UV stability | Flame resistance | ||
| Phthalates (ortho-phthalates) | X | x | x | x | x | x | |
| Trimellitates | x | X | x | ||||
| Aliphatic dibasic esters *4 | X | ||||||
| Meta- and terephthalates; DINCH *1,2 | X | X | X | ||||
| Benzoates *1 | X | X | X | ||||
| Sulfonates *1 | X | X | X | ||||
| Citrates *1 | X | X | X | ||||
| Polyesters | X | X | |||||
| Epoxides | x | x | X | ||||
| Phosphates | X | ||||||
| Extenders (chlorinated paraffins, etc.) *3 | X | ||||||
X denotes primary performance function; x denotes other performance functions (as used in Krauskopf and Goodwin, 2005).
*1: In Krauskopf and Goodwin (2005) these plasticisers were pooled in one group in a similar table, and primary/secondary function distinction may be imprecise here.
*2: Designated as "phthalate-like esters" in Krauskopf and Goodwin (2005). Include substances such as DEHT (terephthalate), DEHIP (metaphthalate) and DINCH (cyclohexane counterpart to DINP).
*3: Extenders are low price oils which are used to extend the effect of other plasticisers, but cannot work as plasticisers alone; e.g. chlorinated paraffins.
*4: Includes adipates.
| Plasticiser in PVC, conc. 40% =67 phr in same PVC resin *1 | Shore A hardness *2 | Volatility,% lost, 1 day at 87 °C over activated carbon | Extracted in water, % | Extracted in kerosene (jet fuel, etc.), % |
|---|---|---|---|---|
| Phthalates: | ||||
| DEHP | 69 | 4.5 | 0.01 | 44 |
| DEHP (PVC2)*3 | 73 | 3.6 | 0.02 | 54.7 |
| DBP | 62 | 45.4 | 0.25 | 9.1 |
| BBP | 68 | 7.7 | 0.07 | 3.4 |
| DIDP | 71 | 1.8 | 0.03 | 74 |
| DINP | 73 | 2.1 | 0.07 | 76.7 |
| Non-phthalates: | ||||
| ASE | 72 | 5.3 | 0.03 | 4.8 |
| ATBC | 73 | 17.8 | 0.09 | |
| DEGD (as single substance) | 69 | 5.5 | 0.75 | 3.4 |
| DGD | 71 | 7.9 | 0.45 | 2.9 |
| DEHT (PVC2)*3 | 76 | 1.9 | 0.09 | 70.8 |
| DINA | 72 | 4.1 | 0.14 | 80.4 |
| TXIB | 76 | 23.7 | 2.83 | 5.2 |
| COMGHA*4 | 88.0 | NA | NA | NA |
| DEHP *4 | 90.0 | NA | NA | NA |
*1: phr = parts per hard resin, meaning parts per weight of hard PVC.
*2: A measure for the plasticiser's efficiency in making PVC flexible; the lower the number, the softer the PVC and the more efficient plasticiser.
*3 Measured performance in another PVC resin (the same for DEHP and EHT).
*4 Data from Danisco; at concentration 40 phr; PVC resin type not specified.
3.3 Alternative plasticisers and polymers used in toys and childcare articles
3.3.1 Toys and childcare articles on the Danish Market
Data on alternative plasticisers and polymers have been obtained by direct contact to Danish manufacturers and larger suppliers of toys and childcare articles. A list of contacted companies can be found in Annex 1. Five out of eleven contacted manufacturers/suppliers have responded with information on alternative plasticisers, whereas four report that they do not have any experience with PVC, DEHP, DBP, BBP or alternatives. All manufacturers have been asked to provide examples of own polymer products containing plasticisers and to specify the applied polymers and plasticisers. Furthermore they were requested information on the technical and economical applicability of the alternative plasticisers.
The results are based on statements by three large companies on the Danish marked for toys and childcare articles. Table 3.4 shows the plasticisers used in PVC products by these three companies.
| Plasticiser | CAS No | Company I | Company II | Company III |
|---|---|---|---|---|
| DINA | 33703-08-1 | X | ||
| TMP | 77-99-6 | X | ||
| ATBC | 77-90-7 | X | X | X |
| DEHT | 6422-86-2 | X | X | X |
| DINCH | 166412-78-8 | X | X | X |
| LG-Flex BET | 610787-76-3 | X | ||
| Mesamoll (ASE) | 91082-17-6 | X |
Chemical name of plasticiser:
DINA: diisononyl adipate,
TMP (trimethylolpropane): 2,2-dihydroxymethybutanol, 2-ethyl-2-hydroxyl-1,3-propanediol,
ATBC: acetyl tributyl citrate,
DEHT: 1,4-benzenedicarboxylic acid, di(2-ethylhexyl) ester,
DINCH: di(isononyl) cyclohexane-1,2-dicarboxylate,
LG-Flex BET: trimethylolpropane, mixed triesters and diesters with benzoic acid and 2-ethylhexanoic acid,
Mesamoll: alkyl sulfonic acid ester of phenol (ASE).
Table 3.5 shows examples of product types and currently applied plasticisers from one of the large toy companies on the Danish marked.
| Product | Part | Alternative plasticiser in PVC | Substituted plasticiser |
|---|---|---|---|
| Dolls | Head | DINA | DEHP or DINP |
| Dolls | Head | ATBC | |
| Inflatable toys | All | DEHT | |
| Inflatable toys | All | TMP | |
| Inflatable toys | All | DINCH | |
| Small vehicles | No information | DINCH |
Chemical name and CAS No: DINA: diisononyl adipate, CAS No 33703-08-1, ATBC: acetyl tributyl citrate, CAS No 77-90-7, DEHT: 1,4-benzenedicarboxylic acid, di(2-ethylhexyl) ester CAS No 6422-86-2, TMP: 2,2-dihydroxymethybutanol, CAS No 77-99-6, DINCH: di(isononyl) cyclohexane-1,2-dicarboxylate, CAS No 166412-78-8.
Experiences with alternative plasticisers
The restriction of the use of DEHP, DBP, BBP, DINP, DIDP and DnOP in toys and childcare articles has challenged the companies on the Danish marked on toys and childcare articles to find alternative solutions for product manufacturing. The companies contacted in this study have various experiences with substitution and some of these are described in the following.
In 2005, a company switched to the alternative ATBC (Citroflex A-4) for all toys for children under 3 years and those of any age which are designed to go to the mouth. This particular plasticiser had been given a favourable opinion by the CSTEE for use in toys. However it suffered from a variety of technical drawbacks when compared with DINP. For instance ATBC would not take decoration and it had high migration into adjacent materials leading to swelling and splitting. There was consequently a need for changes of tools and it had a relatively high cost. The work on alternatives to ATBC suffered technical set backs as the company discovered that alternatives could give rise to mal-odours and poor colour matching in the final PVC. Three potential replacements for DINP were identified: ATBC, DINCH and DEHT. These could be blended in a variety of combinations to achieve softened PVC that performed to the required standards of safety and reliability. These blends could be used in many cases as one-to-one replacements for DINP so major changes to designs and tooling were not necessary.
According to a Danish manufacturer, when the Danish ban came into force, the price of products for the company raised by approximately 50% because the international manufacturers had to produce special deliverances to the Danish marked without phthalates. After the restrictions comprised the entire EU, the prices dropped again. The company estimates that the ban has resulted in a remaining increase in prises of approximately 10-20% because the alternative substances generally are more expensive even after the preliminarily reduced costs related to changing the production.
3.3.2 Toys and childcare articles on the Dutch market
In addition to the information obtained from manufacturers and suppliers of toys and childcare articles in Denmark, data on plasticisers and polymers in toys have been provided by the Dutch Food and Consumer Product Safety Authority, under the Dutch Ministry of Agriculture. The Dutch Food and Consumer Product Safety Authority has investigated plastic softeners in toys and childcare articles for several years.
During May 2007 in total 200 samples of soft plastic toys and 12 samples of soft childcare articles were acquired by The Dutch Food and Consumer Product Safety Authority from the local retail market. These included bath toys, bouncy balls and inflatable aquatic toys as well as bibs, changing table pillows and seats of high chairs. At least 96 brands were represented. Regulated phthalates were analysed quantitatively, whereas other plasticisers, phthalates as well as alternatives, were analysed qualitatively only. The surveys conducted the other years included analyses on regulated phthalates only.
Most examined samples (67%) were made of PVC. Of the examined samples, toys as well as childcare articles, 41% exceeded the legislative limit for DEHP, DBP, BBP, DINP, DIDP and DnOP.
Statistics of the occurrence of non-phthalate plasticisers ranked the alternatives according to the number of products in which they were used as follows (clearly identified substances): DINCH, nonylphenol, TXIB, TBAC, ATBC, DEHTP, DINA, DEHA (see Table 3.6). It is not clear whether the nonylphenol serves as a plasticiser in the plastics. In the literature other applications of nonylphenol in plastics are described, e.g. as an antioxidant.
| Abbreviation | Chemical name | CAS No | Percentage of samples |
|---|---|---|---|
| DINCH | Di(isononyl) cyclohexane-1,2-dicarboxylate | 166412-78-8 | 25 |
| Nonylphenol | 25154-52-3 | 18 | |
| TXIB | Trimethyl pentanyl diisobutyrate | 6846-50-0 | 14 |
| TBAC | Tert-butyl acetate | 540-88-5 | 11 |
| ATBC | Acetyl tributyl citrate | 77-90-7 | 9 |
| DEHTP (same as DEHT) | Di(2-ethyl-hexyl)terephthalate | 6422-86-2 | 7 |
| DINA | diisononyl adipate ester | 33703-08-1 | 6 |
| DEHA | Bis(2-ethylhexyl) adipate | 103-23-1 | 4 |
| DEHS | Dioctyl sebacate | 122-62-3 | 0,6 |
| Bis(2-ethylhexyl)ester | - | 0,6 | |
| DiBA | Diisobutyl adipate | 141-04-8 | 0,6 |
| Other non-specified plasticisers | 0,6 each |
The plasticisers and polymers by product type are shown in Table 3.7. The table includes only the product types for which both phthalate plasticisers and alternatives were found.
| Product | Regulated phthalate | Non-phthalate plasticiser | Polymer |
|---|---|---|---|
| Duck bath toys | DEHP, DINP, DBP | ATBC, DINCH, nonylphenol, TBAC, DINA, TXIB, esters | PVC, PET |
| Other bath toys | DINP, DEHP, DBP | DEHTP, DEP, DIBP, ACTB, DINCH, DEHA, TXIB, nonylphenol, DINA | PVC, TPE, PET, PMMA |
| Balls | DINP, DIDP, DEHP | DEHS, TXIB, | PVC, TPU, PE |
| Dices | DEHP, DINP | DINCH, nonylphenol | PVC |
| Inflatable toys | DINP, DIDP, DEHP | ATBC, DINCH, TXIB, TBAC | PVC |
| Dolls | DEHP, BBP, DINP, DiBA | ATBC, DINCH, DiBA | PVC |
| Puppetry | DINP | DINCH | PVC |
| Strings for making bracelets or key chains | DEHP, BBP, DINP, DBP | ATBC, non-classified phthalates | PVC |
| Toy dinosaurs | DEHP, DINP, DIDP, DBP | TXIB, TBAC, DEP | PMMA, PE/PP, SEBS |
| Toy pig | DINP | Nonylphenol | PVC |
| Swimming bracelets | DEHP, DINP | ATBC, DEHA, DEHT, DINCH, Nonylphenol | PVC, TPE, PET |
| Baby change pad | DEHP, DIDP, DINP | DINCH | PVC |
Compared to similar surveys in 2001, 2004 and 2005 there seems to be only a slight reduction in the use of the six phthalates during the period as shown in Figure 3.2. Notably DBP were found in 30% of the samples in 2004. The percentage had decreased to about 10% in 2007 and further to 1% in 2008 (FCPSA 2008b). BBP is found in only a few percentages of the samples all the year.
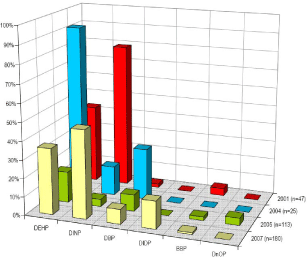
Figure 3.2 Percentage of samples of toys and childcare products from the Dutch retail market that contain 6 specified phthalates (n = number of samples) (FCPSA, 2008a)
3.3.3 Toy s and childcare products on the market in Germany, Austria and Switzerland
A joint study performed in Germany, Austria and Switzerland screened and analysed for all possible plasticisers in 252 samples from 172 toy and childcare products collected in these countries in 2007 (Biedermann-Brem et al., 2008). The study identified the substances shown in Table 3.8 and made detailed statistics of the occurrence, concentrations and mixtures of the identified plasticisers across product groups enabling a ranking of the phthalates as well as the alternatives.
| Abbreviation * | Chemical name |
|---|---|
| ATBC | Acetyl-tributyl-citrate (Citroflex A) |
| DBP | Dibutyl-phthalate |
| DEHA | Di-(2-ethylhexyl)-adipate |
| DEHP | Di-(2-ethylhexyl)-phthalate |
| DEHTP (DEHT) | Di-(2-ethylhexyl)-terephthalate |
| DIBP | Diisobutyl-phthalate |
| DIDP | Diisodecyl-phthalate |
| DINA | Diisononyl-adipate |
| DINCH | Diisononyl-cyclohexane-1,2-dicarboxylate |
| DINP | Diisononyl-phthalate |
| DNOP | Di-(n-octyl)-phthalate |
| ESBO | Epoxidized soy bean oil |
| NPG-EHA-BA | Mixed diesters neopentylglycol-benzoate/2-ethylhexanoate |
| PA | Polyadipate |
| TEHTM | Tri-(2-ethylhexyl)-trimellitate |
| TMP-EHA-BA | Mixed triesters 1,1,1-trimethylol-propane-benzoate/2-ethylhexanoate |
| TXIB | 2,2,4-Trimethyl-1,3-pentanediol-diisobutyrate |
* Abbreviation used in this study in brackets.
The results for each products group involved are shown in Table 3.9, for all plasticiser with occurrence above 4% (main plasticisers in each product). Across all product categories examined, the most important non-phthalate plasticisers used in these products were DINCH, ATBC, DEHT, TXIB and NPG-EHA-BA; see chemicals names in Table 3.8.
Phthalates were detected in 27% of the samples. The phthalates DEHP, DINP, DBP, DIBP, DIDP and DnOP were found. In most cases, the PVC products contained more than one plasticiser; the number of plasticisers in the same product varied between 1 and 5.
3.4 Alternatives recommended by plasticiser producers
In view of the wide spectre of alternative plasticisers available, and in order to get more detailed information on the experience gained on the market with alternatives plasticisers, a number of plasticiser producers were contacted directly with detailed questions on the following issues:
- Proposals for alternative plasticisers for specified traditional DEHP, DBP and BBP applications;
- Level of experience gained on the market with proposed alternatives, according to the manufacturers’ own judgement (4 simplified categories);
- Important processing adjustments, if any, compared to DEHP, DBP and BBP, respectively;
- Limitations in use of alternatives for specified applications;
- Prices of alternatives.
The following plasticiser producers have been contacted based on an initial literature and Internet identification, and asked for this detailed information on their alternatives with focus on non-phthalate alternatives:
- BASF
- Lanxess (formerly Bayer)
- Genovique
- Vertellus (formerly Morflex)
- Eastman Chemicals
- Danisco
- Indo-Nippon Chemicals
- Exxon Mobil
The responses provide an improved indication of, which alternatives are important on the market, and which are emerging or more peripheral. Some producers have not adhered strictly to the market categorisation texts suggested, and here an interpretation was needed to allow for a uniform presentation. Some of the responding companies have not mentioned all their products that otherwise appear to be relevant alternatives to the phthalates judging from their websites. The reasons for this are unknown.
3.4.1 Phthalate alternatives
A number of phthalates are marketed as alternatives to the regulated phthalates and as mentioned above several of the alternatives have a significant market share.
It is highly dependent on the specific application which phthalates are recommended as alternatives. Table 3.10 lists the alternatives recommended by one major manufacturer for five different layers in flooring applications; the main application area for DEHP and BBP.
| Application | Recommended Plasticiser(s) | Advantages |
|---|---|---|
| Top wear layer | Jayflex 77 *1 (replacing BBP/DEHP) *2 | - Lover plastisol viscosity - Improved viscosity stability - Lower cost - Comparable (or better) stain resistance |
| Chemical foam layers | Jayflex DINP or 77 (replacing BBP/DEHP) | - Lower volatility (end use and process) - Lower migration - Lover cost - Equal expansion - Comparable foam structure |
| Impregnation layer | Jayflex DINP (replacing DEHP) | - Lover viscosity - Lover plastisol viscosity - Lower migration - Lower volatility |
| Mechanical foam layer | Jayflex DINP (replacing DEHP at lower BBP concentrations ) | - Lower migration - Lower volatility - Lower cost - Equivalent foam ratio at lower emulsifier concentra- tions |
| Solid PVC layer | Jayflex DINP (replacing DEHP) | - Lower migration - Lower volability - Lower cost |
*1 Diisoheptyl phthalate (DIHP). CAS No. 71888-89-6
*2 DEHP is designated DOP at the webpage.
According to ECPI (2009) 21 different phthalates are in common use. It has been beyond the limits of this study to make a comprehensive assessment of all phthalates that may be used as alternatives to the classified substances. Selected alternatives marketed as alternatives for major applications of the three classified substances are shown in Table 3.11.
| Substances | Abb. | CAS No | recommended alternative to: | Remark |
|---|---|---|---|---|
| Diisononyl phthalate | DINP | 28553-12-0 68515-48-0 |
DEHP; DEHP/DBB | Main all-round alternative to DEHP |
| Diisodecyl phthalate | DIDP | 26761-40-0 271-091-4 |
DEHP | Diisodecyl phthalate (DIDP) is a common phthalate plasticiser, used primarily to soften polyvinyl chloride (PVC). It has properties of volatility resistance, heat stability and electric insulation and is typically used as a plasticiser for heat-resistant electrical cords, leather for car interiors, and PVC flooring. (ECPI, 2009) |
| Diisoheptyl phthalate | DIHP | 71888-89-6 | DEHP/BBP | Marketed as alternative to DEHP/BBP blends in flooring. |
3.4.2 Non-phthalate alternatives
The received responses as regards identification of non-phthalate alternatives to DEHP, DBP and BBP, their application, as well as the level of market experience are presented in Table 3.12, Table 3.13 and Table 3.14 below as recommended by contacted manufacturers.
Besides the results presented in the tables, BASF has reported, that it is their understanding of their customers' experiences that in most, if not all, PVC polymer applications the DEHP functionality may be substituted by applying other plasticisers such as DINP, DPHP (Di- (2-propyl heptyl)phthalate), Hexamoll DINCH or others.
Danisco has further reported that Soft-n-safe (consisting of two substances here designated as COMGHA 1 and COMGHA 2) can be used as alternative to DEHP for various applications.
3.5 Alternative plasticisers applied with processing adjustments
As indicated in the sections above, a large spectre of plasticisers are available, which can influence the final product quality in a number of ways, and additional variety can be obtained by mixing different plasticisers. Besides this, the possibility exists, of accepting that the production conditions or the specifications of the final product are slightly altered, as compared to the present situation. For some products, this may not be critical, whereas for applications requiring specific technical functionalities, such as low temperature flexibility, specific migration thresholds, etc. such deviations may be critical. A closer evaluation of possibilities for substitution of plasticisers with resulting altered performance must be taken in each case.
3.6 Alternatives plasticisers selected for further assessment
Table 3.15 below lists the identified plasticisers and indicates the plasticisers selected for further evaluation in this study (marked in gray).
The selection was based on the following factors:
- Experience with the substances on the marked, based on data collected from suppliers of alternative plasticisers.
- Occurrence as plasticisers in toys and child care articles, based on the studies of plasticisers in these products described above.
- The list should cover the main alternatives for each of the three phthalates DEHP, DBP, and BBP.
- Representation of major substance groups used as plasticisers based on description above.
- Substances with identified significant environment and health effects (CMR or PBT, based on reviews in COWI, 2009 a) were not suggested for further assessment.
3.7 Alternative flexible polymers
Besides the direct alternatives to DEHP, DBP and BBP used as chemicals in various applications, a large number of materials can substitute for flexible PVC in the production of the products.
These alternative materials include, among others, such diverse examples as linoleum and wood for flooring, woven glass fibre and paper for wall coverings, and glass for medical appliances.
The ECHA study on DEHP (COWI, 2009a) concludes that available studies demonstrate that for many applications of DEHP/PVC, alternative materials exist at similar price. Many of the materials seem to have equal or better environment, safety and health performance and cost profiles, but clear conclusions are complicated by the fact that not all aspects of the materials' lifecycles have been included in the assessments.
Due to the wide spectre of products and applications covered, it was decided in this study to limit the considerations for assessment of alternative materials to alternative flexible polymers with characteristics similar to the characteristics of flexible PVC. Based on the information in previous studies, the following flexible polymers are among the principal alternatives to flexible PVC:
- Ethylene vinyl acetate, EVA;
- Low density polyethylene, LDPE;
- Polyolefin elastomers (polyethylene and polypropylene elastomers);
- Several types of polyurethanes (may in some cases be plasticised with phthalates);
- Isobutyl rubber;
- EPDM rubber (may in some cases be plasticised with phthalates);
- Silicone rubber.
3.7.1 Alternative materials suggested for further assessment
A number of studies have been undertaken on replacing PVC with other materials for different applications. For example, an environmental and health assessment of two alternative materials has been conducted by Stuer Lauridsen et al.(2001): PU (polyurethane) and LDPE (low density polyethylene). On the basis of the available data it was not possible to make a full assessment of the materials. This has been the case for several other studies on the subject.
To focus the efforts in this study, it was decided to include an assessment of polyolefin (polyethylene/polypropylene) elastomers as alternatives to flexible PVC - on an overall screening level - of its lifecycle impacts, supplemented by brief description of other flexible polymers mentioned above based on available aggregated reviews.
4 Human health and environmental assessment of alternative plasticisers
- 4.1 ASE (alkylsulphonic phenylester)
- 4.2 ATBC (acetyl, tri-N-butyl citrate)
- 4.3 COMGHA
- 4.4 DEGD (diethylene glycol dibenzoate)
- 4.5 DGD (dipropylene glycol dibenzoate)
- 4.6 DEHT (di-ethylhexyl-terephthalate)
- 4.7 DINA (diisononyl adipate)
- 4.8 DINCH (di-isononyl- cyclohexane-1,2-dicarboxylate)
- 4.9 GTA (glycerol triacetate)
- 4.10 TXIB (trimethyl pentanyl diisobutyrate)
- 4.11 Summary of human health and environmental assessment of alternative plasticisers
Background data for the environmental and human health assessment of the selected alternative plasticisers are presented in Annex 3, which also includes the references to data sources.
4.1 ASE (alkylsulphonic phenylester)
4.1.1 Physico-chemical properties
ASE, a mixture of similar esters of sulfonic acids, phenyl and C10 – C18 alkanes, is a liquid at ambient temperatures. It has low solubility in water (2 mg/L) and low volatility (Vp = 0.01 Pa). It is lipophilic with a log KOW >6.
4.1.2 Human health assessment
Results from pharmacokinetic studies show that a single oral application by gavage of 1000 mg sulfonic acid, C10-21-alkane, Ph esters/kg bw leads to a concentration of 65 μg sulfonic acid, C10-21-alkane, Ph esters/g fat tissue. After 34 days 4μg sulfonic acid, C10-21-alkane, Ph esters/g was still found in the fat tissue. An elimination half-life of 8 days was calculated for the fat tissue. No accumulation was observed in the liver. 20-30% of the dose was excreted in the faeces within 24 hours. When 100 mg of the substance was administered by gavage, the concentration in fat tissue was 22 μg sulfonic acid, C10-21-alkane, Ph esters/g fat tissue after 3 days and 1 μg/g fat tissue after 14 days. No accumulation was observed in the liver.
ASE has low acute toxicity by the oral route with LD50 reported in the range of 26,380 - 31,650 mg/kg bw in the rat. LD50 by the dermal route was found to be > 1,055 mg/kg bw in a rat in a study with no indication of toxicity. ASE was not irritating to rabbit skin when applied to the ears for 24 hours and also not to humans exposed to a saturated patch for 8 hours and followed by a 7 days observation period. Rabbit eyes did not show signs of irritation when exposed to ASE and observed for 7 days.
Subchronic toxicity is studied in a repeated dose 90-day oral toxicity study in the rat. Rats were dosed at 750, 3.000 and 12.000 ppm and a NOAEL was reported at 3,000 ppm corresponding to 228 mg/kg bw in males and 282.6 mg/kg bw in females in spite of significantly dose related absolute and relative liver weights at all dose levels. Effects at the highest dose level included reduced body weight gain, increased feed (females), increased water consumption (males) and increased kidney weight. No accumulation in the liver was observed in repeated dose toxicity studies of shorter duration (28 to 49 days). Elimination half-life for fat tissue was calculated at 15 days.
ASE was negative in Ames test, In vitro Mammalian Cytogenetic Test (OECD 473) and in a HGPRT gene mutation assay, all with and without metabolic activation.
No effects on fertility were seen in what appears to be a three generation reproductive toxicity (fertility) study in rats dosed at 530 mg/kg bw for 6 weeks and observed for 3 months. For the F0-generation, no effects on fertility are reported. The F1-generation is reported to have normal weight gain, normal weight of endocrine organs and normal first oestrus. For the F2 and F3-generation, no effects on fertility and body weight gain are reported. The study dates back to 1956 and details are not available. It is also not clear if a control group has been included in the study. However, due to the limited information about the study and the high degree of uncertainty, it is not possible to draw any conclusions regarding reproductive toxicity from these data.
No other health data was found.
In summary the following profile was identified:
| Acute toxicity | Local effects and sensitisation | ||||
|---|---|---|---|---|---|
| LD50, oral mg/kg bw |
LD50, dermal mg/kg bw |
LC50, inhal.. mg/m³ |
Skin irritation | Eye irritation | Sensitisation |
| 26,380 - 31,650 | > 1,055 | ND | No irritation | No irritation | ND |
| Repeat dose, genotoxicity, carcinogenicity | Reproductive toxicity | |||||
|---|---|---|---|---|---|---|
| Repeat dose, NOAEL mg/kg bw/day |
Genotoxicity | Carcinogenicity | Maternal toxicity mg/kg bw/day |
NOAEL mg/kg bw/day |
Reproductive toxicity | Critical endpoint |
| 228 (m) 282.6 (f) |
Negative | ND | - | 530 | No reliable data | None |
4.1.3 Environmental assessment
Aerobic biodegradation of ASE is found to be 31% in 28 days. Thus, ASE is not readily biodegradable and its log KOW (>6) is indicative of significant potential for bioaccumulation.
Data on effects of ASE on aquatic organisms are few, however, the data in IUCLID indicate low toxicity to fish in OECD acute test with zebrafish (LC0 ≥100 mg/L and LC50 >10,000 mg/L) and similarly a very low toxicity to crustaceans (D. magna); EC >1,000 mg/L and >10,000 mg/L in the two reported tests (OECD acute method). A test with algae (S. suspicatus) gave the same result. It is noted that these test and effect concentrations are far above the reported water solubility of ASE (2 mg/L).
No inhibition of the bacteria Photobacterium phosphoreum was observed at 500 mg/L in one test while in another <20% inhibition occurred at 1.2 g/L. The EC50 for inhibition of activated sludge was >10,000 mg/L (all reported in IUCLID).
The main constituents of sulphonic acids, C10-21-alkane, Ph esters are not considered as PBT. They do not meet the P/vP criteria based on screening data but they meet the screening B criteria. Assessment of ecotoxicity (T) was not carried out during this assessment by the PBT Working Group, PBT List No. 82.
No other environmental effect data have been found.
| Environmental fate | Ecotoxicity | ||||||
|---|---|---|---|---|---|---|---|
| Biodegradation | Bioaccumu-lation | Mobility | Fish | Daphnia | Algae | Micro-organisms | Terrestrial |
| Not readily biodegradable (31% in 28 d) |
Log KOW >6 | ND (Log KOW indicates low mobility) |
LC50 (96 h) >100 mg/L |
EC50 (48 h) >1,000 mg/L |
EC50 (72 h) >10 mg/l |
<20% inhib. at 1.2 mg/l (activated sludge) | ND |
ND = No Data
4.2 ATBC (acetyl, tri-N-butyl citrate)
4.2.1 Physico-chemical properties
ATBC (CAS No. 77-90-7) consists of citrate with three ester bonded butyl groups and one acetyl group bonded to the fourth available oxygen atom. ATBC is a liquid at ambient temperatures and it has a moderate vapour pressure (6.9 Pa at 20 °C). It is sparingly soluble in water (5 mg/L / < 100 mg/L) and quite lipophilic (log KOW = 4.29).
4.2.2 Human health assessment
ATBC is easily absorbed and rapidly metabolised and excreted in the rat. In an absorption study radiolabelled material was recovered at 59 - 70% in urine and cage rinse, 25 - 36% in faeces, 2% expired and 0.36 - 1.26 in tissues and carcass. At least 9 radiolabelled metabolites were found in urine and at least 3 in faeces.
ATBC has low acute toxicity by the oral route in rats with LD50 reported to exceed 30 g/kg bw. No data on acute toxicity by other routes have been found.
ATCB was not irritating to rabbit skin (OECD 404). A study from 1978 in guinea pigs did also not produce irritation, but in an older study from 1955, ATCB produced slight oedema. Patch tests in humans did not produce irritation. ATBC was not irritating to rabbit eyes in a test according to OECD 405. Older studies show slight to moderate irritation. ATCB was not sensitising in guinea pigs or in human volunteers exposed to the substance.
Subchronic toxicity has been studied in several repeated dose toxicity studies of varying quality. In a 90 days study (according to OECD 408) in rats exposed to doses of 100, 300 and 1,000 mg/kg/day, NOAEL was reported at 300 mg/kg bw. All rats survived to scheduled necropsy and no treatment related clinical signs were noted throughout the study. Mean body weights were slightly reduced in both sexes in the high dose group and females in the 300 mg/kg dose group beginning at day 28. These findings were however not statistically significant. Increased liver relative weights for both sexes in the 1,000 mg/kg bw dose group and in the males in the 300 mg/kg bw dose group were not associated with any evidence of hepatotoxicity as evaluated by histopathological examination or clinical chemistry. The only other organ weight change was a slightly increased relative kidney weight for males in the high dose group. NOAEL was based on a few statistically significant differences between the control group and animals administered 1,000 mg/kg bw.
Chronic toxicity was studied in a two-year oral feeding study in rats administered 200, 2000 or 20000 ppm in the diet. Transient reduction in body weight gain was observed in all dose groups from week 5 to 15. Because of this unexplained depression in growth rate, two additional groups of 10 rats received ATCB in concentrations of 200 and 2,000 ppm in the diet for one year. As the findings could not be reproduced it was considered to be an artifact. Most other findings were not statistically significant or not considered treatment related. NOAEL was concluded to be 2,000 ppm (100 mg/kg/day) using a conservative approach as the study lacks in detail and is without GLP.
In a 13-week toxicity study with an In Utero Exposure phase, sensitive reproductive and developmental endpoints were examined. Wistar rats received ATBC in the diet in concentration levels of 100, 300 and 1,000 mg/kg/day. F0 males and females were treated for four weeks prior to mating. F1 male and female offspring were exposed in utero and from birth until start of the 13 week study. F1 offspring selected for the study were then treated for 13 weeks. Based on the results a NOAEL for males was established at 100 mg/kg/day and the NOAEL for females at 300 mg/kg/day. At the highest dose level a slight reduction in body weight gain was seen in both sexes, liver weights were increased and hepatic hypertrophy (common finding at high doses of xenobiotics) was seen in males and females. Weak peroxisome proliferase was measured in males at 300 mg/kg/day and in both sexes at 1,000 mg/kg/day. Slight, reversible variations in urinary composition and plasma electrolyte concentration were considered to be due to adaptation to excretion of high levels of test material and and/or metabolites and were not considered toxicologically significant.
ATBC showed no evidence of mutagenic activity in several Bacterial Reverse Mutation tests (Ames) with and without metabolic activation and also not in Mammalian Cell Gene mutation assays with and without activation. In vitro cytotoxicity was observed in mouse lymphoma cells and less pronounced in HeLa cells. ATCB was not genotoxic in in vivo/in vitrounscheduled DNA synthesis study.
ATBC was administered to rats with the diet in a 2-generation reproductive toxicity study in the following doses: 100, 300 and 1,000 mg/kg/day. NOAEL for both parental animals and offspring was found to be 100 mg/kg/day. No treatment-related clinical observations were noted throughout the study in either F0 or F1 parental animals. Body weights the F1 parental males in the 300 and 1,000 mg/kg/day groups were lower that controls and appeared to be related to treatment. Body weights of the F0 females in the 1,000 mg/kg/day group at the end of pregnancy (gestation days 21 or 22) was significantly lower than control values. No reproductive effects were observed in the F0 and F1 generation and no treatment related abnormalities. Slightly lower body weight and slightly higher mortality was observed among pups in the 300 and 1,000 dose groups, which could be a consequence of reduced water consumption.
No significant treatment related effects on development and embryotoxic effects were observed in a 12 month study in rats.
ATBC showed some signs of neurotoxicity when applied in a 3% acacia to the sciatic nerve in rats and in a 5% suspension of ATBC in 3% gum acacia to the conjunctival sac of the eye of a rabbit. The substance was found to have local anaesthetic action in rabbits and to block neural transmission in rats when placed in contact with a nerve trunk.
In summary the following profile was identified:
| Acute toxicity | Local effects and sensitisation | ||||
|---|---|---|---|---|---|
| LD50, oral mg/kg bw |
LD50, dermal mg/kg bw |
LC50, inhal.. mg/m³ |
Skin irritation | Eye irritation | Sensitisation |
| > 30,000 | > 1,055 | ND | No irritation | No / slight irritation | Not sensitising |
| Repeat dose, genotoxicity, carcinogenicity | Reproductive toxicity | |||||
|---|---|---|---|---|---|---|
| Repeat dose, NOAEL mg/kg bw/day |
Genotoxicity | Carcinogenicity | Maternal toxicity mg/kg bw/day |
NOAEL mg/kg bw/day |
Reproductive toxicity | Critical endpoint |
| 100 | Negative | Negative | NOAEL 100 | 100 (rat) | R and D: No | Decreased bw |
R: Reproductive toxicity; D: Developmental toxicity
4.2.3 Environmental assessment
A number of studies have been conducted to determine the aerobic biodegradability of ATBC. In the modified MITI test with activated sludge inoculum, 80% of the theoretical BOD was reached in 4 weeks. In the static biometer EPA test (EPA 835.3300), ATBC was characterised as readily biodegradable based >60% ThCO2 observed within a 10-14 day window following the lag period. Also in the ASTM D 5338 test, ATBC was found to readily biodegradable as well as ultimately biodedegradable.
A BCF = 250 and a KOC = 1,800 have been calculated for ATBC based on water solubility = 5 mg/L. These values indicate some bioaccumulation potential as well as strong sorption properties i.e. low mobility in soil.
The acute toxicity to fish has been studied using a number of species. The most sensitive end point was found for Pimephales promelas larvae (18 hr) in a 7 day static-renewal test (USPEA Method 1000.0). The LC50 (48 h) was 2.8 mg/L and the LC50 (168 hr) was 1.9 mg/L. In an older (1974) non-guideline flow-through study, the 96 h LC50 for Lepomis macrochirus was estimated at 38-60 mg/L and for the mummichog, Fundalus heteroclitus, to 59 mg/L (both nominal). ECOSAR modelling using P. promelas as model species gave LC50 = 1.67 mg/L.
The water flea Cerodaphnia dubia was used for testing acute toxicity to daphnia using the USEPA 850.1010 method. The 48 h EC50 was determined to be 7.82 mg/L. ECOSAR modelling using D. magna as the model species gave an LC50 = 0.704 mg/L (48 h). The toxicity to algae has not been tested for ATBC, only estimated by ECOSAR with the green alga Selenastrum capricornutum. The 96 hour EC50 was 0.148 mg/L by this method.
| Environmental fate | Ecotoxicity | ||||||
|---|---|---|---|---|---|---|---|
| Biodegradation | Bioaccumu-lation | Mobility | Fish | Daphnia | Algae | Micro-organisms | Terrestrial |
| Ready | BCF = 250 (calculated) | KOC = 1,800 (estimated) | LC50 (48 h) = 2.8 mg/L LC50 (168h) = 1.9 mg/L |
EC50 (48 h) = 7.82 mg/L |
EC50 (96 h) = 0.148 mg/L (calculated) |
ND | ND |
4.3 COMGHA
4.3.1 Physico-chemical properties
12-(Acetoxy)-stearic acid, 2,3-bis(acetoxy)propyl ester is the main constituent (ca. 84%) in a plasticiser consisting of two castor oil derivatives and commonly known as COMGHA (Soft-n-safe). The other main component (ca. 10%) is octadecanoic acid, 2,3-(bis(acetoxy)propyl ester. The CAS number of the mixture, which is a greasy substance, is 736150-63-3. It has a very low volatility (Vp = 0.00000011 Pa at 25 °C), low water solubility (<0.33 mg/L / 7 mg/L) and is highly lipophilic (log KOW = 6.4).
4.3.2 Human health assessment
Toxicokinetic studies on COMGHA show that there is no significant absorption of the material across gastrointestinal epithelium. Based on the results from a 90-days oral toxicity study, it was concluded that there were no marked effects on peroxisomal enzyme activities in liver samples at concentration levels of 0.4%, 1.2% and 3.6% in the diet.
Acute toxicity (OECD 402) of COMGHA by the dermal route has been studied in rat and LD50 found to be > 2,000 mg/kg bw. Other acute toxicity data are not available. COMGHA was not irritating to rabbit skin (OECD 404) and rabbit eyes (OECD 405) and also not a skin sensitizer when studied in a local lymph node assay in mice (OECD 429).
No signs of toxicity were observed in a 28-day repeated dose oral toxicity study (OECD 407) in rats when administered at 3% and 7.5% of the diet/gavage. No effect on the palatability of the diet was observed during the study.
In a 90-day oral toxicity study (OECD 408) with extreme doses: 3, 8.5, 20 ml/kg/day administered by gavage, NOAEL was found to be < 3 ml/kg/day. In a 90-day oral toxicity study (OECD 408) with adequate doses: NOAEL was found to be 5,000 mg/kg/day.
COMGHA was not found mutagenic in Ames test (OECD 471) and no clastogenic activity was seen in the In vitro Mammalian Chromosome Aberration Test (OECD 473). COMGHA was also not mutagenic in In vitro Mammalian Cell Gene Mutation Test (OECD 476).
More studies are planned to investigate genotoxic effects in vivo as well as chronic toxicity, teratogenicity and reproductive toxicity.
In summary the following profile was identified:
| Acute toxicity | Local effects and sensitisation | ||||
|---|---|---|---|---|---|
| LD50, oral mg/kg bw |
LD50, dermal mg/kg bw |
LC50, inhal.. mg/m³ |
Skin irritation | Eye irritation | Sensitisation |
| > 2,000 | ND | ND | No irritation | No irritation | Not sensitising |
| Repeat dose, genotoxicity, carcinogenicity | Reproductive toxicity | |||||
|---|---|---|---|---|---|---|
| Repeat dose, NOAEL mg/kg bw/day |
Genotoxicity | Carcinogenicity | Maternal toxicity mg/kg bw/day |
NOAEL mg/kg bw/day |
Reproductive toxicity | Critical endpoint |
| < 3042 (3 ml) | Negative | ND | ND | ND | ND | - |
4.3.3 Environmental assessment
COMGHA was found to be readily biodegradable when tested by OECD method 301: 98% degradation occurred in 28 days. The log KOW of 6.4 indicates significant bioaccumulation potential and very low mobility in soil.
Acute toxicity to zebrafish was tested using OECD 203 but the LC50 (96 h) could not be determined as it was higher than the solubility of COMGHA. A no observed effect concentration (LC10) after 96 h is stated to be 0.28 mg/L (presumably the highest concentration tested but no detailed information is given).
The acute toxicity to daphnia was EC50 = 0.92 mg/L in the OECD 202 test but COMGHA is, according to the manufacturer, not considered to be acutely toxic at the solubility concentration (<0.33 mg/L). The 72 hour growth inhibition EC50 for algae was 106 mg/L, however a 70-95% loss in test concentration over the test period was observed.
Regarding inhibition of activated sludge respiration (OECD 209), the EC20 (and EC50) was >143 mg/L. No further data on the ecotoxicity of COMGHA is available.
| Environmental fate | Ecotoxicity | ||||||
|---|---|---|---|---|---|---|---|
| Biodegradation | Bioaccumu-lation | Mobility | Fish | Daphnia | Algae | Micro-organisms | Terrestrial |
| Ready | log KOW = 6.4 | "Immobile in soil" | NOEC(LC10) (96h) = 0.28 mg/L |
EC50 (48 h) = 0.92 mg/L |
EC50 (72h) = 106 mg/L |
EC50 >143 mg/L, activated sludge |
ND |
4.4 DEGD (diethylene glycol dibenzoate)
4.4.1 Physico-chemical properties
DEGD is the esterification product of two benzoate groups with diethylene glycol. It has CAS No. 120-55-8. DEGD becomes liquid at temperatures in the range 24-33 °C and has a very low volatility (Vp = 0,000017 Pa at 25 °C). It has a rather low water solubility of 38.3 mg/L and it is moderately lipophilic with a log KOW of 3.0-3.2.
4.4.2 Human health assessment
Metabolism of DEGD was studied in Sprague-Dawley CD rats after single oral doses of 50 mg/kg (low level) and 750 mg/kg (high level). Almost all of single oral doses of 50 and 750 mg/kg of DEGD administered to the rats were adsorbed, metabolized and excreted in the urine within 24 hours of administration. DEGD was metabolized via hydrolysis of the ester bonds to benzoic acid. The free acid was then conjugated with either glycine (major pathway) or glucuronic acid (minor pathway) prior to excretion.
DEGD has low acute toxicity by the oral route in rats with LD50 reported at 4,198 mg/kg bw (OECD 401). Dermal LD50 in rats was found to be > 2,000 mg/kg bw (OECD 402). An acute inhalation toxicity study in the rat was conducted with DEGD resulting in an LC50 > 200 mg/L (4 h).
No dermal reaction was reported following a single semi-occlusive application of DEGD to intact rabbit skin for 4 hours. A single instillation of DEGD into the eye of the rabbit elicited transient very slight conjunctival irritation only. No allergic skin reaction was reported in guinea pigs after repeated skin contact (intradermal and topical) using the Magnusson and Kligman method.
Subchronic toxicity was studied in a repeated dose 13 week oral toxicity study in the rat (OECD 408). Animals received DEGD in the diet in concentration levels of 250, 1000, 1750 or 2500 mg/kg/day. A NOAEL of 1,000 mg/kg bw was established based on the results of the study. There were no findings of toxicological importance at a dosage of 1,000 mg/kg/day or below. In animals receiving 1,750 or 2,500 mg/kg/day, there was an adverse effect on bodyweight gain, changes in clinical pathology parameters and an increased incidence/degree of haemosiderosis in the spleen. In addition, at 2,500 mg/kg/day, a few treatment-related clinical signs were evident, minimal periportal hepatocyte hypertrophy was noted in both sexes. When selected animals previously receiving 2,500 mg/kg/day were maintained off-dose for 4-weeks, all treatment related changes showed evidence of recovery or recovered completely.
No effects were reported in dogs administered up to 300 mg/kg/day of DEGD in their diet for 90 days.
DEGD did not demonstrate mutagenic potential in bacterial (Ames test, OECD 471/2) or mammalian cell (mouse lymphoma cells, OECD 476) systems with and without metabolic activation. No reponse considered to be indicative of clastogenic activity was observed in a In-vitro Mammalian Chromosome Aberration Test in CHL cells (OECD 473).
Prenatal developmental toxicity of DEGD (purity 97.67%) in rats was studied in a test according to US EPA 870.3700 Harmonized Guideline (corresponding to OECD 414). Animals were administered doses of 250, 500 and 1,000 mg/kg/day in the diet. NOEL for maternal toxicity was found to be 1,000 mg/kg/day. At 1,000 mg/kg/day, there were no detectable signs of maternal toxicity; there were no maternal deaths and all females had a live litter at sacrifice. NOAEL for prenatal development was found to be 500 mg/kg/day. A small number of foetuses with cervical ribs was seen at 1,000 mg/kg/day, but not considered indicative of substantial disturbance of morphological development. NOEL for foetal growth and development was 250 mg/kg/day. At 1,000 mg/kg/day mean foetal weights, and consequently litter weight were slightly lower than the control, combined with foetal weight and female foetal weight attaining statistical significance. At 1,000 mg/kg/day 4 foetuses showed cervical ribs, this incidence being higher than the concurrent control and marginally outside the current background control data. Although the incidence of this finding was relatively low, it is considered that a treatment relationship could not be ruled out.
Reproductive toxicity of DEGD (purity 97.67%) in rats was studied in a 2-generation test according to OECD 416 and at dose levels of 1000, 3300 or 10000 ppm in the diet. corresponding to 50, 165 and 500 mg/kg bw/day based on standard conversion factors. There were no obvious toxicological effects of treatment for the two generations on the general condition of the parental animals although a slight disturbance in the pattern of maternal weight change was noted at 10,000 ppm in both generations and at 3,300 ppm in the Fl generation. There was no effect on fertility and reproductive performance at any of the dietary inclusion levels in either generation. Litter parameters at birth of the Fl and F2progeny and their survival to weaning showed no apparent detrimental effects of treatment. However, for the F2 offspring at 10,000 ppm there was a reduction in weight gain from birth to weaning. No abnormal findings were apparent at necropsy of the FO or Fl parental animals, the post weaned unselected Floffspring or the F2 offspring. Organ weight assessment of the FOand Fl parent animals did not suggest any adverse effects on any organs. Assessment of spermatogenesis and histopathology in both parental generations showed that there were no injurious effects on the testes or other reproductive organs. Furthermore, detailed histopathological examination of the tissues from both sexes in both generations did not reveal any adverse effects of treatment. The only possible effect of treatment detected at assessment of organ weights from Fl and F2 offspring was lower absolute and bodyweight relative spleen weights among F2 males and females compared with controls. The evidence from this study suggested that a dietary concentration of 10,000 ppm (500 mg/kg bw/day) should be considered as the NOAEL for the FO and Flparent animals. The NOAEL for the developing offspring is considered to be 3,300 ppm (165 mg/kg bw/day). The NOEL for reproductive parameters is considered to be 10,000 ppm (500 mg/kg bw/day).
Evaluation of estrogenic activity at doses of 500, 1000, 1500 or 2000 mg/kg/day for 7 days by oral gavage in ovariectomized adult Spraque-Dawley (CD) rats using vaginal cornification and the uterotrophic response as the endpoints demonstrated that DEGD did not exhibit estrogenic activity up to and including the maximally tolerated dose.
In summary the following profile was identified:
| Acute toxicity | Local effects and sensitisation | ||||
|---|---|---|---|---|---|
| LD50, oral mg/kg bw |
LD50, dermal mg/kg bw |
LC50, inhal.. mg/m³ |
Skin irritation | Eye irritation | Sensitisation |
| 4,198 | > 2,000 | > 200mg/L | No irritation | Slight irritation | Not sensitising |
| Repeat dose, genotoxicity, carcinogenicity | Reproductive toxicity | |||||
|---|---|---|---|---|---|---|
| Repeat dose, NOAEL mg/kg bw/day |
Genotoxicity | Carcinogenicity | Maternal toxicity mg/kg bw/day |
NOAEL mg/kg bw/day |
Reproductive toxicity | Critical endpoint |
| 1,000 | Negative | ND | 1,000 | 500 250 (NOEL) |
R: No D: Yes |
Foetal bw Cervical ribs |
4.4.3 Environmental assessment
DEGD is found to be readily biodegradable (93% of ThOD in 28 days) in the modified Sturm test (OECD 301B) while in the Closed Bottle Test (OECD 301D) the BOD5/COD ratio was only 0.32 (>0.5 required for ready biodegradability). A KOC of 540 indicates rather low mobility of DEGD in soil, and a moderately high calculated BCF of 120 indicates some bioaccumulation potential.
The aquatic toxicity of DEGD is quite uniform in short term/acute OECD tests between the three main standard groups of test organisms; fish, crustaceans and algae. Thus, the acute (96 h) LC50 to fish (Pimephales promelas) is 3.9 mg/L, while the EC50 (48 h) for daphnia is 6.7 mg/L and the 72 hours growth rate-based EC50 for algae (Seleneastrum capricornutum, now known as Pseudokirchneriella subcapitata) is 11 mg/L. This could indicate a non-specific mode-of-action of DEGD.
The acute toxicity to earthworm (Eisenia foetida) (14 days) was found to be >1,000 ppm while the inhibitory effect (IC50) on the bacterium Pseudomonas putida could not be determined specifically but only be stated as higher than the highest testable concentration of 10 mg/L. Activated sludge respiration was not inhibited at 100 mg/L.
| Environmental fate | Ecotoxicity | ||||||
|---|---|---|---|---|---|---|---|
| Biodegradation | Bioaccumu-lation | Mobility | Fish | Daphnia | Algae | Micro-organisms | Terrestrial |
| Ready | BCF = 120 (calculated) | KOC = 540 (calculated) | LC50 (96 h) = 3.9 mg/L |
EC50 (48 h) = 6.7 mg/L |
EC50 (72 h) = 11 mg/L |
EC50 >10 mg/L (P. putida) NOEC ≥100 mg/L, activated sludge |
LC50 (14 d) >1,000 mg/kg (earthworm) |
4.5 DGD (dipropylene glycol dibenzoate)
4.5.1 Physico-chemical properties
DGD, CAS No. 27138-31-4, is the esterification product of two benzoate groups with dipropylene glycol. DGD is a liquid and is quite similar to DEGD except for two extra methyl groups. It is a substance with low volatility (Vp = 0.00016 Pa at 25 °C), a quite low water solubility of 8.9-15 mg/L and relatively lipophilic character with a log KOW of 3.9.
4.5.2 Human health assessment
Studies show that DGD is rapidly metabolised and excreted from the body and not accumulated in rats. 70% was excreted in the urine within 48 hours of administration as hippuric acid and about 10% was observed in faeces. Half-life of radiocarbon in the blood was 3 hours and for other organs 2-15 hours.
DGD has low acute toxicity by the oral route in rats with LD50 reported at 3,914 mg/kg bw (OECD 401). Dermal LD50 in rats was found to be > 2,000 mg/kg bw (OECD 402). An acute inhalation toxicity study in the rat was conducted with DGD resulting in an LC50 > 200 mg/L (4 h).
No dermal irritation was reported following a single semi-occlusive application of DGD to intact rabbit skin for 4 hours (OECD 404). A single instillation of DGD into the eye of the rabbit elicited transient very slight conjunctival irritation only (OECD 405). No allergic skin reaction was reported in guinea pigs after repeated skin contact (intradermal and topical) using the Magnusson and Kligman method (OECD 406).
Subchronic toxicity was studied in a repeated dose 13 week oral toxicity study in the rat (OECD 408). Animals received DGD in the diet in concentration levels of 250, 1000, 1750 or 2500 mg/kg/day. A NOAEL of 1,000 mg/kg bw (or below) was established based on the results of the study. A few minor intergroup differences were noted at 1,000 mg/kg/day but were insufficient to be of toxicological importance. Higher dosages of 1,750 or 2,500 mg/kg/day were tolerated but the adverse effect on bodyweight was more pronounced, there were increases in circulating enzyme activities, low grade hepatocyte hypertrophy and an increased incidence and degree of hemosiderosis in the spleen in one or both sexes. At 2,500 mg/kg/day, an increased incidence of minimal epithelial hyperplasia was noted in the caecum. When selected animals previously receiving 2,500 mg/kg/day were maintained off dose for 4 weeks, all treatment related effects showed evidence of recovery or recovered completely.
DGD did not demonstrate mutagenic potential in bacterial (Ames test, OECD 471/2) or mammalian cell (mouse lymphoma cells, OECD 476) systems with and without metabolic activation. No response considered to be indicative of clastogenic activity was observed in a In-vitro Mammalian Chromosome Aberration Test in CHL cells (OECD 473) with and without activation.
Prenatal developmental toxicity of DGD (purity 94.84%) in rats was studied in a test according to US EPA 870.3700 (corresponding to OECD 414). Animals were administered doses of 250, 500 and 1,000 mg/kg/day in the diet. NOEL for maternal toxicity was found to be 1,000 mg/kg/day. At 1,000 mg/kg/day, there were no detectable signs of maternal toxicity; there were no maternal deaths and all females had a live litter at sacrifice. NOAEL for prenatal development was found to be 500 mg/kg/day. A small number of foetuses with cervical ribs was seen at 1,000 mg/kg/day. NOEL for foetal growth and development was 250 mg/kg/day. There were no effects of treatment on prenatal survival or growth. At 1,000 mg/kg/day, treatment was associated with a small but definite increase in the number of foetuses with cervical ribs.
Reproductive toxicity of DGD (purity 94.84%) in rats was studied in a 2-generation test according to OECD 416 and at dose levels of 1000, 3300 or 10000 ppm in the diet corresponding to 50, 165 and 500 mg/kg bw/day based on standard conversion factors. There were no obvious toxicological effects of treatment for the two generations on the general condition of the parental animals or on their fertility and reproductive performance.
Litter parameters at birth of the Fland F2 progeny and their survival to weaning showed no apparent detrimental effects of treatment. However, in both the Fland F2 offspring at 10,000 ppm there was a slight reduction in weight gain during days 14-21 of age and this finding may be linked to the transition to direct exposure to the test material as the offspring weaned onto solid diet at the same dietary inclusion levels as their parents.
No abnormal findings were apparent at necropsy of the FO or Fl parental animals, the post weaned unselected Fl offspring or the F2 offspring. Organ weight assessment of the FO and Fl parent animals did not suggest any adverse effects on any organs. Assessment of spermatogenesis and histopathology in both parental generations showed that there were no injurious effects on these testes or other reproductive organs. Furthermore, detailed histopathological examination of the tissues from both sexes in both generations did not reveal any adverse effects of treatment. Regarding survival and growth of the offspring, there were no unequivocal adverse effects. However, a slight reduction in bodyweight gain during days 14 to 21 (F1 and F2), likely due to the neonatal consumption of the dam’s treated diet, and a slight reduction in spleen weights only observed in the F2 generation are of questionable toxicological relevance. The evidence from this study suggested that a dietary concentration of 10,000 ppm (500 mg/kg bw/day) should be considered as the NOEL for the F0 and F1 parent animals. The NOAEL for survival and growth of the offspring is considered to be 10,000 ppm (500 mg/kg bw/day).
Evaluation of estrogenic activity in a uterotrophic Assay at doses of 500, 1000, 1500 or 2000 mg/kg/day for 7 days by oral gavage in ovariectomized (ovaries removed and no natural source of oestrogen) adult Spraque-Dawley (CD) rats using vaginal cornification and the uterotrophic response as the endpoints demonstrated that DGD did not exhibit estrogenic activity up to and including the maximally tolerated dose.
In summary the following profile was identified:
| Acute toxicity | Local effects and sensitisation | ||||
|---|---|---|---|---|---|
| LD50, oral mg/kg bw |
LD50, dermal mg/kg bw |
LC50, inhal.. mg/m³ |
Skin irritation | Eye irritation | Sensitisation |
| 3,914 | > 2,000 | > 200mg/L | No irritation | Slight irritation | Not sensitising |
| Repeat dose, genotoxicity, carcinogenicity | Reproductive toxicity | |||||
|---|---|---|---|---|---|---|
| Repeat dose, NOAEL mg/kg bw/day |
Genotoxicity | Carcinogenicity | Maternal toxicity mg/kg bw/day |
NOAEL mg/kg bw/day |
Reproductive toxicity | Critical endpoint |
| < 1,000 | Negative | ND | 1,000 (NOEL) | 500 250 (NOEL) |
R: No D: Yes |
Foetal bw Cervical ribs |
D: Developmental
4.5.3 Environmental assessment
DGD is found to be readily biodegradable (85% of ThOD in 28 days) in the modified Sturm test (OECD 301B) while in the Closed Bottle Test (OECD 301D) the BOD5/COD ratio was only 0.29 (>0.5 required for ready biodegradability). 75% was degraded after 120 days in an anaerobic biodegradation test with a pass level of 60% (US EPA 796.3140, corresponding to OECD 311) and therefore considered to be ultimately biodegradable under anaerobic conditions. The log KOW of 3.9 indicates some bioaccumulation potential and, at the same time, a likely low mobility in soil.
The LC50 of fish (P. promelas) exposed to DGD for 96 hours was found to be 3.7 mg/L (OECD 203), the 48 hour EC50 for daphnia 19.3 mg/L (OECD 202), and the 72 hours growth rate-based EC50 for algae (Seleneastrum capricornutum, now known as Pseudokirchneriella subcapitata) is 11 mg/L. The corresponding 72 h NOEC was 1.0 mg/L. These quite uniform values across three main taxonomic groups could indicate a non-specific mode-of-action of DGD.
The acute toxicity of DGD to earthworm (Eisenia foetida) (14 days) was found to be >1,000 ppm while the inhibitory effect (IC50) on the bacterium Pseudomonas putida could not be determined specifically but only be stated as higher than the highest testable concentration of 10 mg/L. Activated sludge respiration was not inhibited at 100 mg/L.
| Environmental fate | Ecotoxicity | ||||||
|---|---|---|---|---|---|---|---|
| Biodegradation | Bioaccumu-lation | Mobility | Fish | Daphnia | Algae | Micro-organisms | Terrestrial |
| Ready Ultimately biodegradable under anaerobic conditions |
Log KOW = 3.9 | ND | LC50 (96 h) = 3.7 mg/L |
EC50 (48 h) = 19.3 mg/L |
EC50 (72 h) = 4.9 mg/L NOEC (72 h) = 1.0 mg/L |
EC50 >10 mg/L (P. putida) NOEC ≥100 mg/L, activated sludge |
LC50 (14 d) >1,000 mg/kg (earthworm) |
4.6 DEHT (di-ethylhexyl-terephthalate)
4.6.1 Physico-chemical properties
DEHT, CAS No. 6422-86-2, is a phthalate ester stoekiometrically equal to DEHP, i.e. phthalate ester bound to two ethylhexyl groups, but with a different spatial structure, because one of the carboxylic groups is placed differently on the benzyl ring ("tere" means tertiary, or third, because the carboxylic group is placed on the third carbon atom counted from the first carboxyl group).
DEHT is a liquid with low volatility (Vp = 0.0029 Pa) and a very low solubility in water; determined to be 0.4 μg/L in a recent (2002) GLP study using the slow-stir method. Previously reported, higher solubilities (in the low or sub-mg/L range) are now believed to be incorrectly determined. The log KOW of DEHT is as high as 8.39.
4.6.2 Human health assessment
DEHT has been shown in both in vitro and in vivo studies to have the potential to undergo complete hydrolysis to yield terephthalic acid and 2-ethylhexanol (2-EH), which are rapidly eliminated. Results of these metabolism studies also indicate DEHT was not well absorbed within the gastrointestinal tract, with 36% of it recovered in the faeces still intact. A study to assess dermal absorption rate indicated that DEHT has a very low potential to penetrate the skin (0.103 µg/cm²/hr), which further limits systemic exposure potential.
DEHT has low acute toxicity by the oral route in rats with LD50 reported at >3,200 mg/kg bw (male rat, no guideline) and >5,000 mg/kg bw (TSCA FHSA Regulations (1979): 16 CFR Part 1500.40). Dermal LD50 in male guinea pigs was found to be > 19,670 mg/kg bw. No deaths occurred following inhalation exposure of mice for 4 hr to "saturated" vapours; however, mucosal irritation, loss of coordination and decreased mobility were noted. Recovery occurred in 24 hours.
DEHT was concluded to be a slight dermal irritant in male guinea pigs with no evidence of percutaneous absorption following a single-dose occlusive dermal application and 24 hours exposure. DEHT produces slight irritation to rabbit eyes in a study using a procedure similar to OECD 405.
In studies with some limitations, no skin sensitization was observed in humans or guinea pigs.
Subchronic toxicity was evaluated in a 90-days repeated dose toxicity study where rats were fed diets containing DEHT in concentrations of 0.1, 0.5 or 1%. Study was conducted in a manner similar to the one described in the U.S. EPA guideline, 799.9310 TSCA. The only significant treatment related difference between controls and treated animals was increased relative liver weight in the 1.0% dose group. NOEL was 0.5% corresponding to approximately 500 mg/kg/day. In a 21-days repeated oral toxicity study in rats NOEL was also 0.5% (≈ 500 mg/kg/day) based on increased relative liver weight in females at 1.0%.
DEHT did not produce mutagenicity in Ames tests (procedure similar to OECD 471) and also no response considered to be indicative of clastogenic activity in doses up to 1,000 nL/mL in a In-vitro Mammalian Chromosome Aberration assays in CHO cells (procedure similar to OECD 473) with and without activation.
DEHT was evaluated for combined chronic toxicity and carcinogenicity. The test substance was administered in the diets of male and female Fischer-344 inbred rats at concentrations of 20, 142, and 1,000 mg/kg/day. Clinical evaluations revealed no treatment-related signs, however, eye opacities (cataracts) occurred frequently in all groups. At 1,000 mg/kg/day, body weights and female liver weights were reduced. There were no consistent reductions in food consumption. There were no treatment-related effects evident from the gross and histopathologic examinations conducted at 6 and 12 months. At 18 months, two basic lesions of the females in the 1,000 mg/kg/day level appear to be associated with treatment. These were hyperplasia and/or transitional cell adenomas of the urinary bladder and adenomas or adenocarcinomas of the uterus.
In another combined chronic toxicity and carcinogenicity study (industry study) in F-344 rats DEHT was administered in doses of 1,500, 6,000 and 12,000 ppm (79, 324 and 666 mg/kg bw/day (m) and 102, 418 and 901 mg/kg bw/day) in the diet. It was concluded that the oral administration of DEHT via the diet was well tolerated at all dose levels. There was no effect upon tumour incidence and therefore the NOEL for tumorigenicity was at least 666 mg/kg bw/day in males and 901 mg bw/day in females. Toxic responses were confined to low weight gain and food conversion efficiency in males and females receiving 6,000 and 12,000 ppm. Consequently the NOEL for chronic toxicity in the study was 1,500 ppm (79 mg bw/day (m) and 102 mg bw/day (f)).
Reproductive toxicity of DEHT in Sprague-Dawley rats was studied in a 2-generation test according to OECD 416 and at dose levels of 3000, 6000 or 10000 ppm in the diet. The NOAEL for reproductive toxicity was 1.0% in the diet (500-700 mg/kg bw/day for males and 800-1,000 mg/kg bw/day for females; highest dose tested), and the NOAEL for parental and offspring toxicity based on reduced body weight gains was 0.3% (150-200 mg/kg bw/day for males and 250-300 mg/kg bw/day for females). Mean maternal body weights and body weight gains were reduced for F0 and F1 females in the 1.0% group throughout pregnancy and decreased mean terminal body weights were noted in F1 males and females given 0.6% or 1.0% test material. The results of this study, in conjunction with the 90-day study which also showed no effect of DEHT on histology of reproductive organs indicate that DEHT has a low potential to induce reproductive toxicity.
Developmental toxicity was evaluated in a dietary study following OECD Test Guideline 414 and at dose levels of 3000, 6000 or 10000 ppm in the diet. The NOEL for maternal toxicity was 0.6% (458 mg/kg/day) and the NOEL for developmental toxicity was 1.0% (747 mg/kg/day; highest dose tested).
The ability of DEHT to induce anti-androgenic like effects in male offspring was assessed by giving pregnant rats 750 mg/kg DEHT by gavage on gestation day 14 until postnatal day (PND) 3. No changes indicative of a feminization effect were induced in male pups. NOEL for maternal toxicity and teratogenicity was 750 mg/kg.
Results of an uterotrophic assay in which immature females were given up to 2,000 mg/kg/day DEHT by gavage on PND 19-21 also indicate that DEHT does not possess estrogenic activity.
In summary the following profile was identified:
| Acute toxicity | Local effects and sensitisation | ||||
|---|---|---|---|---|---|
| LD50, oral mg/kg bw |
LD50, dermal mg/kg bw |
LC50, inhal.. mg/m³ |
Skin irritation | Eye irritation | Sensitisation |
| > 5000 | > 19,670 | No deaths, saturated vapour (mice) | Slight irritation | Slight irritation | Not sensitising |
| Repeat dose, genotoxicity, carcinogenicity | Reproductive toxicity | |||||
|---|---|---|---|---|---|---|
| Repeat dose, NOAEL mg/kg bw/day |
Genotoxicity | Carcinogenicity | Maternal toxicity mg/kg bw/day |
NOAEL mg/kg bw/day |
Reproductive toxicity | Critical endpoint |
| 500 | Negative | Negative | 458 (rat) | 747 (highest dose tested) | R: Low potential D: No |
- |
D: Developmental
4.6.3 Environmental assessment
In a study from 1986, performed according to the EPA aerobic biodegradation guideline, the biodegradability of DEHT was found to be 56% in 28 days, corresponding to a classification as inherently biodegradable. The BCF of 393 (in oysters, determined in an EPA protocol study) indicates a medium potential to bioaccumulate, and the KOC of 2,000 a high sorptivity to soil organic matter.
A 7 days flow-through study with Salmo gairdneri using acetone to enhance solubility of DEHT resulted in an LC50 of ≥ 0.25 mg/L (measured), and a 71 days early life-stage study with the same species gave a NOEC ≥ 0.28 mg/L (measured). The acute toxicity (immobilization) to Daphnia magna was determined in a standard 48 hour static test and found to be EC50 ≥ 1.4 mg/L while the 21 days NOEC (reproduction) for D. magna in a flow-through test was 0.76 mg/L (both measured). Inhibition of growth of Seleneastrum capricornutum(now known as Pseudokirchneriella subcapitata) in the standard 72 hours static test did not occur at the possible test concentrations and the EC50 was therefore just stated to be ≥ 0.86 mg/L.
In the OECD 218 test on sediment- dwelling organisms (larvae of Chironomus riparius), the EC50 after 28 days was determined to ≥ 950 mg/kg while the NOEC for emergence was 180 mg/kg. In a 3 hour activated sludge inhibition test according to OECD 209 the EC50 was found to be higher than 10 mg/L.
| Environmental fate | Ecotoxicity | ||||||
|---|---|---|---|---|---|---|---|
| Biodegradation | Bioaccumu-lation | Mobility | Fish | Daphnia | Algae | Micro-organisms | Terrestrial |
| Inherent | BCF = 393 | KOC = 2,000 | LC50 (7 d) > 0.25 mg/L NOEC (71d) ≥ 0.28 mg/L |
EC50 (48 h) > 1.4 mg/L NOEC (21d) ≥ 0.76 mg/L |
EC50 (72 h) > 0.86 mg/L |
EC50 >10 mg/L, activated sludge |
ND |
4.7 DINA (diisononyl adipate)
4.7.1 Physico-chemical properties
DINA is formed by an adipate (hexanoic acid) ester bound with two C-9 alkanes. The CAS No. is 33703-08-1. It is a liquid at room temperature and it appears to be moderately volatile (Vp < 10 Pa at 20 °C). It has a low water solubility of < 1 mg/L and is extremely lipophilic with a log KOW = 9.24.
4.7.2 Human health assessment
No data on toxicokinetics have been identified.
DINA has low acute toxicity by the oral and dermal route with oral LD50 in rats reported at > 5,000 mg/kg bw and dermal LD50 in rabbits reported at > 3,160 mg/kg bw.
DINA was not irritating to rabbit skin (OECD 404) or rabbit eyes (OECD 405). No allergic skin reaction was reported in guinea pigs after repeated skin contact (intradermal and topical) using the Magnusson and Kligman method (OECD 406).
Subchronic toxicity was evaluated in a repeated dose 13 week oral toxicity study in the rat at dose levels up to 500 mg/kg/day. NOAEL in males was 500 mg/kg/day. Increased relative kidney weight was observed at 500 mg/kg/day but no change in absolute kidney weight and histopathological changes was seen. It was concluded that there were no significant findings at any dose level. In a 13 week oral toxicity study in dogs fed at doses up to 3% for 8 weeks and 6% for 5 weeks, NOAEL was 1% in the diet corresponding to approximately 274 mg/kg/day. Adverse effects at the high dose included decreased body weight and food consumption, increased liver weight, elevated enzyme levels, liver and kidney discoloration, and histopathological changes in the liver and kidneys.
DINA did not produce mutagenicity in Ames tests or mammalian cell (mouse lymphoma cells) systems with and without metabolic activation. No other data on health effects was found. Bridging data gaps and read across to the structural analogue, adipic acid, bis (2-ethylhexyl) ester (C22) (CAS No. 103-23-1) and three other structurally similar alkyl diesters (C12-C32) was suggested in the US EPA HPV programme for reproductive and developmental toxicity. These diesters show no or low potential for reproductive and developmental toxicity.
In summary the following profile was identified:
| Acute toxicity | Local effects and sensitisation | ||||
|---|---|---|---|---|---|
| LD50, oral mg/kg bw |
LD50, dermal mg/kg bw |
LC50, inhal.. mg/m³ |
Skin irritation | Eye irritation | Sensitisation |
| > 5000 | > 3,160 | ND | No irritation | No irritation | Not sensitising |
| Repeat dose, genotoxicity, carcinogenicity | Reproductive toxicity | |||||
|---|---|---|---|---|---|---|
| Repeat dose, NOAEL mg/kg bw/day |
Genotoxicity | Carcinogenicity | Maternal toxicity mg/kg bw/day |
NOAEL mg/kg bw/day |
Reproductive toxicity | Critical endpoint |
| 274 | Negative | ND | 1000 (rat) | - | No data1) | - |
- Data from structurally similar diesters show low potential for reproductive and developmental toxicity.
4.7.3 Environmental assessment
DINA is readily biodegradable, it has been found to degrade by 82% in 28 days in the modified MITI test (OECD 301C) and by > 90% in the EEC manometric respirometric method. No data on mobility in soil have been found, but the extremely high log KOW of 9.24 indicates a very low mobility. The BCF in a 21 day test with D. magna was 1,102-2,031 while in a 35 day test with the blue mussel, Mytilus edulis, a BCF = 11,000 was found. No results for BCF in fish have been identified but an estimate from the USEPA gave a BCF = 3.2.
The tests performed with DINA have all been carried out at concentrations exceeding the aqueous solubility i.e. by the use of solubility enhancing solvents such as DMF or acetone. The acute (96 h) toxicity to fish was determined in a test with Leuciscus idus according to DIN 38412. The LC50 was > 500 mg/L (nominal), which corresponded to > 2.6 mg/L (measured). The 79/831/EEC static acute immobilization test was conducted with Daphnia magna, and an EC50 > 100 mg/L was determined. A 21days NOEC > 100 mg/L was found for effects on reproduction of D. magna when using the OECD 202, part 2 test method. The 72 hour EC50 for algae was > 100 mg/L.
| Environmental fate | Ecotoxicity | ||||||
|---|---|---|---|---|---|---|---|
| Biodegradation | Bioaccumu-lation | Mobility | Fish | Daphnia | Algae | Micro-organisms | Terrestrial |
| Ready | BCF ≥ 1,100 BCF (estimated) = 3.2 |
ND | LC50 (96 h) > 500 mg/L (nominal) LC50 (96 h) > 2.6 mg/L (measured) |
EC50 (48 h) > 100 mg/L NOEC (21d) > 100 mg/L |
EC50 (72 h) > 100 mg/L |
EC50 >10,000 mg/L (P. putida) EC20 > 1,000 mg/L, activated sludge |
ND |
4.8 DINCH (di-isononyl- cyclohexane-1,2-dicarboxylate)
4.8.1 Physico-chemical properties
DINCH (CAS No. 166412-78-8) is the hydrogenated parallel to DINP, with the difference that the ring structure is cyclohexane (a cyclic alkane) instead of a benzene ring (an aromate). It is a colourless liquid at 20 °C with a very low water solubility of <0.02 mg/L, low volatily (Vp = 0.000022 Pa) and highly lipophilic character (log KOW >6.2).
4.8.2 Human health assessment
DINCH is rapidly absorbed after oral administration and readily eliminated. After 24 hours approximately 80% of the radioactivity is excreted, after 48 hours more than 90% is excreted via urine and mainly via faeces. There is no indication of bioaccumulation. The characterisation of metabolites after oral and intravenous administration of DINCH indicates two main pathways: the partial hydrolysis of DINCH to the mono-isonyl ester followed by conjugation to glucuronic acid, which is the most abundant metabolite in bile, or the hydrolysis of the remaining ester bond to yield free cyclohexane dicarboxylic acid, the predominant urinary metabolite.
DINCH has low acute toxicity by the oral (OECD 423) with LD50 in rats reported at > 5,000 mg/kg bw. Dermal LD50 in rabbits (OECD 402) was reported at > 2,000 mg/kg bw.
DINCH was slightly irritating to rabbit skin (OECD 404) with mean scores for erythema of 2.0 in one animal and 1.7 in two animals. DINCH was not irritating to rabbit eyes (OECD 405). There was no evidence of skin sensitisation in guinea pigs in a study according to OECD 406.
DINCH was studied in a 28-days oral repeated dose toxicity study (OECD 407) in rats at concentrations of 600, 3000 and 15000 ppm in the diet. Doses of 15,000 ppm caused changes in clinical chemistry parameters in animals of both sexes. Indications of mild renal function impairment (urinary epithelial cells, elevated serum Na+/K+) were observed in male rats. Female rats showed signs that may be associated with hepatic microsomal enzyme induction, characterised by stimulation of γ-glutamyltransferase synthesis and by increased excretion of bilirubin due to stimulation of phase II reactions. NOAEL was established as 3,000 ppm (318 mg/kg bw/day (males) and 342 mg/kg bw/day (females)) in this study, based on the absence of effects on clinical chemistry parameters at this intake level.
In a 90-days oral repeated dose toxicity study (OECD 408) in rats DINCH was administered in the diet at concentrations of 1500, 4500 and 15000 ppm. NOAEL was established at 107.1 mg/kg bw/day (males) and 389.4 mg/kg bw/day (females) in this study, based on kidney weight changes in both sexes and the appearance of degenerated epithelial cells in the urine of males.
In a combined chronic toxicity/carcinogenicity study (OECD 453) rats were administered doses of 40, 200 or 1,000 mg/kg bw/day in the diet. After 24 months of treatment, dose-related follicular cell hyperplasia and increased number of follicular adenomas were observed in the thyroid glands of male rats administered 200 mg/kg bw/day and in both genders administered 1,000 mg/kg bw/day. The thyroid glands are a target organ for the effects of the notified substance in rats. There was a dose-related increased incidence of follicular adenomas in the thyroid gland of mid and high dose male rats and high dose female rats. However, thyroid effects in rats are potentially secondary effects associated with liver enzyme induction and of limited relevance to humans. Such an indirect mechanism is plausible based on the findings of increased GGT activity and lower serum bilirubin levels in this study, and supported by further studies (see special studies below) on enzyme induction and cell proliferation. NOAEL was established at 40 mg/kg bw/day (males) and 200 mg/kg bw/day (females) based on liver weight changes (both sexes) and kidney weight changes (males).
DINCH did not produce mutagenicity in Ames tests (OECD 471) or in in vitro mammalian CHO cells with and without metabolic activation. No clastogenic activity was seen in a chromosome aberration assay (OECD 473) with and without activation. DINCH was also not found to be clastogenic or aneuploidogenic in a in vivo mouse nucleus test (OECD 474).
Developmental toxicity was examined in rabbits (OECD 414) at doses of 100, 300 or 1,000 mg/kg bw/day in the diet. DINCH did not have any adverse effects on maternal toxicity, gestational parameters or developmental toxicity up to 1,000 mg/kg bw/day. NOAEL was established at 1,000 mg/kg bw/day. In a similar study in rats (OECD 414) with animals dosed at 200, 600 or 1,200 mg/kg bw/day, NOAEL was established at 1,200 mg/kg bw/day due to absence of adverse effects on maternal toxicity and prenatal development. Same conclusions were obtained in a study following elements of OECD 414 and OECD 415 (one-generation reproduction toxicity test) with rats dosed at 750 and 1,000 mg/kg bw/day. NOAEL for maternal and development toxicity was established at 1,000 mg/kg bw/day.
Toxicity to reproduction was studied in a two-generation study with animals dosed at 100, 300 or 1,000 mg/kg bw/day. Under the conditions of this reproduction study, the NOAEL for fertility and reproductive performance was established at 1,000 mg/kg bw/day for F0 and F1generation rats of both genders. The NOAEL for general toxicity was 1,000 mg/kg bw/day (F0 rats of both genders) and 100 mg/kg bw/day for the F1 male and female rats (based on tubular vacuolisation and flaky thyroid follicular colloid). The NOAEL for developmental toxicity (growth and development of offspring) was 1,000 mg/kg bw/day for the F1 and F2 pups.
In summary the following profile was identified:
| Acute toxicity | Local effects and sensitisation | ||||
|---|---|---|---|---|---|
| LD50, oral mg/kg bw |
LD50, dermal mg/kg bw |
LC50, inhal.. mg/m³ |
Skin irritation | Eye irritation | Sensitisation |
| > 5,000 | > 2,000 | ND | Slight irritation | No irritation | Not sensitising |
| Repeat dose, genotoxicity, carcinogenicity | Reproductive toxicity | |||||
|---|---|---|---|---|---|---|
| Repeat dose, NOAEL mg/kg bw/day |
Genotoxicity | Carcinogenicity | Maternal toxicity mg/kg bw/day |
NOAEL mg/kg bw/day |
Reproductive toxicity | Critical endpoint |
| 107 | Negative | Negative | 1000 (rat) | 1000 | R and D: No | - |
4.8.3 Environmental assessment
With only 41% degradation in the CO2 evolution test (OECD 301B), DINCH cannot be classified as readily biodegradable. The BCF of 189 indicates a moderate bioaccumulation potential but 90% depuration of the substance occurred within 1.6 days. The log KOW of >6.2 indicates high sorption potential.
The acute toxicity to fish was tested with zebrafish using the 96 hour static EC-test method and found to be LC50 >100 mg/L. Similarly, the acute EC50 for daphnia was found to be higher than the highest test concentration in OECD 202 of 100 mg/L. In a 21 days reproduction test (OECD 211) no effects occurred at the highest test level of 0.021 mg/L (measured) and the NOEC was therefore determined to be ≥0.021 mg/L. The rate based 72 hour EC50 for algae (Scenedesmus subspicatus) was found to be >100 mg/L and the corresponding NOEC ≥100 mg/L.
DINCH was found to be virtually non-toxic to activated sludge (EC50 >1,000 mg/L) and to earthworms in the 14 days acute artificial soil test (LC50 >1,000 mg/kg).
| Environmental fate | Ecotoxicity | ||||||
|---|---|---|---|---|---|---|---|
| Biodegradation | Bioaccumu-lation | Mobility | Fish | Daphnia | Algae | Micro-organisms | Terrestrial |
| Not readily biodegradable (41% in 28 d) |
BCF = 189 | ND | LC50 (96 h) >100 mg/L |
EC50 (48 h) >100 mg/L NOEC (21d) ≥0.021 mg/L |
EC50 (72 h) >100 mg/L NOEC (72h) ≥100 mg/L |
EC50 >1,000 mg/L, activated sludge |
LC50 (14 d) >1,000 mg/kg (earthworm) |
4.9 GTA (glycerol triacetate)
4.9.1 Physico-chemical properties
GTA ("Triacetin") is an ester of glycerol and three acetate groups. Its CAS no. is 102-76-1. It is a liquid at room temperature with a relatively low vapour pressure of 0.33 Pa at 25 °C. It has high solubility in water (58,000-70,000 mg/L) and a correspondingly low log KOW of 0.21-0.36.
4.9.2 Human health assessment
GTA is rapidly absorbed following ingestion and metabolised like other shorter-chain triglycerides. Several studies confirmed that GTA is hydrolysed to glycerol and acetic acid by digestive enzymes, particularly lipases, liver or plasma carboesterases. GTA is readily hydrolyzed to free glycerol and acetic acid, when incubated with rat intestine in vitro. The chemical infused in dogs undergoes intravascular hydrolysis and the majority of the resulting acetate is oxidized nearly quantitatively. The substance has been shown to be a source of liver glycogen and when fed in amounts equal in caloric value to 15% glucose it was utilised as efficiently as glucose.
The acute oral and dermal toxicity of GTA is low. In an oral acute toxicity study in rats (OECD 401), a limit dose of 2,000 mg/kg bw caused no mortality and no signs of systemic toxicity during the 14-day observation period. The LD50 in rats by gavage is determined to be >2,000 mg/kg bw for both sexes, and dermal LD50 in rabbits and guinea pigs were >2,000 mg/kg bw. Acute inhalation toxicity is considered to be very low, since the LC50 in an acute inhalation toxicity study in rats was >1,721 mg/m³ for both sexes (OECD 403) and repeated daily exposure of rats to 73,700 mg/m³ produced no sign of toxicity after 5 days.
GTA was not found irritating to rabbit skin and eyes in studies following OECD 404 and 405. GTA did not induce sensitisation in guinea pigs.
In a combined repeat dose and reproductive/developmental screening toxicity test (OECD 422), rats were exposed to 40, 200 or 1,000 mg/kg/day by oral gavage for 44 days from 2 weeks prior to mating for males and for 41 - 48 days from 14 days before mating to day 3 postpartum for females. GTA had no effects on clinical signs, body weight, food consumption, and organ weight or necropsy findings. No histopathological changes ascribable to the compound were observed in either sex. There were no abnormalities in haematological or blood chemical parameters in males. The NOAEL for repeated dose oral toxicity is thus considered to be 1,000 mg/kg bw/day for both sexes.
An inhalation study was conducted in rats given GTA for 90 days at a dose of 249 ppm (2,220 mg/m³). No signs of toxicity were noted during the exposure. The NOAEL is considered to be 249 ppm (2,220 mg/m³) for 90 days. Although the inhalation study is considered to be useful, it does not fully comply with the current testing protocol.
GTA did not induce gene mutation in Ames test at concentrations up to 5,000 ug /plate (OECD 471 and 472). Induction of chromosome aberrations was observed in the Chinese hamster lung cells at the highest concentration (2.2 mg/mL, 10 mM) in the presence of metabolic activation (OECD 473). Because of high toxicity (75%) that might be caused by low pH (4.9) at the end of the treatment, the chromosomal aberration observed might not be biological relevant. Under un-physiological culture condition, such as low pH, it was reported that the frequency of chromosomal aberrations could be increased. Polyploidy was not induced under any of the conditions tested. Results were equivocal, but taking all data into consideration, GTA could be considered to be non-genotoxic.
The combined repeated dose and reproductive/developmental toxicity study in rats at doses of 40, 200 or 1,000 mg/kg bw/day (OECD 422) showed no statistically significant adverse effects on reproductive parameters ( mating index, fertility index, gestation length, numbers of corpora lutea and implantations, implantation index, gestation index, delivery index, parturition and maternal behaviour at delivery and lactation). In addition, there were no significant differences in numbers of offspring or live offspring, the sex ratio, the live birth index, the viability index or body weight. Developmental toxicity, clinical signs of toxicity, and change in necropsy findings were not found in offspring. Therefore, the NOAEL is considered to be 1,000 mg/kg bw/day for parental animals and offspring.
In summary the following profile was identified:
| Acute toxicity | Local effects and sensitisation | ||||
|---|---|---|---|---|---|
| LD50, oral mg/kg bw |
LD50, dermal mg/kg bw |
LC50, inhal.. mg/m³ |
Skin irritation | Eye irritation | Sensitisation |
| > 2,000 | > 2,000 | > 1,721 | No irritation | No irritation | Not sensitising |
| Repeat dose, genotoxicity, carcinogenicity | Reproductive toxicity | |||||
|---|---|---|---|---|---|---|
| Repeat dose, NOAEL mg/kg bw/day |
Genotoxicity | Carcinogenicity | Maternal toxicity mg/kg bw/day |
NOAEL mg/kg bw/day |
Reproductive toxicity | Critical endpoint |
| 1000 | Negative | ND | 1000 (rat) | 1000 | R and D: No | - |
4.9.3 Environmental assessment
The bulk of data on aerobic biodegradability of GTA suggests that the substance is readily biodegradable, e.g. 77% degradation after 14 days based on BOD, 93% after 28 days based on ThCO2 and 94% after 28 days based on TOC (OECD methods 301B, 301C and 301 D). The KOC of 10.5 indicates high mobility in soil (corresponds well with high water solubility and low log KOW), and the calculated BCF = 1.3 implies an insignificant bioaccumulation potential.
The acute toxicity of GTA to fish has been studied on a number of species such as Pimephales promelas, Oryzias latipes, Cyprinus carpio, Brachydanio rerio and Leuciscus idus. In the test with O. letipes, the lethal level was not reached at the highest test concentration of 100 mg/L, and among the other species the LC50 ranged from 165 to 300 mg/L with P. promelas being the most sensitive species (tested with OECD 203, DIN38412 or ISO 7346/2 (conforming to OECD 203)). In a prolonged test with O. latipes the 14 days LC50 was > 100 mg/L (nomimal) (OECD 204).
EC50'ies for acute toxicity to D. magna range from 380 to 811 mg/L with the most sensitive result being obtained with the DIN 39412 Teil 11 test, which conforms to OECD 202. The 21 days NOEC on reproduction of D. magna was 100 mg/L in the OECD 211 test. Inhibition of growth of Seleneastrum capricornutum (now known as Pseudokirchneriella subcapitata) in the standard 72 hours static test (OECD 201) could not be determined but was > 1,000 mg/L. The NOEC (72 h) was determined to be 556 mg/L. The toxicity to bacteria, Pseudemonas putida, was determined using the EN ISO 10712 guideline. A 16 hours NOEC = 3,000 mg/L was determined.
| Environmental fate | Ecotoxicity | ||||||
|---|---|---|---|---|---|---|---|
| Biodegradation | Bioaccumu-lation | Mobility | Fish | Daphnia | Algae | Micro-organisms | Terrestrial |
| Ready | BCF = 1.3 | KOC = 10.5 | LC50 (96 h) = 165 mg/L LC50 (14 d) > 100 mg/L |
EC50 (48 h) = 380 mg/L NOEC (21d) = 100 mg/L |
EC50 (72 h) > 940 mg/L NOEC (72h) = 556 mg/L |
NOEC (16h) = 3,000 mg/L (P. putida) |
ND |
4.10 TXIB (trimethyl pentanyl diisobutyrate)
4.10.1 Physico-chemical properties
TXIB is an ester of the branched alkane trimethyl pentanyl with two butyrate groups. Its CAS no. is 6846-50-0. The vapour pressure is only 0.089 Pa, the water solubility low (1-2 mg/L; 15 mg/L) and the lipophilicity quite high (log KOW is 4.1 or more).
4.10.2 Human health assessment
TXIB was rapidly adsorped, metabolized and excreted. The major route of elimination was urine (47 – 72% total dose) within 5 - 10 days and the majority of this occurring in the first 72 hours. Radioactivity in faeces accounted for 14 – 31% of the dose with elimination being essentially complete by 7 days with the majority isolated after 48 hours. Radiolabeled CO2 was not detected. In total, excretions accounted for 95-99% of the dose. Residual radioactivity of treated animals approached control by two weeks. Identification of metabolites showed the faeces to contain both 2,2,4-trimethyl pentanediol (TMPD) and TXIB-3-14C indicating esterase cleavage of the two isobutyrates. A small potion of the absorbed material in the urine was unchanged TXIB-3-14C while the majority consisted of metabolites consistent with complete cleavage to the glycol (TMPD) parent molecule. Although much of the urinary metabolite was unidentified it does, nonetheless, represent rapidly cleared material.
TXIB has low acute toxicity by the oral route with LD50 in rats reported at > 3,200 mg/kg bw. Dermal LD50 in rabbit (OECD 402) was reported at > 2,000 mg/kg bw. LC50 in rats exposed to 0.12 mg/L or 5.3 mg/L for 6 hours was > 5.3 mg/L.
Skin irritation was studied in guinea pigs and rabbits. In guinea pigs TXIB was slightly irritating to the skin and in rabbits (OECD 404) no irritation was observed. TXIB was not found irritating to rabbit eyes (OECD 405).
TXIB was not found sensitising to skin in guinea pigs in a test following a protocol similar to OECD 406. In a study on human volunteers using a modified Draize procedure , TXIB was found non-irritating ad did not induce any evidence of sensitisation.
TXIB was studied in a 103 days oral repeated dose toxicity study in rats receiving concentrations of 1% and 1.0% in the diet. There was a slight, significant increase in the relative liver weights in the 1.0% group and in the absolute liver weights in the 1.0% male group when compared to controls. NOAEL was established at 0.1%. In another study rats were exposed to the same concentrations for 52 or 99 days (I: 52 days TXIB diet, II: 99 days TXIB diet, III: 52 days TXIB + 47 days control diet or 52 days control diet + 47 days TXIB diet). From the study it appeared that high doses of TXIB cause significant adaptive changes in the rat liver, and these changes are reversible if the animal is returned to normal diet. NOAEL was 0.1%. In dogs (beagles) fed a diet with 0.1, 0.35 or 1.0% TXIB for 13 weeks, NOAEL was established at 1.0% because of no toxicological significant findings related to treatment.
In a combined repeat dose and reproductive/developmental screening toxicity test (OECD 422), rats were exposed to 30, 150 or 750 mg/kg/day. A NOEL at 30 mg/kg/day was established based on effects on liver and kidneys in the higher dose groups. When evaluating the reproductive toxicity, NOEL (Parental) and NOEL (F1 offspring) was established at 750 mg/kg/day, as no effects on reproduction (mating, fertility and oestrus cycle, dams during pregnancy and lactation, pubs after birth) related to treatment were observed.
In another combined study (OECD 421 with additional sperm motility asssessment) TXIB was administered in doses of 91, 276 or 905 mg/kg /day in males and 120, 359 and 1,135 mg/kg/day in females. Statistically significant reproductive effects observed in the high dose group include reduced number of implantation sites, reduced mean litter weights on postnatal (PND) 0, reduced mean number of live pubs on PND 4, decreased mean absolute epididymal sperm counts, and reduced absolute and relative testicular sperm counts. The mean number of live pubs per litter was also reduced on PND 0. NOAEL for reproductive and developmental toxicity was 276 mg/kg/day on males and 359 mg/kg/day in females based on reduced number of implantation sites and reduced mean number of live pubs on PND 0.
TXIB did not produce mutagenicity in Ames tests (Japanese guideline) or in in vitro mammalian CHL cells (Japanese guideline) with and without metabolic activation.
In summary the following profile was identified:
| Acute toxicity | Local effects and sensitisation | ||||
|---|---|---|---|---|---|
| LD50, oral mg/kg bw |
LD50, dermal mg/kg bw |
LC50, inhal.. mg/m³ |
Skin irritation | Eye irritation | Sensitisation |
| > 3,200 | > 2,000 | > 5,3 mg/L (6h) | No / slight irritation | No irritation | Not sensitising |
| Repeat dose, genotoxicity, carcinogenicity | Reproductive toxicity | |||||
|---|---|---|---|---|---|---|
| Repeat dose, NOAEL mg/kg bw/day |
Genotoxicity | Carcinogenicity | Maternal toxicity mg/kg bw/day |
NOAEL mg/kg bw/day |
Reproductive toxicity | Critical endpoint |
| 1000 | Negative | ND | 1000 (rat) | 276 (m) | R: Yes D: No |
Red. no. implantations sites and mean no. of live pubs |
4.10.3 Environmental assessment
TXIB was found to be inherently biodegradable in the OECD 301C test and the BCF (carp) determined to be in the range 5.2-31 (OECD 305C), i.e. a low bioaccumulation potential. The BCF of 4.1 (or more) indicates a low mobility of TXIB in soil.
Acute toxicity of TXIB to fish: LC50 (96 h) = 18 mg/L (OECD 203; Oryzias latipes). In one test (OECD 202) on Daphnia magna the EC50 was found to be 300 mg/L while in another EC50 >1.46 mg/L. The 14 days NOEC (reproduction) for D. magna was determined to 3.2 mg/L in a test basically performed according to OECD principles but apparently not fulfilling all test requirements. The 72 hours biomass-based EC50 for algae (Seleneastrum capricornutum, now known as Pseudokirchneriella subcapitata) has been determined to 8.0 mg/L using the OECD 201 method. The corresponding 72 h NOEC was 5.3 mg/L.
| Environmental fate | Ecotoxicity | ||||||
|---|---|---|---|---|---|---|---|
| Biodegradation | Bioaccumu-lation | Mobility | Fish | Daphnia | Algae | Micro-organisms | Terrestrial |
| Inherent | BCF = 5.2-31 | ND | LC50 (96 h) = 18 mg/L |
EC50 (48 h) >1.46 mg/L NOEC (14d) = 3.2 mg/L |
EC50 (72h) = 8.0 mg/L NOEC = 5.3 mg/L |
ND | ND |
4.11 Summary of human health and environmental assessment of alternative plasticisers
4.11.1 Human health assessment summary
Table 4.11 provides an overview of the toxicological properties of the selected alternatives.
All substances have been tested for acute toxicity for at least one exposure route, sensitisation (except ASE), subchronic toxicity and mutagenicity. All substances except ASE, COMGHA and DINA have been tested for both reproductive and developmental toxicity.
With regard to carcinogenicity only ATBC, DEHT and DINCH have been tested in combined chronic toxicity and carcinogenicity studies. For DEGD, DGD and DEHT estrogenic activity has been tested in a uterotrophic assay without positive response.
Most data used for the evaluation are considered of good quality, i.e. studies following accepted guidelines (OECD or US EPA) or studies considered acceptable at the time they were carried out. For some of the studies little information is available to evaluate the quality. However, key information is obtained from IUCLID data sheets, USEPA or OECD HPV robust summaries. Studies evaluated under the USEPA HPV programme are considered to be reliable without restrictions or reliable with restrictions (Klimisch codes 1 and 2, and restrictions in cat. 2 generally not severe).
From the overview it can be seen that all ten substances have low acute toxicity. With regard to local effects most substances are non-irritating to skin and eyes or only produce slight irritation which would not lead to classification. None of the tested substances show any sensitising potential.
Effects from repeated dose toxicity studies mainly include reduced body weight gain, increased organ weights (liver and/or kidney) and for some substances also changes in clinical chemistry or clinical pathology parameters. However, more serious pathological effects were not observed. Only GTA did not show any adverse effects in repeat dose toxicity studies.
Studies to evaluate the potential for reproductive/developmental toxicity primarily show toxic effects on parents and offspring resulting in effects on body weight or relative organ weights. Effects on foetal growth and development of cervical ribs were observed in prenatal development studies with DEGD and DGD. With regard to DEHT it was concluded that DEHT has a low potential to induce reproductive toxicity. TXIB showed statistically significant reproductive and developmental toxicity in a combined study (repeat dose/reproductive-developmental screening). Effects included reduced number of implantation sites, reduced mean litter weights and reduced mean number of live pubs.
Carcinogenicity has only been evaluated for three substances in combined studies with negative outcome.
Click here to see: Table 4.11 Overview of toxicological properties of selected alternatives
4.11.2 Environmental assessment summary
Table 4.12 summarises the main data on environmental fate (biodegradation, bioaccumulation and mobility) and ecotoxicological effects (fish, daphnia and algae) of the 10 studied phthalate alternatives. The data on effects on bacteria have been omitted in the summary table because the effects were generally negligible at relevant exposure levels, while terrestrial data were so sparse that a comparative evaluation cannot be made for these organisms anyway.
Useful fate data regarding biodegradability (in water) and bioaccumulative properties (either as BCF or log KOW) are available for all alternatives while other fate data are quite variable and incomplete. With regard to ecotoxicological effect data, results from short-term tests with the base-set of organisms - fish, crustaceans and algae - exist for all 10 substances although the duration of some studies deviate from the current OECD standard. The low solubility of many of the phthalate alternatives has rendered it necessary to enhance solubility by means of organic solvents in order to be able to carry out the tests.
Overall, the data obtained are of good quality i.e. they are mostly based on studies performed according to accepted guideline procedures, and the studies have been evaluated (e.g. in the USEPA HPV robust summaries) to be reliable without restrictions or reliable with restrictions (Klimisch codes 1 and 2, and restrictions in cat. 2 generally not severe).
None of the 10 studied alternatives fulfil the criteria for being PBT or vPvB substances.
| Substance | Environmental fate | Ecotoxicity | ||||
|---|---|---|---|---|---|---|
| Biodegradation | Bioaccumu-lation | Mobility | Fish | Daphnia | Algae | |
| ASE | Not readily biodegradable (31% in 28 d) |
Log KOW >6 | ND (log KOW indicates low mobility) |
LC50 (96 h) >100 mg/L |
EC50 (48 h) >1,000 mg/L |
EC50 (72 h) >10 mg/l |
| ATBC | Ready | BCF = 250 (calculated) | KOC = 1,800 (estimated) | LC50 (48 h) = 2.8 mg/L LC50 (168h) = 1.9 mg/L |
EC50 (48 h) = 7.82 mg/L |
EC50 (96 h) = 0.148 mg/L (calculated) |
| COMGHA | Ready | Log KOW = 6.4 | "Immobile in soil" | NOEC(LC10) (96h) = 0.28 mg/L | EC50 (48 h) = 0.92 mg/L |
EC50 (72h) = 106 mg/L |
| DEGD | Ready | BCF = 120 (calculated) | KOC = 540 (calculated) | LC50 (96 h) = 3.9 mg/L |
EC50 (48 h) = 6.7 mg/L |
EC50 (72 h) = 11 mg/L |
| DGD | Ready Ultimately biodegradable under anaerobic conditions |
Log KOW = 3.9 | ND | LC50 (96 h) = 3.7 mg/L |
EC50 (48 h) = 19.3 mg/L |
EC50 (72 h) = 4.9 mg/L NOEC (72 h) = 1.0 mg/L |
| DEHT | Inherent | BCF = 393 | KOC = 2,000 | LC50 (7 d) > 0.25 mg/L NOEC (71d) ≥ 0.28 mg/L |
EC50 (48 h) > 1.4 mg/L NOEC (21d) ≥ 0.76 mg/L |
EC50 (72 h) > 0.86 mg/L |
| DINA | Ready | BCF ≥ 1,100 BCF (estimated) = 3.2 |
ND | LC50 (96 h) > 500 mg/L (nominal) LC50 (96 h) > 2.6 mg/L (measured) |
EC50 (48 h) > 100 mg/L NOEC (21d) > 100 mg/L |
EC50 (72 h) > 100 mg/L |
| DINCH | Not readily biodegradable (41% in 28 d) |
BCF = 189 | ND | LC50 (96 h) >100 mg/L |
EC50 (48 h) >100 mg/L NOEC (21d) ≥0.021 mg/L |
EC50 (72 h) >100 mg/L NOEC (72h) ≥100 mg/L |
| GTA | Ready | BCF = 1.3 | KOC = 10.5 | LC50 (96 h) = 165 mg/L LC50 (14 d) > 100 mg/L |
EC50 (48 h) = 380 mg/L NOEC (21d) = 100 mg/L |
EC50 (72 h) > 940 mg/L NOEC (72h) = 556 mg/L |
| TXIB | Inherent | BCF = 5.2-31 | ND | LC50 (96 h) = 18 mg/L |
EC50 (48 h) >1.46 mg/L NOEC (14d) = 3.2 mg/L |
EC50 (72h) = 8.0 mg/L NOEC = 5.3 mg/L |
GTA (triacetin) appears to be easily biodegradable; it does not have bioaccumulative properties and has very moderate toxicity in the aquatic environment.
DEGD, DGD and DINA also come out rather favourable (if using the estimated BCF of 3.2 for DINA), while ATBC and COMGHA come out negatively despite their degradability because of their aquatic toxicities and bioaccumulative properties. ASE and DINCH both have low acute toxicities to aquatic organisms but are not easily degradable and have high log KOW values. DEHT is also not easily biodegradable and is bioaccumulative but its aquatic toxicity cannot be fully evaluated based on the data available.
4.11.3 Health and environmental assessment overview
A simplified overview of the main toxicological and ecotoxicological properties of the evaluated substances is shown in Table 4.13.
In the table a rough overview of the quality and completeness of data is presented using the scoring system indicated in note 4 to the table.
Click here to see: Table 4.13 Overview of main toxicological and ecotoxicological properties
5 Technical and economical assessment of alternative plasticisers
- 5.1 ASE (Alkylsulfonic phenylester)
- 5.2 ATBC (Acetyl tri-n-butyl Citrate)
- 5.3 "COMGHA1" (12-(Acetoxy)-stearic acid, 2,3-bis(acetoxy)propyl ester)
- 5.4 DEGD (diethylene glycol dibenzoate)
- 5.5 DGD (dipropylene glycol dibenzoate)
- 5.6 DEHT (di-ethyl-hexyl-terephthalate)
- 5.7 DINA (diisononyl adipate)
- 5.8 DINCH (di-isononyl-cyclohexane-1,2dicarboxylate)
- 5.9 GTA (glycerol triacetate)
- 5.10 TXIB (trimethyl pentanyl diisobutyrate)
- 5.11 Summary and discussion of technical and economical assessment of alternative plasticisers
5.1 ASE (Alkylsulfonic phenylester)
ASE is a mixture of similar esters of sulfonic acids, phenyl and C10 – C18 alkanes (mixture CAS 70775–94–10). It is marketed by Lanxess (formerly Bayer) under the product name Mesamoll.
Producer's description (extracts)
The producer Lanxess presents ASE as having the following characteristics (Lanxess, 2009b):
- Outstanding gelling capacity with a large number of polymers including PVC and polyurethanes, resulting in lower processing temperatures and shorter processing times.
- High saponification resistance, especially compared to DEHP, due to ASE's chemical structure; this is especially beneficial for articles which come into contact with water and alkalis.
- Good compatibility with a large number of polymers such as polyurethane (PU), polyvinyl chloride (PVC), natural rubber (NR), styrene-butadiene rubber (SBR), blends of styrene-butadiene rubber and butadiene rubber (SBR/BR), isobutylene-isoprene rubber (IIR), acrylonitrile-butadiene rubber (NBR) and chloroprene rubber (CR)
- Outstanding resistance to weathering and light.
- Good dielectric properties which give plasticised PVC outstanding weldability at high frequencies leading to shorter cycle times than with other plasticisers.
Application and market experience
Lanxess has provided information on application areas for ASE among the traditional DEHP, DBP and BBP applications shown in Table 5.1. The table also indicates the level of market experience in each application area according to Lanxess (2009; interpreted from qualitative text by the report authors). Note that Lanxess has indicated significant market experience for most applications, indicating both general plasticiser characteristics and coverage of several of the special performance characteristics of DBP and BBP.
A Danish study (Nilsson et al., 2002) demonstrated the feasibility of ASE as alternative to phthalates in waterbeds (in the plastisol saturated textile lining), where it is used today.
| Application | Market experience *1 |
|---|---|
| Substituting for DEHP | |
| Polymer applications: | |
| Calendering of film, sheet and coated products | 2 |
| Calendering of flooring, roofing, wall covering | 4 |
| Extrusion of hose and profile | 2 |
| Extrusion of wire and cable | 2 |
| Extrusion of miscellaneous products from compounds | 2 |
| Injection moulding of footwear and miscellaneous | ? |
| Spread coating of flooring | 2 |
| Spread coating of coated fabric, wall covering, coil coating, etc. | 2 |
| Car undercoating | 2 |
| Non polymer applications: | |
| Adhesives/sealant, rubber | 2 |
| Lacquers and paint | 2 |
| Printing ink | 2 |
| Production of ceramics | |
| Substituting for DBP | |
| Plasticiser in PVC | 2 |
| Plasticiser in other polymers | 2 |
| Adhesives | 2 |
| Printing inks | 2 |
| Miscellaneous: | |
| Sealants | 2 |
| PU foam sealants | 2 |
| Nitrocellulose paints | 2 |
| Film coatings | 3 |
| Glass fibre production | |
| Cosmetics | |
| Substituting for BBP | |
| Polymer applications: | |
| General PVC (e.g. for moulded plastic parts) | 2 |
| Plastisol coating, for flooring | 2 |
| Extrusion or spreadcoating: Leather and cloth coating (e.g. for furniture, shoes, bags, suitcases) | 2 |
| Films, calendering (e.g. for packaging, calendered flooring, wall covering, etc.) | 2 |
| Non polymer applications: | |
| Sealants (polysulfide based, polyurethane foam sealants, acrylic based; e.g. for windows, construction etc.) | 2 |
| Coatings and inks (e.g. for car care products, construction, paper, board) | |
| Adhesives (polymer based, e.g. for construction, paper) | 2 |
*1: Market experience categories interpretation: 1) Main alternative on market. 2) Significant market experience. 3) Examples of full scale experience. 4) Pilot/lab scale experience.
Key characteristics
Table 5.2 below describes some key characteristics of ASE as alternative to DEHP, DBP and BBP.
| Parameter | Value | Remarks |
|---|---|---|
| Efficiency( as plasticiser in PVC compared to DEHP)*1 | NA | |
| Price (primo 2009) | €1,75/kg | Lanxess (2009) |
| Price relative to DEHP (≈0.8-1€/kg in 2008/2009; 1€ used for calculations) | 175% | |
| Effective price relative to DEHP | NA | |
| Compatibility/solubility in PVC | Good (Lanxess, 2009, 2009b) | |
| Permanency (migration, evaporation, extraction) | + | High resistance to extraction by saponification (extraction with soap water), (Lanxess, 2009, 2009b) |
| Processability (fusing speed and temperature, viscosity, etc.) | + | Low gelling temperature. Faster gellation/fusing speed than DEHP; lower than BBP and DBP (Wilson, 1995; Lanxess, 2009, 2009b) |
| Limitations in use, if any, noted by supplier | Not suitable for polysulfide based sealants (Lanxess, 2009, 2009b) |
Notes: *1: Effectiveness indicator, also called substitution factor, indicating the concentration of plasticiser in PVC needed, compared to DEHP, to achieve a specified flexibility according to a well defined method. DEHP has substitution factor 1 per definition, while plasticisers needing lower concentration (more effective) have lower values, and plasticisers needing higher concentrations have higher effectiveness value. *2: According to Lanxess, 2009, 2009b. NA = not available
Table 5.3 below shows some performance data for ASE compared to DEHP. As shown, performance parameters are quite close to DEHP's, except for a higher resistance to kerosene (jet-fuel, lipophilic solvent).
| Plasticiser in PVC, conc. 40% =67 phr in same PVC resin | Shore A hardness | Volatility,% lost, 1 day at 87 °C over activated carbon | Extracted in water,% | Extracted in kerosene (jet fuel, etc.),% |
|---|---|---|---|---|
| DEHP | 69 | 4.5 | 0.01 | 44 |
| ASE | 72 | 5.3 | 0.03 | 4.8 |
Wilson (1995) states, that were it not for the higher prices of ASE, it could easily be used as an alternative to the general purpose phthalate plasticisers in a wide range of applications. Its structure gives it some advantages over phthalates for certain processes and aggressive environments. The higher polarity results in faster gelling speed than can be achieved with other plasticisers at similar molecular weight and volatility. ASE has high resistance to degradation from weathering, microorganisms and alkaline media.
Conclusions
Based on the above mentioned, it seems reasonable to conclude that ASE appears - based on technical observations only - to be an actual general plasticiser alternative to DEHP. The producer, Lanxess, has indicated significant market experience for most traditional DEHP, DBP and BBP uses, indicating both general plasticiser characteristics and coverage of several of the special performance characteristics of DBP and BBP.
ASE was reported as used for toys by Danish toy manufacturers (with contract production in China), but was not found in the surveys of plasticisers in toys in the Netherlands and Switzerland.
The significantly higher price currently may, however, likely be an impediment to widespread substitution. The potential for attaining reduced prices with increased production volume has not been investigated by the research made for this study.
5.2 ATBC (Acetyl tri-n-butyl Citrate)
ATBC consists of citrate with three ester bonded butyl groups and one acetyl group bonded to the fourth available oxygen atom, see the structure below. The CAS no. is 77-90-7. It is marketed by Vertellus (formerly Morflex), under the product name Citroflex A-4, and by Jungbunzlauer under the products name CITROFOL® BII.
Citrates (ATBC; acetyl tributyl citrate)
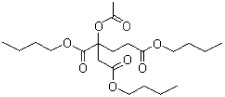
Producer's description (extracts)
Vertellus (formerly Morflex) has characterised ATBC as follows in their sales material (Vertellus, 2009b): ATBC is compatible with PVC resin, as well as with a range of other polymers. ATBC has mostly been used in products used for sensitive purposes such as medical products food contact products and children's toys. It is, however, too extractable to be useful in some of the applications in the medical area where contact with lipids is important. For such uses, the larger molecule n-butyryltri-n-hexyl Citrate is recommended by Vertellus. The higher molecular weight citric acid esters, including ATBC, are effective replacements for di-(2-ethylhexyl) phthalate (DEHP) and di-(2-ethylhexyl) adipate (DEHA). ATBC also has similar characteristics to some of the lower molecular weight phthalates in a variety of polymers. ATBC is widely used in food contact polymers. It provides many improvements over DBP in cellulose nitrate films, including lower volatility, better resistance to yellowing, and better adhesion to metals. ATBC is effective in solution coating both paperboard and foil. It is a good plasticizer for vinyl toys. ATBC Special is developed and recommended for medical articles and similar sensitive applications. It is manufactured in a unique, patented process. A special version for use in pharmaceutical coatings is sold as ATBC, PG.
Application and market experience
Vertellus has provided information on application areas for ATBC among the traditional DEHP, DBP and BBP applications shown in Table 5.4. The table also indicates the level of market experience in each application area according to Vertellus (2009).
| Application | Market experience *1 |
|---|---|
| Substituting for DEHP | |
| Polymer applications: | |
| Calendering of film, sheet and coated products | 3 |
| Calendering of flooring, roofing, wall covering | |
| Extrusion of hose and profile | 3 |
| Extrusion of wire and cable | |
| Extrusion of miscellaneous products from compounds | 2 |
| Injection moulding of footwear and miscellaneous | |
| Spread coating of flooring | |
| Spread coating of coated fabric, wall covering, coil coating, etc. | |
| Car undercoating | |
| Non polymer applications: | |
| Adhesives/sealant, rubber | 2 |
| Lacquers and paint | |
| Printing ink | 2 |
| Production of ceramics | |
| Other applications (added to list by producer) | |
| PVC medical articles | 2 |
| Toy and childcare articles | 1 |
| Substituting for DBP | |
| Plasticiser in PVC | 2 |
| Plasticiser in other polymers | |
| Adhesives | 3 |
| Printing inks | 2 |
| Miscellaneous: | |
| Sealants | 3 |
| PU foam sealants | 4 |
| Nitrocellulose paints | 2 |
| Film coatings | 3 |
| Glass fibre production | |
| Cosmetics | |
| Substituting for BBP | |
| Polymer applications: | |
| General PVC (e.g. for moulded plastic parts) | |
| Plastisol coating, for flooring | |
| Extrusion or spreadcoating: Leather and cloth coating | |
| Films, calendering (e.g. for packaging, calendered flooring, wall covering, etc.) | |
| Non polymer applications: | |
| Sealants (polysulfide based, polyurethane foam sealants, acrylic based; e.g. for windows, construction etc.) | |
| Coatings and inks (e.g. for car care products, construction, paper, board) | 3 |
| Adhesives (polymer based, e.g. for construction, paper) | |
| Nails polish | 1 |
*1: Market experience categories interpretation: 1) Main alternative on market. 2) Significant market experience. 3) Examples of full scale experience. 4) Pilot/lab scale experience.
Note that Vertellus has indicated significant market experience for toys, medical articles, and certain non-polymer applications substituting for both DEHP, DBP and BBP, indicating both general plasticiser characteristics and coverage of some of the special performance characteristics of DBP and BBP.
In a study of alternatives to flexible PVC with phthalates, PVC products plasticised with ATBC were reported to match all of the technical requirements (Postle et al., 2000). Similarly, in a lab test of different alternatives to DEHP for their suitability as plasticisers in PVC for medical uses ATBC was found suitable on all technical parameters tested (Karbæk, 2003).
Key characteristics
According to ECPI, acetyl tributyl citrate is traditionally used in electrical coatings and casings because of its solvating characteristics. It is also used in inks, hair sprays and aerosol bandages (ECPI, 2009).
The table below (from Vertellus, 2009b) compares various characteristics of medical grade PVC with 50 parts by PVC weight plasticiser (= plasticiser concentration in PVC product 33,3% by weight), 2.5 parts by PVC weight stabiliser (Cl/Zn) and 0.25 parts by PVC weight lubricant (stearic acid), milled and pressure died to sheet (mechanical tests by ASTM methods; Vertellus, 2009b). A-4 is ATBC. T4 and Tf are torsion flex indicators at specific conditions (see Vertellus, 2009b).
Table 5.5 Comparison of ATBC (=A-4) with DEHP and DEHA for various parameters, from Vertellus (2009).
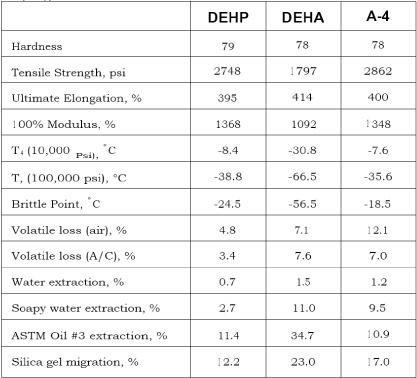
As shown, the mechanical tests and oil extraction for DEHP and A-4 = ATBC are very close, while losses to air and aqueous liquids are a factor 2-3 higher for ATBC. According to the reference, extraction and volatilization properties can be adjusted by adding another plasticiser, for example the less expensive epoxidized soybean oil (ESO).
Table 5.6 describes some key characteristics of ATBC as alternative to DEHP, DBP and BBP.
| Parameter | Value | Remarks |
|---|---|---|
| Efficiency( as plasticiser in PVC compared to DEHP)*1 | Equal to or slightly above 1 judged from Shore A hardness. | |
| Price (primo 2009) | NA | |
| Price relative to DEHP | 300% | ExxonMobil (2009); Karbæk (2003) |
| Effective price relative to DEHP | NA | |
| Compatibility/solubility in PVC | Compatible | |
| Permanency (migration, evaporation, extraction) | Extractability in medical appliances with lipid (fat) contact not found optimal, but losses to oil is found similar to DEHP. Losses to air and aqueous liquids are a factor 2-3 higher than for DEHP (Vertellus, 2009b). | |
| Processability (fusing speed and temperature, etc.) | High solvating | |
| Limitations in use, if any, noted by supplier in data for this study | None noted |
Notes: *1: Efficiency indicator, also called substitution factor, indicating the concentration of plasticiser in PVC needed, compared to DEHP, to achieve a specified flexibility according to a well defined method. DEHP has substitution factor 1 per definition, while plasticisers needing lower concentration (more effective) have lower values, and plasticisers needing higher concentrations have higher effectiveness value. NA = not available
Table 5.7 below shows some performance data for ATBC compared to DEHP, DBP and BBP. As shown, ATBC results in a similar hardness as DEHP and BBP, and volatility and extractability higher than DEHP and BBP, but lower than DBP (DBP is the smallest, most mobile molecule of these phthalates).
| Plasticiser in PVC, conc. 40% =67 phr in same PVC resin | Shore A hardness | Volatility,% lost, 1 day at 87 °C over activated carbon | Extracted in water,% | Extracted in kerosene (jet fuel, etc.),% |
|---|---|---|---|---|
| DEHP | 69 | 4.5 | 0.01 | 44 |
| DBP | 62 | 45.4 | 0.25 | 9.1 |
| BBP | 68 | 7.7 | 0.07 | 3.4 |
| ATBC | 73 | 17.8 | 0.09 |
A Danish toy producing company switched from DINP to ATBC (Citroflex A-4) for all toys for children under 3 years and for toys which are designed to go to the mouth. This particular plasticiser had been given a favourable opinion by the CSTEE for use in toys. However it suffered from a variety of technical drawbacks when compared with DINP. For instance ATBC would not take decoration, it had high migration into adjacent materials leading to swelling and splitting, and there was a need for tooling changes. Development led to the use of a mixture of ATBC, DINCH and DEHT, which could be blended in a variety of combinations to achieve softened PVC that performed to the required standards of safety and reliability with the existing production setup. These blends could be used in many cases as one-to-one replacements for DINP so that major changes to designs and tooling were not necessary.
Conclusions
The performance of ATBC on some parameters seems similar to DEHP, indicating technical suitability for substitution for some purposes. The producer, Vertellus, has indicated significant market experience for toys, medical articles, and certain non-polymer applications substituting for both DEHP, DBP and BBP, indicating both general plasticiser characteristics and coverage of some of the special performance characteristics of DBP and BBP.
ATBC was found in 9% and 10%, respectively, of products analysed in two European studies of large samples of toys and childcare articles. It was also reported as used for toys by Danish toy manufacturers (with contract production in China); used alone it did however not perform adequately in the established toy production setup due to migration to adjacent materials, print resistance, etc. Used in combination with DINCH and DEHT it could be used with no major processing changes.
The higher extractability in aqueous solutions and the higher volatility may reduce the performance of ATBC as a plasticiser in PVC, and could perhaps limit its use for certain applications. Similarly, the extractability in medical appliances with lipid (fat) contact may perhaps limit its use for certain medical applications.
The price of ATBC is significantly higher than the price of DEHP, and this may represent a major impediment for its wider use as alternative to DEHP; DBP and BBP.
5.3 "COMGHA1" (12-(Acetoxy)-stearic acid, 2,3-bis(acetoxy)propyl ester)
12-(Acetoxy)-stearic acid, 2,3-bis(acetoxy)propyl ester is the main constituent (ca. 84%) in a plasticiser consisting of two castor oil derivatives designated as "COMGHA" (by SCENIHR, 2008) and marketed as Soft-n-Safe by Danisco. The other main component (ca. 10%) is octadecanoic acid, 2,3-(bis(acetoxy)propyl ester. Both main substances are shown below (from, SCENIHR, 2008). The remaining 5% are excess glycerine from the production. In the following technical text, this mixed product is therefore described. The CAS number of the mixture is 736150-63-3.
 "COMGHA1" , 12-(Acetoxy)-stearic acid, 2,3-bis (acetoxy)propyl ester;
"COMGHA1" , 12-(Acetoxy)-stearic acid, 2,3-bis (acetoxy)propyl ester;
CAS: 330198-91-9
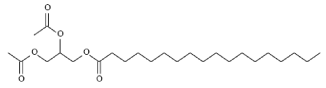 "COMGHA2" , Octadecanoic acid, 2,3-(bis(acetoxy) propyl ester;
"COMGHA2" , Octadecanoic acid, 2,3-(bis(acetoxy) propyl ester;
CAS: 33599-07-4
Producer's description (extracts)
Danisco characterises COMGHA/Soft-n-safe as follows (Danisco, 2009b):
COMGHA is an efficient, one-to-one replacement for most conventional plasticisers, such as phthalates. In tests, the quality, durability and functional properties achieved have proven equivalent to phthalate-based solutions. Not only that, COMGHA can be directly applied, without any further alteration to the formulation or processing.
COMGHA has been tested against traditional plasticisers in many PVC applications. Compared with plasticisers such as DEHP, DINP and DOA, the efficiency and reliability of COMGHA is consistently on top.
The main application areas for COMGHA are:
- Food contact applications
- Plastisol production
- Medical devices
COMGHA has been compared with DEHP in numerous flexible PVC applications that perform a sensitive medical role, including tubing and medical film. The test results show that COMGHA meets all requirements in the extrusion, calendaring and injection moulding applications where it has been evaluated. In medical applications where plasticiser migration is a particular concern, COMGHA demonstrated high extraction resistance in aqueous and oily solvents.
Compared with traditional plasticisers such as DEHP and DINP, COMGHA performs consistently well in applications such as vinyl flooring, wallpaper, shrink wrap film, textile dyes, ink applications, adhesives and sealants. In application tests with toys for young children, COMGHA provided the same level of efficiency as DEHP, when measured according to the Shore A scale.
While COMGHA has a significantly higher molecular weight than DEHP and a comparable molecular weight to TOTM, its efficiency remains on top. This makes it a viable candidate for applications that demand low volatility. TGA analysis has shown that COMGHA is considerably less volatile than DEHP under all conditions. Although the novel plasticiser is more volatile than a high end permanency plasticiser as TOTM, the lower loading level required means overall volatility is reduced.
Application and market experience
Danisco has provided information on application areas for COMGHA among the traditional DEHP, DBP and BBP applications shown in Table 5.8. The table also indicates the level of market experience in each application area according to Danisco (2009). Note that COMGHA still has relative moderate market experience, albeit with many examples of full scale usage and pilot/lab scale tests, and significant market experience in plastisol applications and cosmetics.
| Application | Market experience *1 |
|---|---|
| Substituting for DEHP | |
| Polymer applications: | |
| Calendering of film, sheet and coated products | 3 |
| Calendering of flooring, roofing, wall covering | 3 |
| Extrusion of hose and profile | 3 |
| Extrusion of wire and cable | 3 |
| Extrusion of miscellaneous products from compounds | 3 |
| Injection moulding of footwear and miscellaneous | 3 |
| Spread coating of flooring | 2 |
| Spread coating of coated fabric, wall covering, coil coating, etc. | 3 |
| Car undercoating | |
| Non polymer applications: | |
| Adhesives/sealant, rubber | 4 |
| Lacquers and paint | 4 |
| Printing ink | 3 |
| Production of ceramics | |
| Substituting for DBP | |
| Plasticiser in PVC | 2 |
| Plasticiser in other polymers | 2 |
| Adhesives | 4 |
| Printing inks | 3 |
| Miscellaneous: | |
| Sealants | 4 |
| PU foam sealants | |
| Nitrocellulose paints | |
| Film coatings | |
| Glass fibre production | 4 |
| Cosmetics | 2 |
| Substituting for BBP | |
| Polymer applications: | |
| General PVC (e.g. for moulded plastic parts) | 4 |
| Plastisol coating, for flooring | 3 |
| Extrusion or spreadcoating: Leather and cloth coating (e.g. for furniture, shoes, bags, suitcases) | 2 |
| Films, calendering (e.g. for packaging, calendered flooring, wall covering, etc.) | 3 |
| Non polymer applications: | |
| Sealants (polysulfide based, polyurethane foam sealants, acrylic based; e.g. for windows, construction etc.) | |
| Coatings and inks (e.g. for car care products, construction, paper, board) | |
| Adhesives (polymer based, e.g. for construction, paper) |
*1: Market experience categories interpretation: 1) Main alternative on market. 2) Significant market experience. 3) Examples of full scale experience. 4) Pilot/lab scale experience.
Key characteristics
Table 5.9 below shows selected comparisons from Danisco (2009b) between COMGHA, DEHP and DINP for selected parameters (more parameters are shown in Danisco, 2009b). Note that COMGHA has very similar characteristics as DEHP and DINP.
Table 5.9 shows comparisons of COMGHA, DEHP and DINP for selected parameters.
| Plasticiser (at 40 phr) | Shore A, after 15 sec | Tensile strength, MPa | 100% modulus, MPa | Max. elongation,% |
|---|---|---|---|---|
| COMGHA | 88.0 | 25.0 | 9.1 | 367 |
| DEHP | 90.0 | 22.2 | 8.5 | 320 |
| DINP | 91.5 | 24.1 | 9.3 | 344 |
Table 5.10 describes some key characteristics of COMGHA as alternative to DEHP, DBP and BBP.
| Parameter | Value | Remarks |
|---|---|---|
| Efficiency( as plasticiser in PVC compared to DEHP)*1 | ≈1 | (Danisco, 2009b) |
| Price | 3.5 €/kg | Danisco (2009) |
| Price relative to DEHP (≈0.8-1€/kg in 2008/2009; 1€ used for calculations) | ≈350% | |
| Effective price relative to DEHP | ≈350% | |
| Compatibility/solubility in PVC | Compatible | |
| Permanency (migration, evaporation, extraction) | Much lower extractability than DEHP in acidic water solutions and ethanol/water solutions. Lower extractability in sunflower oil (Danisco, 2009b) | |
| Processability (fusing speed and temperature, viscosity, etc.) | Higher viscosity than DEHP (Danisco, 2009b) | |
| Limitations in use, if any, noted by supplier in data for this study | None noted |
Notes: *1: Efficiency indicator, also called substitution factor, indicating the concentration of plasticiser in PVC needed, compared to DEHP, to achieve a specified flexibility according to a well defined method. DEHP has substitution factor 1 per definition, while plasticisers needing lower concentration (more effective) have lower values, and plasticisers needing higher concentrations have higher effectiveness value. NA = not available
Both substances in COMGHA are derived from castor oil produced from castor beans. According to Danisco, the beans must be handpicked which sets limits to production volume and cost reductions (Danisco, 2009). Research is therefore ongoing to produce the substances from other, more abundant, biological substrates (Buck Jensen, 2009).
Conclusions
According to the producer, Danisco, COMGHA still has relative moderate market experience, albeit with many examples of full scale usage and pilot/lab scale tests, and significant market experience in some plastisol application and cosmetics. The producers found technically good performance on key parameters indicating a potential for substituting for DEHP and perhaps for some traditional DBP/BBP uses.
The significantly higher price than DEHP is a major impediment to a wider application of COMGHA. New research initiated needs yet to prove potential for increased production at lower prices.
5.4 DEGD (diethylene glycol dibenzoate)
DEGD is the esterification product of two benzoate groups with diethylene glycol, see structural formula below. Its CAS No. is 120-55-8. It is marketed by Genovique in a mixture with two other dibenzoates under the product name Benzoflex 2088. The two other dibenzoates are dipropylene glycol dibenzoate (DGD; CAS 27138-31-4) and triethylene glycol dibenzoate (CAS 120-56-9). In this section, the mixed product is described. Note that DGD is described separately in the toxicological assessment. DGD is quite similar to DEGD in structure except for two extra methyl groups.
DEGD; diethylene glycol dibenzoate)
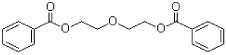
(diagram from www.chemblink.com)
Producer's description (extracts)
Benzoflex® 2088 is a high solvating plasticizer primarily known for its exceptional performance in polyvinyl acetate and water-based adhesive systems. It displays good wet tack, set times and open times. It also improves adhesion in acrylic latex caulks (Genovique, 2009b).
In Europe, Benzoflex® 2088 is Genovique’s most cost effective replacement for fast fusing phthalate plasticizers used in vinyl applications. It can substitute for phthalates such as BBP, DBP, DIHP and DIBP. It has had its greatest success replacing phthalates in plastisol application, the largest of which is resilient flooring. Over the past five years Benzoflex® 2088 has been established as largest volume non-phthalate, fast fusing plasticizer used in resilient flooring in Europe. Most plastisols were formulated with phthalates in mind, so utilizing an alternative chemistry, like benzoates, requires formulation adjustments (Genovique, 2009).
Application and market experience
Genovique has provided information on application areas for Benzoflex 2088 among the traditional DEHP, DBP and BBP applications shown in Table 5.11. The table also indicates the level of market experience in each application area according to Genovique (2009; interpreted from qualitative text by the report authors). Note the significant market experience in several of the traditional DBP and BBP specialty plasticiser applications and certain DEHP applications, notably in the non-polymer (adhesives, sealants, etc.) and PVC spread coating (plastisol) application fields.
In a study of plasticiser alternatives for non-PVC applications (COWI, 2000), DEGD was proposed by market actors as a substitute for phthalates in adhesives and sealants.
| Application | Market experience *1 |
|---|---|
| Substituting for DEHP | |
| Polymer applications: | |
| Calendering of film, sheet and coated products | 4 |
| Calendering of flooring, roofing, wall covering | 3 |
| Extrusion of hose and profile | 3 |
| Extrusion of wire and cable | 3 |
| Extrusion of miscellaneous products from compounds | 2 |
| Injection moulding of footwear and miscellaneous | 2 |
| Spread coating of flooring | 2 |
| Spread coating of coated fabric, wall covering, coil coating, etc. | 2 |
| Car undercoating | 3 |
| Non polymer applications: | |
| Adhesives/sealant, rubber | 1 |
| Lacquers and paint | 2 |
| Printing ink | 2 |
| Production of ceramics | |
| Substituting for DBP | |
| Plasticiser in PVC | 1 |
| Plasticiser in other polymers | |
| Adhesives | 1 |
| Printing inks | |
| Miscellaneous: | |
| Sealants | |
| PU foam sealants | |
| Nitrocellulose paints | 2 |
| Film coatings | |
| Glass fibre production | |
| Cosmetics | |
| Substituting for BBP | |
| Polymer applications: | |
| General PVC (e.g. for moulded plastic parts) | |
| Plastisol coating, for flooring | 1 |
| Extrusion or spreadcoating: Leather and cloth coating (e.g. for furniture, shoes, bags, suitcases) | 2 |
| Films, calendering (e.g. for packaging, calendered flooring, wall covering, etc.) | 4 |
| Non polymer applications: | |
| Sealants (polysulfide based, polyurethane foam sealants, acrylic based; e.g. for windows, construction etc.) | 1 |
| Coatings and inks (e.g. for car care products, construction, paper, board) | |
| Adhesives (polymer based, e.g. for construction, paper) | 1 |
*1: Market experience categories interpretation: 1) Main alternative on market. 2) Significant market experience. 3) Examples of full scale experience. 4) Pilot/lab scale experience.
Key characteristics
DGD is a commonly used benzoate. According to Krauskopf and Godwin (2005), benzoates are generally strong solvates due to the high aromaticity, as are lower molecular weight phthalates such as BBP. Commercial practice includes the use of up to 10–20% of the plasticiser system as “strong solvating” type plasticisers, such as aryl-alkyl phthalates (e.g. BBP), benzoates, etc.
Table 5.12 below describes some key characteristics of Benzoflex 2088 as alternative to DEHP, DBP and BBP.
| Parameter | Value | Remarks |
|---|---|---|
| Efficiency( as plasticiser in PVC compared to DEHP)*1 | ||
| Price (primo 2009) | ||
| Price relative to DEHP | "Slightly higher" than DEHP and DBP; equivalent to BBP (Genovique, 2009) | |
| Effective price relative to DEHP | ||
| Compatibility/solubility in PVC | Compatible | |
| Permanency (migration, evaporation, extraction) | App. double volatility as DEHP in plastisols. Three times higher water extraction than DEHP. Much lower organic solvents extraction than DEHP (Genovique, 2009). | |
| Processability (fusing speed and temperature, viscosity, etc.) | Lower gelling/fusing temperature in PVC than with BBP and DEHP. Slightly higher viscosity in plastisols (Genovique, 2009) | |
| Limitations in use, if any, noted by supplier in data for this study |
Notes: *1: Efficiency indicator, also called substitution factor, indicating the concentration of plasticiser in PVC needed, compared to DEHP, to achieve a specified flexibility according to a well defined method. DEHP has substitution factor 1 per definition, while plasticisers needing lower concentration (more effective) have lower values, and plasticisers needing higher concentrations have higher effectiveness value. NA = not available
Table 5.13 below shows some performance data for DEGD (as single substance) compared to DEHP, DBP and BBP. As shown, DEGD has similar characteristics to BBP on these parameters, except for a factor 10 higher extractability in water.
| Plasticiser in PVC, conc. 40% =67 phr in same PVC resin | Shore A hardness | Volatility,% lost, 1 day at 87 °C over activated carbon | Extracted in water,% | Extracted in kerosene (jet fuel, etc.),% |
|---|---|---|---|---|
| DEHP | 69 | 4.5 | 0.01 | 44 |
| DBP | 62 | 45.4 | 0.25 | 9.1 |
| BBP | 68 | 7.7 | 0.07 | 3.4 |
| DEGD (as single substance) | 69 | 5.5 | 0.75 | 3.4 |
Conclusions
The producer, Genovique, has indicated significant market experience in several of the traditional DBP and BBP specialty plasticiser applications and certain DEHP applications, notably in the non-polymer (adhesives, sealants, etc.) and PVC spread coating (plastisol) application fields. According to the producer, Benzoflex 2088 has become the main non-phthalate alternative to DBP/BBP in vinyl flooring production in Europe. The higher extractability in water may limit its use for some applications. Prices are indicated as "slightly higher" than DEHP and DBP; equivalent to BBP by Genovique.
5.5 DGD (dipropylene glycol dibenzoate)
DGD is the esterification product of two benzoate groups with dipropylene glycol, see structural formula below. Its CAS No. is 27138-31-4. It is marketed by Genovique under the product name Benzoflex 9-88. DGD is quite similar to DEGD except for two extra methyl groups.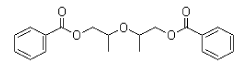
DGD; dipropylene glycol dibenzoate
Producer's description (extracts)
DGD is a high solvating plasticizer that has been used for many years in a wide variety of applications. Its diverse uses include resilient flooring, adhesives, artificial leather cloth and caulk (Genovique, 2009b).
DGD can be used as a replacement for BBP and DBP in vinyl applications. Its gel fusion temperature is identical to BBP and DBP in vinyl plastisol applications allowing it to be as close to a drop-in replacement for BBP and DBP as possible (Genovique, 2009).
Application and market experience
Genovique has provided information on application areas for DGD among the traditional DEHP, DBP and BBP applications shown in Table 5.14. The table also indicates the level of market experience in each application area according to Genovique (2009; interpreted from qualitative text by the report authors). Note the significant market experience in sealants, adhesives, coatings and inks as well as in PVC spread coating (plastisols), extrusion and injection moulding. DGD seems to be capable of substituting for both DEHP, DBP and BBP, indicating coverage of some general plasticiser features as well as some of the special performance characteristics of DBP and BBP.
In a study of plasticiser alternatives for non-PVC applications (COWI, 2000), DGD was proposed by market actors as a substitute for phthalates in adhesives and sealants.
| Application | Market experience *1 |
|---|---|
| Substituting for DEHP | |
| Polymer applications: | |
| Calendering of film, sheet and coated products | 4 |
| Calendering of flooring, roofing, wall covering | 3 |
| Extrusion of hose and profile | 3 |
| Extrusion of wire and cable | 3 |
| Extrusion of miscellaneous products from compounds | 2 |
| Injection moulding of footwear and miscellaneous | 2 |
| Spread coating of flooring | 2 |
| Spread coating of coated fabric, wall covering, coil coating, etc. | 2 |
| Car undercoating | 3 |
| Non polymer applications: | |
| Adhesives/sealant, rubber | 1 |
| Lacquers and paint | 2 |
| Printing ink | 2 |
| Production of ceramics | |
| Substituting for DBP | |
| Plasticiser in PVC | 1 |
| Plasticiser in other polymers | |
| Adhesives | |
| Printing inks | |
| Miscellaneous: | |
| Sealants | |
| PU foam sealants | |
| Nitrocellulose paints | 2 |
| Film coatings | |
| Glass fibre production | |
| Cosmetics | |
| Substituting for BBP | |
| Polymer applications: | |
| General PVC (e.g. for moulded plastic parts) | |
| Plastisol coating, for flooring | 1 |
| Extrusion or spreadcoating: Leather and cloth coating (e.g. for furniture, shoes, bags, suitcases) | |
| Films, calendering (e.g. for packaging, calendered flooring, wall covering, etc.) | 4 |
| Non polymer applications: | |
| Sealants (polysulfide based, polyurethane foam sealants, acrylic based; e.g. for windows, construction etc.) | 1 |
| Coatings and inks (e.g. for car care products, construction, paper, board) | 1 |
| Adhesives (polymer based, e.g. for construction, paper) |
*1: Market experience categories interpretation: 1) Main alternative on market. 2) Significant market experience. 3) Examples of full scale experience. 4) Pilot/lab scale experience.
Key characteristics
DGD is a commonly used benzoate. According to Krauskopf and Godwin (2005), it's preferred use is in PVC flooring products, owing to its strong solvating strength, and it reportedly controls plasticiser bleeding into asphalt adhesives. In vinyl sheet flooring, the benzoate enhances processing, while the low molecular weight contributes a hardened, stain resistant surface, due to volatilization, similar to the effect of BBP in flooring. Benzoates are generally strong solvents due to the high aromaticity, as are lower molecular weight phthalates such as BBP. Commercial practice includes the use of up to 10–20% of the plasticiser system as “strong solvating” type plasticisers, such as aryl-alkyl phthalates (e.g. BBP), benzoates, etc.
Table 5.15 describes some key characteristics of DGD as alternative to DEHP, DBP and BBP.
| Parameter | Value | Remarks |
|---|---|---|
| Efficiency( as plasticiser in PVC compared to DEHP)*1 | 0.98 | TURI (2006, citing industry) |
| Price | 0.73 USD/Lb | TURI (2006, citing industry) |
| Price relative to DEHP (2006: 0.70 USD/Lb) |
2009: "Slightly higher" 2006: 104% |
2009: "Slightly higher" according to Genovique (2009); also compared to DBP prices. Equivalent to BBP prices. 2006: Calculated from (TURI, 2006, citing industry); Same price relative to BBP. |
| Effective price relative to DEHP | 102% | |
| Compatibility/solubility in PVC | Compatible | |
| Permanency (migration, evaporation, extraction) | Lover volatility than its competitor BBP (Wilson, 1995). | |
| Processability (fusing speed and temperature, viscosity, etc.) | High solvating, fast fusing; competing with BBP (Wilson, 1995, and others). TURI (2006): Compounding easier than with DEHP; calendering: no issues identified. Some general plasticiser like applications will require blends with slower fusing plasticizers (Genovique, 2009). |
|
| Limitations in use, if any, noted by supplier in data for this study | None noted |
Notes: *1: Efficiency indicator, also called substitution factor, indicating the concentration of plasticiser in PVC needed, compared to DEHP, to achieve a specified flexibility according to a well defined method. DEHP has substitution factor 1 per definition, while plasticisers needing lower concentration (more effective) have lower values, and plasticisers needing higher concentrations have higher effectiveness value. NA = not available
Table 5.16 below shows some performance data for DGD compared to DEHP, DBP and BBP. As shown, DGD has very similar characteristics to BBP on these parameters, except for a higher extractability in water.
| Plasticiser in PVC, conc. 40% =67 phr in same PVC resin | Shore A hardness | Volatility,% lost, 1 day at 87 °C over activated carbon | Extracted in water,% | Extracted in kerosene (jet fuel, etc.),% |
|---|---|---|---|---|
| DEHP | 69 | 4.5 | 0.01 | 44 |
| DBP | 62 | 45.4 | 0.25 | 9.1 |
| BBP | 68 | 7.7 | 0.07 | 3.4 |
| DGD | 71 | 7.9 | 0.45 | 2.9 |
Wilson (1995) states that the consumption of benzoates had so far been minor in Europe although they were well known in the USA, where they were established plasticisers in the PVC flooring industry. Here they were used as fast fusing stain resistant plasticisers. Wilson states the most commercially important benzoate as DGD, which is broadly competitive with BBP, had the advantage of somewhat lower volatility.
Wilson (1995) also emphasises that DGD had replaced much of the previous C4 phthalate use (i.e. DBP, BBP) in PVA adhesives in the USA. In the mid 1990s this substitution had not yet happened in Europe, partly due to higher benzoate prices in Europe, partly due to less regulatory (health and environment) pressure at that time.
Karbæk (2003), who tested DGD and other plasticisers in PVC for medical purposes, found its poor resistance to extraction by water a major drawback for its use in medical applications. Otherwise DGD performed technically well compared to DEHP on all tested parameters, except for tensile strain at break and flexibility at low temperatures.
BBP is mentioned as a critical component in seals for insulating double glaz-ing (BBP Information Centre, 2009), but it has not been specifically investigated whether DGD or other benzoates can substitute for BBP for this particular application.
Conclusions
Genovique, the producer of DGD, has indicated significant market experience in sealants, adhesives, coatings and inks as well as in PVC spread coating (plastisols), extrusion and injection moulding. The qualities of DGD seem especially suitable for substitution of BBP, while it may also substitute for some traditional uses of DEHP and DBP. The fact that DGD has for many years been a well known and much used competitor to BBP in USA, especially in the flooring industry and in PVA adhesives, indicates a clear potential for substituting DGD for BBP, from a technical point of view. DGD and benzoates may already have played a part in the observed reductions (COWI, 2009c) of BBP usage in Europe.
The price of DGD seems also to be largely competitive with low molecular weight phthalates such as BBP. Currently the effective price is only slightly higher than the price of BBP. As with many other substitution processes, a price decrease may occur if the market increases in open competition between producers.
5.6 DEHT (di-ethyl-hexyl-terephthalate)
DEHT is a phthalate ester stoekiometrically equal to DEHP, i.e. phthalate ester bound to two ethylhexyl groups, but with a different spatial structure, because one of the carboxylic groups is placed differently on the benzyl ring; see diagram of the structure below ("tere" means tertiary, or third, because the carboxylic group is placed on the third carbon atom counted from the first carboxyl group). DEHT is marketed by Eastman Chemical Company under the product name Eastman 168. It is also marketed by LG Chem under the name LGflex GL300. There are several other producers world wide.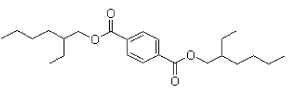
Terephthalate, (DEHT; di 2-ethylhexyl terephthalate); CAS no. 6422-86-2
Producer's description (extracts)
Eastman Chemicals has given the following short presentation of DEHT (Eastman, 2009b): DEHT is a good general purpose plasticizer for PVC, with performance equal or better than most orthophthalate plasticizers (Eds.: "ortho" is the form for most common phthalate plasticisers, such as in DEHP). It offers good performance properties, good low temperature flexibility, resistance to extraction by soapy water and good non-migration properties. In plastisols, DEHT results in low initial viscosity and good keeping viscosity.
Applications/Uses according to Eastman (2009b):
- Bottle caps and closures
- Coatings
- Coatings for cloth
- Electric connectors
- Flexible film
- Pavement striping compounds
- Sheet vinyl flooring
- Toys
- Traffic cones
- Vinyl compounding
- Vinyl gloves
- Vinyl products
- Vinyl water stops
- Walk-off mats
Application and market experience
Eastman Chemicals has provided information on application areas for DEHT among the traditional DEHP, DBP and BBP applications shown in Table 5.17. The table also indicates the level of market experience in each application area according to Eastman (2009; interpreted from qualitative text by the report authors). Note that Eastman has indicated significant market experience for all traditional DEHP uses, except car undercoating and production of ceramics. No traditional DBP and BBP applications have been indicated by Eastman, signalling that they consider DEHT as general plasticiser (such as DEHP, DINP and DIDP).
| Application | Market experience *1 |
|---|---|
| Substituting for DEHP | |
| Polymer applications: | |
| Calendering of film, sheet and coated products | 2 |
| Calendering of flooring, roofing, wall covering | 2 |
| Extrusion of hose and profile | 2 |
| Extrusion of wire and cable | 2 |
| Extrusion of miscellaneous products from compounds | 2 |
| Injection moulding of footwear and miscellaneous | 2 |
| Spread coating of flooring | 2 |
| Spread coating of coated fabric, wall covering, coil coating, etc. | 2 |
| Car undercoating | |
| Non polymer applications: | |
| Adhesives/sealant, rubber | 2 |
| Lacquers and paint | 2 |
| Printing ink | 2 |
| Production of ceramics | |
| PVC medical articles | 2 |
| Toy and childcare articles | 2 |
*1: Market experience categories interpretation: 1) Main alternative on market. 2) Significant market experience. 3) Examples of full scale experience. 4) Pilot/lab scale experience.
Key characteristics
Table 5.18 below shows selected comparisons from Eastman (2009c) between DEHT ("168"), DEHP ("DOP"), TXIB and other plasticisers of interest for selected parameters (more parameters are shown in Eastman 2009c). Note that DEHT has similar extraction values to DEHP in oil and hexane, and a factor two lower in soapy water. Volatility at elevated temperatures (expressed as activated carbon extraction) is 50%-67% of the volatility of DEHP. The low temperature flexibility of DEHT in PVC is equal to that of DEHP, whereas 100% modulus is app. 20% higher than for DEHP indicating slightly lower efficiency of DEHT (substitution factor 1.03) than DEHP. DEHT has about half the viscosity of DEHP in plastisols.
Table 5.18 Comparison between DEHT ("168"), DEHP ("DOP"), TXIB and other plasticisers (from Eastman, 2009c)
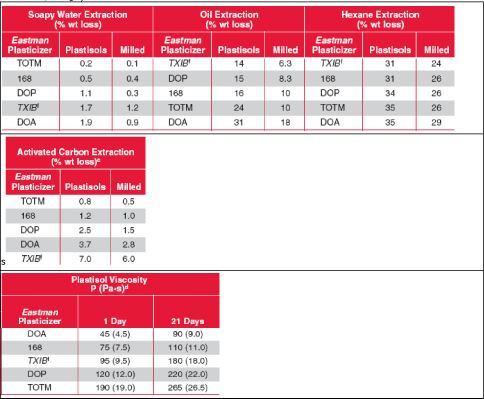
Notes: Plastisols contain 60 phr plasticiser; milled and calendered PVC contains 50 phr plasticiser; phr meaning parts per 100 parts hard PVC by weight. a) Stress at which PVC is elongated 100%. Lower value indicates higher efficiency of plasticiser (ASTM D638). b) Temperature at which the shown stiffness (torsion) is reached (ASTM D1043). c) Indication of volatility at elevated temperatures (ASTM D1203). d) Brookfield viscosity, determined with a number 4 spindle at 6rpm and 23 C.
Table 5.19 shows another performance data set for DEHT compared to DEHP. As shown, DEHT results in quite similar hardness as DEHP on these parameters. The volatility of DEHT is somewhat lower, and the extractability in water and kerosene is somewhat higher.
| Plasticiser in PVC, conc. 40% =67 phr in same PVC resin*1 | Shore A hardness*2 | Volatility,% lost, 1 day at 87 °C over activated carbon | Extracted in water,% | Extracted in kerosene (jet fuel, etc.),% |
|---|---|---|---|---|
| DEHP (PVC2) | 73 | 3.6 | 0.02 | 54.7 |
| DEHT (PVC2) | 76 | 1.9 | 0.09 | 70.8 |
Table 5.20 below describes some generalised key characteristics of DEHT as alternative to DEHP.
| Parameter | Value | Remarks |
|---|---|---|
| Efficiency( as plasticiser in PVC compared to DEHP)*1 | 1.03 | TURI (2006) |
| Price | 0.74 USD/Lb | TURI (2006) |
| Price relative to DEHP (2006: 0.70 USD/Lb) | 106% | TURI (2006) Also according to Krauskopf and Godwin (2005), DEHT is commercially available at similar price as DEHP. |
| Effective price relative to DEHP | 109% | TURI (2006) |
| Compatibility/solubility in PVC | Compatible | |
| Permanency (migration, evaporation, extraction) | Similar to DEHP, slightly higher permanence on some parameters, slightly lower on others, see above (Eastman, 2009c). | |
| Processability (fusing speed and temperature, viscosity, etc.) | See some values in Table 5.18 above | Overall very similar to DEHP and DINP (Eastman, 2009; TURI, 2006). Half the viscosity in plastisols as DEHP (advantage of DEHT); slightly higher gelling temperatures than DEHP (Eastman, 2009c). |
| Limitations in use, if any, noted by supplier in data for this study | Poor weatherability properties for external applications such as roofing and coil coating (Eastman, 2009) |
Notes: *1: Efficiency indicator, also called substitution factor, indicating the concentration of plasticiser in PVC needed, compared to DEHP, to achieve a specified flexibility according to a well defined method. DEHP has substitution factor 1 per definition, while plasticisers needing lower concentration (more effective) have lower values, and plasticisers needing higher concentrations have higher effectiveness value. NA = not available.
In practice, terephthalates are more commonly used in the USA than elsewhere (TURI, 2006).
Conclusions
All available information indicates that, technically, DEHT may be a good substitute for most or all traditional DEHP uses. In practice, terephthalates are more commonly used in the USA than elsewhere. DEHT was found in 7% and 10%, respectively, of products analysed in two European surveys of large samples of toys and childcare articles. It was also reported as used for toys by Danish toy manufacturers (with contract production in China).
According to 2006 information, DEHT is slightly more expensive than DEHP. As with many other substitution processes, a price decrease may occur if the market increases in open competition between producers.
5.7 DINA (diisononyl adipate)
DINA is formed by an adipate (hexanoic acid) ester bound with two C-9 alkanes, see diagram. It is marketed by ExxonMobil under the product name Jayflex-DINA, by BASF under the product name Plastomoll DNA, and formerly by Lanxess under the product name Adimoll® DN.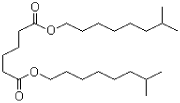
DINA = di isononyl adipate; CAS 33703-08-1
Producers' descriptions (extracts)
BASF has given the following short presentation of DINA (BASF, 2009): DINA is a nearly colourless, clear and practically anhydrous liquid with a hardly noticeable odour. It is soluble in the usual organic solvents and is miscible and compatible with all of the monomeric plasticizers commonly used in PVC. In water DINA is soluble only in very small amounts. Owing to its chemical structure, DINA permits – preferably, in combination with phthalates and polymeric plasticizers – the production of plasticized PVC products with exceptionally good low temperature properties. PVC plasticized with DINA has far less volatility than, for example, PVC plasticized with DOA.
ExxonMobil has given the following short presentation of DINA (ExxonMobil, 2009b):
Improved permanent adipate widely used in a variety of end-uses.
- Low temperature flexibility, low freezing point
- Lower volatility/higher permanence than DOA
- Enhanced viscosity stability and air release properties in plastisols
- Partial replacement for phthalates to improve low temperature flexibility
Applications:
- Films
- Shrink wrap
- Electrical wire jacket
Application and market experience
Neither BASF nor ExxonMobil has wished to provide detailed feedback for this project on DINA's suitability for substituting for DEHP, DBP and BBP.
In a study of plasticiser alternatives for non-PVC applications (COWI, 2000), DINA was proposed by market actors as a substitute for phthalates in adhesives, printing inks, paint and lacquer, and rubber.
Key characteristics
The family of adipic acid esters used in PVC applications improves low temperature performance relative to phthalates and give significantly lower plastisol viscosities in plastisol applications, due to the lower inherent viscosities of the plasticisers themselves (ECPI, 2009).
Table 5.21 shows some performance data for DINA compared to DEHP, DBP and BBP. As shown, DINA has similar hardness and volatility as DEHP, but higher extractability in water and kerosene.
| Plasticiser in PVC, conc. 40% =67 phr in same PVC resin | Shore A hardness | Volatility,% lost, 1 day at 87 °C over activated carbon | Extracted in water,% | Extracted in kerosene (jet fuel, etc.),% |
|---|---|---|---|---|
| DEHP | 69 | 4.5 | 0.01 | 44 |
| DBP | 62 | 45.4 | 0.25 | 9.1 |
| BBP | 68 | 7.7 | 0.07 | 3.4 |
| DINA | 72 | 4.1 | 0.14 | 80.4 |
Table 5.22 describes some generic characteristics of DINA as alternative to DEHP, DBP and BBP.
| Parameter | Value | Remarks |
|---|---|---|
| Efficiency( as plasticiser in PVC compared to DEHP)*1 | 0.98 | |
| Price (primo 2009) | NA | |
| Price relative to DEHP | 150-200% ≈130% |
ExxonMobil (2009) Arbeitsgemeinschaft PVC und Umwelt e.V. (2006; statement for adipates in general) |
| Effective price relative to DEHP | NA | |
| Compatibility/solubility in PVC | Compatible | |
| Permanency (migration, evaporation, extraction) | Far less volatility in PVC than DOA (BASF, 2009); DOA is slightly more volatile than DEHP (Eastman, 2009c) | |
| Processability (fusing speed and temperature, viscosity, etc.) | Enhanced viscosity stability and air release properties in plastisols | |
| Limitations in use, if any, noted by supplier in data for this study | NA |
Notes: *1: Efficiency indicator, also called substitution factor, indicating the concentration of plasticiser in PVC needed, compared to DEHP, to achieve a specified flexibility according to a well defined method. DEHP has substitution factor 1 per definition, while plasticisers needing lower concentration (more effective) have lower values, and plasticisers needing higher concentrations have higher effectiveness value. NA = not available
Conclusions
It has not been possible to get detailed application data compared to DEHP, DBP and BBP for DINA from producers. Among the adipates, DEHA (or DOA) is the most used. It was however not included for further investigation in this study because it was reported by SCENIHR (2008) to have reproductive toxicity. Instead DINA was chosen for further study. In PVC DINA has similar hardness and volatility as DEHP, but higher extractability in water and kerosene. DINA has mostly been used for low temperature PVC applications and in PVC film/wrapping. According to the producers, PVC plasticized with DINA has far less volatility than, for example, PVC plasticized with DEHA. DINA seems therefore potentially more suitable than DEHA, from a technical point of view, as an alternative to DEHP than DEHA. DINA seems currently only to have a small European market and data for DINA are scarcer than for DEHA. In fact two producers have ceased marketing of DINA on the European market. However, DINA was found in 6% and 4%, respectively, of products analysed in two European studies of large samples of toys and childcare articles. It was also reported as used for toys by Danish toy manufacturers (with contract production in China). The data available for this study does not allow clear-cut conclusions as regards DINA's suitability as alternative to DEHP, but DINA could perhaps be worth investigating in future technical explorations for alternatives.
The price for DINA is currently somewhat higher than for DEHP, but as mentioned the market is small and DINA is used as a specialty plasticiser.
5.8 DINCH (di-isononyl-cyclohexane-1,2dicarboxylate)
DINCH is the hydrogenated parallel to DINP, with the difference that the ring structure is cyclohexane (a cyclic alkyl hydrocarbon) instead of a benzene ring (an aromate). Its CAS no. is 166412-78-8. It is marketed by BASF as Hexamoll DINCH. There may be other producers outside the EU.
Producer's description (extracts)
BASF describes DINCH as follows (BASF, 2009): The combination of an good toxicological profile and a very low migration rate makes DINCH the plasticizer of choice for medical devices made with soft PVC products such as tubes for internal feeding and haemodialysis bags, respiratory tubes, catheters, gloves and breathing masks.
DINCH is the ideal additive for toys. Thanks to its low migration rate, lack of odour and technical suitability, DINCH is the plasticizer of choice for toys and children’s articles such as dolls, inflatables and balls, figurines, modelling clay, swimming aids, baby and childcare articles, wire and cable for toys.
Thanks to its toxicological profile, its low migration rate, and especially the low solubility in water and ethanol, this additive is developed for food contact applications such as cling film, hoses, sealants and cap closures, crown corks, artificial wine corks, gaskets and gloves.
DINCH is also suitable for other applications where safety is needed, also outside PVC: Thermoplastics and polar rubbers, coatings and printing inks, dispersions, adhesives, cosmetics (e.g. nail polish), masterbatches, artificial leather, textile coatings (e.g. rain coats), erasers, film and sheets (e.g. lifestyle bags).
Application and market experience
BASF has not wished to provide detailed feedback for this project on DINCH's suitability for substituting for DEHP, DBP and BBP.
In 2007, BASF raised its DINCH production capacity from 25,000 to 100,000 tonnes/yr (MPW, 2008), indicating an increasing demand.
Key characteristics
Table 5.23 below describes some key characteristics of DINCH as alternative to DEHP, DBP and BBP.
| Parameter | Value | Remarks |
|---|---|---|
| Efficiency (as plasticiser in PVC compared to DEHP)*1 | NA | |
| Price | $0.91 /lb | TURI (2006) |
| Price relative to DEHP (2006: 0.70 USD/Lb) | 130% | TURI (2006) |
| Effective price relative to DEHP | NA | |
| Compatibility/solubility in PVC | Compatible | |
| Permanency (migration, evaporation, extraction) | Migration levels eight times lower than that of DEHP (MPW, 2008). | |
| Processability (fusing speed and temperature, viscosity, etc.) | NA | |
| Limitations in use, if any, noted by supplier in data for this study | NA |
Notes: *1: Efficiency indicator, also called substitution factor, indicating the concentration of plasticiser in PVC needed, compared to DEHP, to achieve a specified flexibility according to a well defined method. DEHP has substitution factor 1 per definition, while plasticisers needing lower concentration (more effective) have lower values, and plasticisers needing higher concentrations have higher effectiveness value. NA = not available
Conclusions
The producer’s sales appraisal indicates a relatively wide usage of DINCH for general plasticiser purposes, where safety is prioritised. The production capacity has been raised substantially, indicating a growing demand and market experience. More detailed information of applications substituting for DEHP; DBP and BBP has not been available from the producer. DINCH was the most frequently found plasticiser in two European studies of large samples of toys and childcare articles. It was found in 25% and 48%, respectively, of the analysed products. It was also reported as used for toys by Danish toy manufacturers (with contract production in China). The data available does not allow a closer assessment of DINCH's technical suitability as alternative to DEHP, DBP and BBP.
Sales prices in 2006 were somewhat higher than DEHP. As with many other substitution processes, a price decrease may occur if the market increases in open competition between producers.
5.9 GTA (glycerol triacetate)
GTA is an ester of glycerol and three acetate groups, 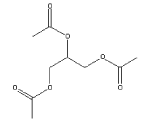 see diagram below. Its CAS no. is 102-76-1. It is marketed by Lanxess and Eastman under the name Triacetin.
see diagram below. Its CAS no. is 102-76-1. It is marketed by Lanxess and Eastman under the name Triacetin.
Glyceryl triacetate (GTA, Triacetin)
Producer's description (extracts)
Lanxess (2009) presents GTA as follows: Triacetin is used for the solidification of acetyl cellulose fibres in the manufacture of cigarette filters. The water content must be kept constant to achieve constant solidification. Triacetin is also used as a support for flavourings and essences in the food industry and as a plasticiser for chewing gum. In technical applications, Triacetin is used for example as a core sand binder in the metal foundry sector. Another application is inks and printing inks. Triacetin is used as a highly effective plasticiser for cellulose-based plastics.
The major features of Triacetin are:
- good suitability for the solidification of acetyl cellulose fibres for the manufacture of cigarette filters
- very good dissolving power for a number of organic substances
- good plasticising effect for various plastics such as cellulose acetates or celluloseacetobutyrates
- good plasticising effect for cellulose-based paints
- good compatibility with natural and synthetic rubber
- good light resistance
Eastman presents GTA as follows (Eastman, 2009d): GTA is used as a plasticizer for cellulosic resins and is compatible in all proportions with cellulose acetate, nitrocellulose, and ethyl cellulose. GTA is useful for imparting plasticity and flow to laminating resins, particularly at low temperatures, and is also used as a plasticizer for vinylidene polymers and copolymers. It serves as an ingredient in inks for printing on plastics, and as a plasticizer in nail polish. GTA is approved by the FDA for food packaging and many other food-contact applications.
Application and market experience
Lanxess has provided information on application areas for GTA among the traditional DEHP, DBP and BBP applications shown in Table 5.24. The table also indicates the level of market experience in each application area according to Lanxess (2009; interpreted from qualitative text by the report authors). Note that Lanxess has indicated significant market experience for adhesives, coatings and inks and examples of full scale experience in a number of other non-polymer traditional DBP and BBP uses. Lanxess has not indicated use of GTA in any traditional DEHP uses.
| Application | Market experience *1 |
|---|---|
| Substituting for DEHP | |
| Plasticiser in PVC | |
| Plasticiser in other polymers | |
| Adhesives | 2 |
| Printing inks | 3 |
| Miscellaneous: | |
| Sealants | |
| PU foam sealants | |
| Nitrocellulose paints | 3 |
| Film coatings | |
| Glass fibre production | |
| Cosmetics | |
| Substituting for BBP | |
| Polymer applications: | |
| General PVC (e.g. for moulded plastic parts) | |
| Plastisol coating, for flooring | |
| Extrusion or spreadcoating: Leather and cloth coating (e.g. for furniture, shoes, bags, suitcases) | |
| Films, calendering (e.g. for packaging, calendered flooring, wall covering, etc.) | |
| Non polymer applications: | |
| Sealants (polysulfide based, polyurethane foam sealants, acrylic based; e.g. for windows, construction etc.) | |
| Coatings and inks (e.g. for car care products, construction, paper, board) | 2 |
| Adhesives (polymer based, e.g. for construction, paper) |
*1: Market experience categories interpretation: 1) Main alternative on market. 2) Significant market experience. 3) Examples of full scale experience. 4) Pilot/lab scale experience.
Key characteristics
Table 5.25 describes some key characteristics of GTA as alternative to DEHP, DBP and BBP.
| Parameter | Value | Remarks |
|---|---|---|
| Efficiency (as plasticiser in PVC compared to DEHP)*1 | NA | |
| Price (primo 2009) | €1,50/KG | Lanxess (2009) |
| Price relative to DEHP (≈0.8-1€/kg in 2008/2009; 1€ used for calculations) | 150% | |
| Effective price relative to DEHP | NA | |
| Limitations in use, if any, noted by supplier in data for this study | NA |
Notes: *1: Efficiency indicator, also called substitution factor, indicating the concentration of plasticiser in PVC needed, compared to DEHP, to achieve a specified flexibility according to a well defined method. DEHP has substitution factor 1 per definition, while plasticisers needing lower concentration (more effective) have lower values, and plasticisers needing higher concentrations have higher effectiveness value. NA = not available
Conclusions
According to a producer, GTA can substitute for DBP and BBP in adhesives, inks and coatings. The price of GTA is somewhat higher that DEHP (and DBP and BBP). The data available does not allow a closer assessment of GTA's technical suitability as alternative to DEHP, DBP and BBP.
5.10 TXIB (trimethyl pentanyl diisobutyrate)
TXIB is an ester of the branched alkane trimethyl pentanyl with two butyrate groups. Its CAS no. is 6846-50-0. Until 2006, it was marketed by Eastman as TXIB plasticiser, but since then it has been renamed TXIB formulation additive.
TXIB
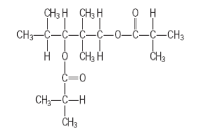
(Eastman, 2009e)
Producer's description (extracts)
Eastman has given the following presentations of TXIB (Eastmann, 2006, 2009f): TXIB formulation additive is a superior primary plasticiser for PVC plastisols. It has good compatibility with PVC, and it is compatible with all common primary and secondary plasticisers. TXIB provides low viscosity characteristics in plastisols with good viscosity stability over time. TXIB is the lowest viscosity (9 cps) additive available to the flexible PVC industry. TXIB is completely compatible with PVC in all proportions and is usually blended with general-purpose plasticizers such as DOP or DOTP. The unique low viscosity makes this material particularly suitable for PVC plastisols and often allows adding additional fillers to the plastisol, resulting in a cost savings. TXIB has efficiency generally equal to DOP, which makes substitution in a vinyl formulation very easy. It imparts a dry surface to the vinyl, excellent resistance to staining, and physical properties equivalent to DOP. TXIB is also used in inks, coatings, urethane elastomers, and nail polish lacquers.
Application/Uses:
- Automotive OEM
- Coatings for automotive plastics
- Lithographic and letterpress oil-based inks
- Nail care
- Phthalate-free diluent for MEKP formulations
- Plastisols
- Sheet vinyl flooring
- Toys/Sporting goods
- Traffic cones
- Vinyl compounding
- Vinyl gloves
- Wall coverings
Application and market experience
Eastman has not wished to provide detailed feedback for this project on TXIB's suitability for substituting for DEHP, DBP and BBP. Eastman has informed that TXIB cannot be used as a direct replacement for DEHP, DBP or BBP. TXIB is used to lower the viscosity of plastisols; one function DBP has been used for. TXIB does however not increase gellation speed for faster production, as DBP and BBP is used for. TXIB cannot work as a primary plasticiser (Eastman, 2009).
In a study of plasticiser alternatives for non-PVC applications (COWI, 2000), TXIB was proposed by market actors as a substitute for phthalates in adhesives and sealants.
Key characteristics
Table 5.26 below shows selected comparisons from Eastman (2009c) between TXIB, DEHP ("DOP"), DEHT ("168") and other plasticisers of interest for selected parameters (more parameters are shown in reference). Note that TXIB in PVC has lower extractability in oil and hexane than DEHP, but higher in soapy water. The modulus (resistance to elongation) and low temperature flexibility is similar to DEHP, while the volatility of TXIB is a factor three higher than DEHP. Plastisol viscosities are lower than for DEHP.
Table 5.26 Comparison between DEHT ("168"), DEHP ("DOP"), TXIB and other plasticisers (from Eastman, 2009c)
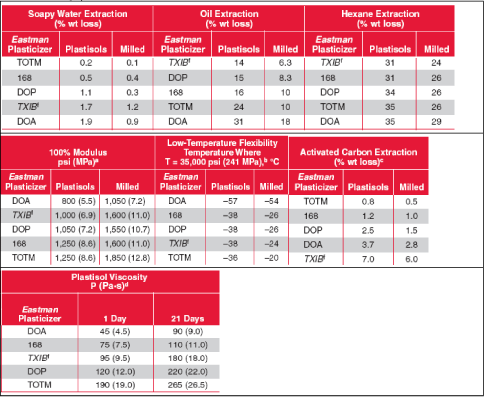
Notes: Plastisols contain 60 phr plasticiser; milled and calendered PVC contains 50 phr plasticiser; phr meaning parts per 100 parts hard PVC by weight. a) Stress at which PVC is elongated 100%. Lower value indicates higher efficiency of plasticiser (ASTM D638). b) Temperature at which the shown stiffness (torsion) is reached (ASTM D1043). c) Indication of volatility at elevated temperatures (ASTM D1203). d) Brookfield viscosity, determined with a number 4 spindle at 6rpm and 23 C.
Table 5.27 below shows some other performance dataset for TXIB compared to DEHP, DBP and BBP. As shown, here TXIB has higher hardness (lower efficiency) than the phthalates, high volatility (yet lower than DBP) and much higher extractability in water. Extractability in kerosene is higher than BBP, but lower than DEHP and DBP.
| Plasticiser in PVC, conc. 40% =67 phr in same PVC resin | Shore A hardness | Volatility,% lost, 1 day at 87 °C over activated carbon | Extracted in water,% | Extracted in kerosene (jet fuel, etc.),% |
|---|---|---|---|---|
| DEHP | 69 | 4.5 | 0.01 | 44 |
| DBP | 62 | 45.4 | 0.25 | 9.1 |
| BBP | 68 | 7.7 | 0.07 | 3.4 |
| TXIB | 76 | 23.7 | 2.83 | 5.2 |
Table 5.28 below describes some key characteristics of TXIB as alternative to DEHP, DBP and BBP.
| Parameter | Value | Remarks |
|---|---|---|
| Efficiency (as plasticiser in PVC compared to DEHP)*1 | Close to 1 (Eastman, 2009f) | |
| Price (primo 2009) | NA | |
| Price relative to DEHP | NA | |
| Effective price relative to DEHP | NA | |
| Compatibility/solubility in PVC | Compatible | |
| Permanency (migration, evaporation, extraction) | High volatility, see below (Wilson, 1995) | |
| Processability (fusing speed and temperature, etc.) | Gives low viscosity to plastisols (Eastman, 2009f) | |
| Limitations in use, if any, noted by supplier in data for this study | NA |
Notes: *1: Efficiency indicator, also called substitution factor, indicating the concentration of plasticiser in PVC needed, compared to DEHP, to achieve a specified flexibility according to a well defined method. DEHP has substitution factor 1 per definition, lower = more effective, higher = less effective. NA = not available
According to Wilson (1995), TXIB shows unique performance parameters in PVC plastisols. It is a useful component of plastisols formulated to give hard end products since it confers little viscosity at low levels of addition. It has been widely used in cushion vinyl flooring for this purpose, usually in conjunction with BBP; this use has however declined greatly in the 1990s as its high volatility causes unacceptable emissions from end products.
Conclusions
TXIB seems to have some technically relevant characteristics as plasticiser. TXIB was found in many products analysed, 25% and 11% respectively, in two European studies of large samples of toys and childcare articles (its presence need not have been as a primary plasticiser). Given the fact that the producer does not consider TXIB an alternative to DEHP, DBP or BBP, and the information that the usage of TXIB in vinyl flooring has declined in the 1990s due to high emissions from end products, it seems that TXIB should maybe not be seen as a suitable alternative to any of these substances.
5.11 Summary and discussion of technical and economical assessment of alternative plasticisers
The technical description in this report of ten selected alternatives to DEHP, DBP and BBP is based on the producers' assessments of relevant application fields and experience on the market, as well as the evidence of already established practises, especially in the toys, foodstuffs and medical product fields, but also for other end-uses. Based on the available information, a number of suitable alternative plasticisers have been identified for most applications. Some of the alternative plasticisers have a broad application scope, others are more specialised. A summary of the assessed alternatives is given in Table 5.29. A summary of some technical performance parameters for the assessed alternatives compared to DEHP, DBP, BBP and other selected phthalates is given below in annex 3. For some of the substances the manufacturers have provided information on market experience. The data are summarised in Table 5.30. Some of the substances, for which specific information on market experience has not been supplied (e.g. DINCH), certainly have some experience as indicated by the production volumes of the substances.
Industry may have, or may get in the future, more knowledge on the possibilities and limitations of the described plasticisers, which can feed into the decision-making process as regards regulation of DEHP, DBP and BBP. So far, the dominance for many years of DEHP and other ortho-phthalate plasticisers may have naturally limited the motivation to get more full scale experience with other plasticiser types. Under other circumstances, driven by other priorities, the experience with so far less favoured plasticisers would inevitably increase. The expenses associated with such additional research and implementation has not been assessed in this study. The research would however not have to start from scratch, as many relevant substances have been investigated for plasticiser characteristics in early research.
DINP and DIDP have become dominating alternatives to DEHP due to their closeness in performance to DEHP, their availability and their only moderately higher costs. As mentioned, these ortho-phthalates were not included for deeper assessment in this study, because they are already well described, technically and environmentally.
In some cases, blends of different alternative plasticisers may be needed to attain the desired technical characteristics. This is also a well known practice with many DEHP uses. More blending may be needed with some of the non-phthalate alternatives to achieve general plasticiser characteristics. By way of example, a Danish toy producing company attempted a switch to ATBC as primary plasticiser in all PVC toys etc. for small children. However in the existing production setup, it suffered from a variety of technical drawbacks when compared with DINP: ATBC would not take decoration, it had high migration into adjacent materials leading to swelling and splitting, and there was a consequent need for tooling changes. Development led to the use of a mixture of ATBC, DINCH and DEHT, which could be blended in a variety of combinations to achieve softened PVC that performed to the required standards with the existing production setup, and could be used as a one-to-one alternative to DINP. Also, some of the marketed plasticiser products consist of several substances, pre-mixed to provide desired performance characteristics. The products Soft-n-safe and Benzoflex 2088 described in this report are examples of such mixed plasticiser products.
| Substance | Overall technical assessment |
|---|---|
| ASE | ASE is a general plasticiser alternative to DEHP. The producer has indicated significant market experience for most traditional DEHP, DBP and BBP uses. |
| ATBC | The performance of ATBC on some parameters seems similar to DEHP, indicating technical suitability for substitution of DEHP for some applications. The higher extractability in aqueous solutions and the higher volatility may reduce the performance of ATBC as a plasticiser in PVC. The data available does not allow a closer assessment of ATBC's technical suitability as alternative to DEHP, DBP and BBP |
| COMGHA | The producer has indicated significant market experience in several of the traditional DBP and BBP specialty plasticiser applications and certain DEHP applications, notably in the non-polymer (adhesives, sealants, etc.) and PVC spread coating (plastisol) application fields. According to the producer, Benzoflex 2088 (with DEGD) has become the main non-phthalate alternative to DBP or BBP in vinyl flooring production in Europe. The higher extractability in water may limit its use for some applications. |
| DEGD | According to the producer, COMGHA still has relative moderate market experience, albeit with many examples of full scale usage and pilot/lab scale tests, and significant market experience in some plastisol application and cosmetics. The producer found good performance on key technical parameters indicating a potential for substituting for DEHP and perhaps for DBP and BBP in some traditional uses og these substances. |
| DGD | DEHT is a general plasticiser alternative to DEHP. Today, terephthalates like DEHT are more commonly used in the USA than elsewhere. |
| DEHT | DINA has mostly been used for low temperature PVC applications and in PVC film/wrapping . The data available for this study does not allow clear-cut conclusions as regards DINA's suitability as alternative to DEHP |
| DINA | The producer’s sales appraisal indicates a relatively wide usage of DINCH for general plasticiser purposes. DINCH was the most frequently found plasticiser in two European surveys of plasticisers in toys and childcare articles. The data available does not allow a closer assessment of DINCH's technical suitability as alternative to DEHP, DBP and BBP. |
| DINCH | The fact that DGD for many years has been a well known and much used competitor to BBP, especially in PVC flooring and in PVA adhesives, indicates a clear potential for substituting DGD for BBP, from a technical point of view. DGD may probably also substitute for some traditional uses of DEHP and DBP. |
| GTA | According to a producer, GTA can substitute for DBP and BBP in adhesives, inks and coatings. The data available does not allow a closer assessment of GTA's technical suitability as alternative to DEHP, DBP and BBP. |
| TXIB | TXIB was found in more than 10% of the samples in surveys of plasticisers in toys and childcare articles. However, the producer does not consider TXIB an alternative to DEHP, DBP or BBP, and the usage of TXIB in vinyl flooring has declined in the 1990’s due to high emissions from end products. Consequently, TXIB seems not to be a suitable alternative to DEHP, DBP or BBP. |
| Application | ASE | GTA | DGD | Mix of DGD, DEGD, TGD | ATBC | COMGHA |
|---|---|---|---|---|---|---|
| Substitute for DEHP | ||||||
| Polymer applications: | ||||||
| Calendering of film, sheet and coated products | 2 | 2 | 4 | 4 | 3 | 3 |
| Calendering of flooring, roofing, wall covering | 4 | 2 | 3 | 3 | 3 | |
| Extrusion of hose and profile | 2 | 2 | 3 | 3 | 3 | 3 |
| Extrusion of wire and cable | 2 | 2 | 3 | 3 | 3 | |
| Extrusion of miscellaneous products | 2 | 2 | 2 | 2 | 2 | 3 |
| Injection moulding of footwear and miscellaneous | ? | 2 | 2 | 2 | 3 | |
| Spread coating of flooring | 2 | 2 | 2 | 2 | 2 | |
| Spread coating | 2 | 2 | 2 | 2 | 3 | |
| Car undercoating | 2 | 3 | 3 | |||
| PVC medical articles | 2 | 2 | ||||
| Toy and childcare articles | 2 | 1 | ||||
| Non polymer applications: | 0 | |||||
| Adhesives/sealant, rubber | 2 | 2 | 1 | 1 | 2 | 4 |
| Lacquers and paint | 2 | 2 | 2 | 2 | 4 | |
| Printing ink | 2 | 2 | 2 | 2 | 2 | 3 |
| Production of ceramics | ||||||
| Substitute for DBP | ||||||
| Plasticiser in PVC | 2 | 1 | 1 | 2 | 2 | |
| Plasticiser in other polymers | 2 | 2 | ||||
| Adhesives | 2 | 2 | 1 | 3 | 4 | |
| Printing inks | 2 | 3 | 2 | 3 | ||
| Miscellaneous: | ||||||
| Sealants | 2 | 3 | 4 | |||
| PU foam sealants | 2 | 4 | ||||
| Nitrocellulose paints | 2 | 3 | 2 | 2 | 2 | |
| Film coatings | 3 | 3 | ||||
| Glass fibre production | 4 | |||||
| Cosmetics | 2 | |||||
| Substitute for BBP | ||||||
| Polymer applications: | ||||||
| General PVC (e.g. for moulded plastic parts) | 2 | 4 | ||||
| Plastisol coating, for flooring | 2 | 1 | 1 | 3 | ||
| Extrusion or spread coating | 2 | 2 | 2 | |||
| Films, calendering | 2 | 4 | 4 | 3 | ||
| Non polymer applications: | ||||||
| Sealants | 2 | 1 | 1 | |||
| Coatings and inks) | 2 | 1 | 3 | |||
| Adhesives | 2 | 1 | ||||
| Nail polish | 1 |
*1: Market experience categories interpretation: 1) Main alternative on market. 2) Significant market experience. 3) Examples of full scale experience. 4) Pilot/lab scale experience
Prices of alternative plasticisers
Some of the alternative plasticisers investigated have similar or only slightly higher prices than the relevant competing substances among DEHP, DBP and BBP; see Table 5.31 below. Others have higher or substantially higher prices. As shown in the table, the non-phthalate alternatives DGD, DEGD and DEHT were in the same price range as the DEHP, DBP and BBP, (as well as the ortho-phthalate alternatives DINP and DIDP), whereas ASE, DINA , DINCH and GTA were somewhat more expensive and ATBC and COMGHA were considerably more expensive (counted as direct relative price). We have not been able to find a price for TXIB for this study. Note that here BBP and DEHP had the same price per weight unit in this case. In older literature, BBP is reported to be a specialty plasticiser with higher price than DEHP.
Prices of chemicals (and other industrial products) tend to decrease as production capacity and competition is increased. Different chemicals are however based on different raw materials and more or less complex and resource demanding chemical synthesis technologies. This of course sets limits to the minimum prices attainable even in a mature market, and some of the alternative plasticisers described may likely remain at higher price levels. It should be noted that the prices of DEHP have dropped significantly over the last decade or more.
According to a Danish manufacturer, when the Danish ban of DEHP and 5 other phthalates in toys etc. came into force, the price of products for one Danish toy company rose by approximately 50% because the international manufacturers had to produce special deliverances to the Danish marked without phthalates. Later, when the ban comprised the entire EU the prices dropped again. The company estimates that the ban has resulted in a remaining increase in prises of approximately 10-20% because the alternative substances generally are more expensive even after the preliminary reduced costs related to changing production methods.
| Substance | Price | Relative price to DEHP,% | Substitution factor | Effective relative price,% | Remarks´´´´´´ |
|---|---|---|---|---|---|
| Phthalates and other reference plasticisers: | |||||
| DEHP (2006) | 0.70 USD/Lb | - | - | - | TURI (2006) |
| DEHP (2006-2009) | ≈0.8-1 €/kg | - | - | - | ExxonMobil (2009), Arbeitsgemeinschaft PVC (2006) |
| BBP (2006) | 0.70 USD/Lb | 100% | 0.94 | 94% | TURI (2006) |
| DINP (2006) | 0.73 USD/Lb | 104% | 1.06 | 111% | TURI (2006) |
| DIDP (2006) | 0.77 USD/Lb | 110% | 1.10 | 121% | TURI (2006) |
| Assessed alternatives: | |||||
| ASE (2009) | 1,75 €/kg | 175% *1 | NA | NA | Lanxess (2009) |
| ATBC | NA | 300% | NA | NA | ExxonMobil (2009); Karbæk (2003) |
| COMGHA | 3.5€/kg | ≈350% *1 | ≈1 | ≈350% | Danisco (2009) |
| Benzoflex 2088 (with DEGD) | "Slightly higher" | Genovique (2009) | |||
| DGD (2006) | 0.73 USD/Lb | 104% | 0.98 | 102% | TURI (2006) |
| DEHT (2006) | 0.74 USD/Lb | 106% | 1.03 | 109% | TURI (2006) |
| DINA | 150-200% | ExxonMobil (2009) | |||
| DINCH (2006) | 0.91 USD/Lb | 130% | NA | NA | TURI (2006) |
| GTA | €1,50/KG | 150% *1 | NA | NA | Lanxess (2009) |
| TXIB | NA | NA | NA | NA |
Notes: *1: DEHP price in 2006-2009 ≈0.8-1 €/kg; 1 € is used for calculations. NA = Not available for this study.
6 Assessment of alternative flexible polymers
A number of studies have been undertaken on replacing PVC with other materials for different applications. The conclusion of many studies has been that it - on the basis of the available data - was not possible to make a full assessment of the materials.
To focus the efforts in this study, it was decided to include an assessment of polyolefin (polyethylene/polypropylene) elastomers as alternatives to flexible PVC - on an overall screening level - of its lifecycle impacts, supplemented by brief description of other flexible polymers based on avail-able aggregated reviews.
6.1 Assessment of polyolefin elastomers
A detailed life cycle assessment (LCA) was performed by Stripple et al. (2007) assessing the life cycle impacts of three flexible polymers in their use as urinary catheters, a disposable medical care product consisting of a thin flexible tube and a cone-shaped flexible connector in one end. The materials assessed were (Stripple et al., 2007, Melitek, 2006, 2009):
- DEHP-plasticised PVC;
- Thermoplastic polyurethane (TPU[1]);
- An elastomer, marketed by Melitek (Denmark) under the product name Meliflex (here designated PO), based on polypropylene, styrene block co-polymer polyethylene and (non-phthalate) additives in ppt concentrations.
The functional unit was one year's supply of catheters for one person. The LCA seems comprehensive and was performed by the independent institute IVL in Sweden. It was conducted using 4 different LCA methods, the Eco-indicator 99 system, the CML 2 system, and the EPS 2000 system, as well as a classification and characterisation in line with the EPD system (environmental product declaration system).
The result varied somewhat depending on the assessment methodology used, but in broad lines the TPU elastomer was assessed as having higher environmental impact than the PO elastomer and plasticised PVC, whereas the last two were assessed as having quite equal overall impacts. It should be noted that while some human toxicity aspects were included, the health effects of DEHP could not be included in the assessment, as no conclusive toxicity data suited for the methodology had been identified. The major assessed effects appeared quite influenced by energy resource depletion and energy related emissions.
The overall conclusion can be drawn that a DEHP-free medical grade flexible polymer is available. It is primarily based on low toxic olefins and has similar or lower life cycle impacts than plasticised PVC. The material, Meliflex, is more expensive per weight, but as less material is needed per unit of the medical functions investigated, this partly outbalances the price difference (Melitek, 2009). Meliflex is designed and produced specifically for pharmaceutical packaging and medical devices applications. Grades are available for tubing extrusion, films blown by cast extrusion, for injection moulding and for blow moulding.
Previously an environmental and health assessment of two alternative materials has been conducted by Stuer Lauridsen et al., 2001: PU (polyurethane) and LDPE (low density polyethylene). On the basis of the available data it was not possible to make a full assessment of the materials. It is however in the report recognised that LDPE has a low toxicity and that LDPE does not release large quantities of monomers or oligomers.
A study on alternatives to soft PVC in building materials among others concludes that polyethylene and other polyolefins have better environmental characteristics than PVC for many applications in the building industry (Andersson, 2002).
6.2 Summary on other flexible polymers
As described above, another DEHP-free flexible medical grade material, TPU, is available. The LCA performed by Stripple et al. (2007) indicated, that its life cycle is more energy-intensive and have higher emissions of prioritised pollutants. According to Stripple et al. (2007), while plasticised PVC is the traditional flexible material of choice in the medical market, TPU also has a significant part of the market. No price data have been collected for TPU for this study.
Alternative materials for toys
Postle et al. (2000) note that a number of companies have undertaken substitution to entirely different plastic products rather than simply different plasticisers. For those products which are specifically intended to be placed in the mouth, the substitute plastics which appeared to be most widely used were polyethylene (PE) and ethylene vinyl acetate (EVA). These materials can reportedly be used adequately in the products in question. However, the technical performance of the final product has been indicated to be often slightly inferior to that obtained with PVC. For example, products produced from these materials may sometimes have lower resistance to biting and tearing than plasticised PVC. The products may also have reduced longevity. In terms of the wider range of toys and childcare articles, plastics which are reported to be used as substitutes for plasticised PVC include various forms of polyethylene (LDPE, and LLDPE) styrenic block copolymers and again EVA, as shown in Table 6.1.
Table 6.1 Summary of technical suitability and use of alternative flexible materials for use in toys (Postle, et al, 2000)

Alternative materials for medical devises
The Toxics Use Reduction Institute (TURI, 2006) investigated a number of alternatives materials for three application areas: Resilient flooring, wall coverings and medical devices for neonatal care. For medical application several alternative materials were assessed for both sheet (EVA, polyolefins and glass) and tubing (polyolefins, silicone and TPU) applications. Many manufacturers were offering non-DEHP and/or non-PVC alternatives for both sheet and tubing uses. The study does not provide a clear conclusion for medical applications. For the detailed assessment summary reference is made to the study report. Products utilising the alternative materials, either singly or in multi-layer laminates, were commercially available for sheet and tubing device applications with the notable exception of red blood cell storage.
Review of life cycle assessments (LCA) of PVC and alternative materials
In a study for the European Commission, Baitz et al. (2004 compiled an overview of the publicly available information on LCA on PVC and competing materials, for a variety of applications. Approximately 100 LCAs related to PVC were identified, of these 30 included comparisons at the application level. For roofing applications the study concludes that higher quality of the systems (thermal conductivity per thickness of roofing sheet layers) as well as the accuracy of the laying and maintenance processes have a large influence over the reduction of environmental impacts. Additionally, the study concludes that ‘green roofing’ (e.g. planting on the roof) further decreases environmental impacts because of the subsequent longer lifetime of the roofing systems. Three polymer solutions (one PVC system and two competing systems) have the potential to perform better, with similar environmental impacts on global warming, acidification and ozone formation over the life cycle. The study reports that some polymer solutions tend to have lower environmental impacts than competitive systems. Few comparative LCA studies pertaining to consumer goods are available. No useful general conclusions on material comparisons could be drawn.
In a review of various studies on alternative materials to plasticised PVC it is concluded that the available reviewed studies demonstrate that for many applications of DEHP/PVC alternative materials exist at similar prices (COWI, 2009a). Many of the materials seems to have equal or better environmental, health and safety, performance and cost profiles, but clear conclusion are complicated by the fact that not all aspects of the materials’ lifecycles have been included in the assessments.
Pedersen (1999) produced a simplified matrix indicating a gross differentiation of polymer materials in some overall categories of health and environment impacts. It is presented in Table 6.2 based on its presentation in the publication Nordic Ecolabelling (2007). Being simplified, and being based on late 1990 knowledge, the table should likely be interpreted with some caution. Note that the low impact classes 1 and 2 include the substances polyethylene (PE), poly(isobutylene) (PIB), ethylene vinyl acetate (EVA), styrene ethylene butylene styrene co-block polymer (SEBS), styrene isoprene block polymer and silicone.
Conclusions
A number of flexible polymers are available which can substitute for many traditional uses of flexible PVC. Polyethylene (PE), polyolefin elastomers, different polyurethane (PU) qualities, ethylene vinyl acetate (EVA) and different rubber types are examples of among others. For many flexible PVC uses, also other substitute materials than flexible polymers exist. The LCA-based, application-focused assessments are few, and often clear-cut conclusions could not be made. But many materials exist with seemingly equal or better environmental, health and safety, performance and cost profiles. The assessment made here does not allow for a more detailed analysis of possibilities and limitations in the coverage of alternative flexible polymers.
| Category | Description | Material |
|---|---|---|
| 1 | The polymer materials in this category contain particularly health or environmentally hazardous substances, which are crucial for the manufacturing or for the properties in use of the polymer. The substances added or generated in the production, use or disposal phase may require special end-of-pipe precautions or protective equipment and may result in significant health or environmental impacts. It should be noted that where the necessary end-of-pipe precautions and protective equipment are adequately installed during manufacturing, the impacts on health and environment can be made negligible. |
Polyethylene – PE Poly (isobutylene) – PIB Ethylene vinyl acetate – EVA |
| 2 | The polymer materials in this category contain health or environmental hazardous substances, which are crucial for the manufacturing or for the properties in use of the polymer. The substances added or generated in the production, use or disposal phases may not according to law, require any special end-of pipe treatment or special for protective equipment but might have health or environmental impacts. The polymer materials, which fulfil the first criteria in Category 1 but require large energy consumption to manufacture or which generate relatively low levels of energy upon incineration, are also listed in Category 2. |
Styrene ethylene butylene styrene co-block polymer – SEBS Styren isoprene block polymer Silicone |
| 3 | The polymer materials in this category contain particularly health or environmentally hazardous substances, which are crucial for the manufacturing or for the properties in use of the polymer. The substances added or generated in the production, use or disposal phase may require special end-of-pipe precautions or protective equipment and may result in significant health or environmental impacts. It should be noted that where the necessary end-of-pipe precautions and protective equipment are adequately installed during manufacturing, the impacts on health and environment can be made negligible. |
Latex/Natural rubber (cispolyisoprene) – NR Polyvinyl chloride not plasticized with DEHP – PVC (soft) Thermoplastic Polyurethane – TPU Polyurethane foam – PUR foam |
| 4 | The polymer materials in this category are regarded as particularly hazardous to health and environment. This category includes polymer materials that otherwise would be in category 1-3 but which contain additives considered as hazardous to health and environment. | Polyvinyl chloride plasticized with DEHP – PVC(soft) Halogenated additives Additives with heavy metals Fire-retardant based on bisphenols or diphenyl Plasticizers based on DEHP Other additives with the ability to act as endocrine disrupters |
[1] The assumed TPU composition studied was based on hydrogenated methylene diisocyanate (HMDI), polytetramethylene ether glycol (PTMEG) and 1,4-butadiol.
References
[Data sources for the environmental and health assessment are included in Annex 3]
Andersson, M. 2002. Formidlingsprojekt om alternativer til blød PVC i byggebranchen. Arbejdsrapport fra Miljøstyrelsen Nr. 24/2002. The Danish Environmental Protection Agency, Copenhagen.
Arbeitsgemeinschaft PVC und Umwelt e.V., 2006. Plasticiser market data. January 2006, accessed August 2009 at http://www.agpu.com/fileadmin/user_upload/information_
herunterlade/Marktdaten%20Weichmacher_230106.lin_en.pdf
ATSDR. Toxicological Profile for Di-n-octylphthalate (DNOP). 1997. U.S. Department of Health and Human Services, Agency for Toxic Substances and Disease Registry, Atlanta. http://www.atsdr.cdc.gov/toxprofiles/tp95-c4.pdf
Baitz, M. , J. Kreißig, E. Byrne, C. Makishi, T. Kupfer, N. Frees, N. Bey, M.S. Hansen, A. Hansen, T. Bosch, V. Borghi, J. Watson and M. Miranda. 2004. Life Cycle Assessment of PVC and of principal competing materials. PE Europe GmbH and others for the European Commission.
BASF, 2009. Technical leaflet on Plastomoll DNA, BASF, accessed June 2009 at: http://www.plasticizers.basf.com/icms/streamer?fid=217082
BASF, 2009: Product information on Hexamoll DINCH, accessed August 2009 at http://www.hexamoll.com/icms/basf_6/en/dt.jsp?setCursor=1_216770
BBP Information Centre. 2009. An information resource on the plasticiser butyl benzyl phthalate (BBP). An initiative of the European Council for Plasticisers and Intermediates (ECPI), Brussels.
Biedermann-Brem, S., M. Biedermann, S. Pfenninger, M. Bauer, W. Altkofer, K. Rieger, U. Hauri, C. Droz, K. Grob. 2008. Plasticisers in PVC toys and childcare products: What succeeds the phthalates?. Market Survey 2007. Chromatographia 68: 227-234.
Brandt, U.K. and E. Hansen. 2009. Ftalater i afgiftsbelagte produkter [phthalates in products subject to tax]. COWI for Danish Environmental Protection Agency (unpublished).
Buck Jensen, M. 2009. Højteknologifonden støtter udvikling af alternativ til phthalater (in Danish; Hi-tech foundation supports development of alternative to phthalates). Ingeniøren, June 2009. Accessed July 2009 at http://ing.dk/artikel/99245-hoejteknologifonden-stoetter-udvikling-af-alternativ-til-phthalater?highlight=castor+olie
Cadogan, D. 2006. Plasticisers: An update. Presentation at: Plasttekniske Dager, Oslo, 8-9 November 2006. Cadogan on behalf of European Council for Plasticisers and Intermediates (ECPI).
Christensen, C.L., L. Høibye and E. Hansen. 2007. Forbrug af phthalater i Danmark i historisk perspektiv [Consumption of phthalates in a historic perspective]. COWI A/S for Danish Environmental Protection Agency (unpublished).
COWI, 2000. Kortlægning og vurdering af substitutionsmuligheder for phthalater i udvalgte produkter (Survey and assessment of substitution options for phthalates in selected products; in Danish). Environmental project no. 560, Danish Environmental Protection Agency, Copenhagen.
COWI. 2009a. Data on manufacture, import, export, uses and releases of bis(2-ethylhexyl phthalate) (DEHP) as well as information on potential alternatives to its use. COWI in cooperation with Entec and IOM for European Chemicals Agency (ECHA), Helsinki.
COWI. 2009b. Data on manufacture, import, export, uses and releases of dibutyl phthalate (DBP) as well as information on potential alternatives to its use. COWI in cooperation with Entec and IOM for European Chemicals Agency (ECHA), Helsinki.
COWI. 2009c. Data on manufacture, import, export, uses and releases of benzyl butyl phthalate (BBP) as well as information on potential alternatives to its use. COWI in cooperation with Entec and IOM for European Chemicals Agency (ECHA), Helsinki.
Danisco, 2009. Personal communication. 2009.
Danisco, 2009b. Product description "COMGHA - The sustainable plasticiser for PVC". Danisco April 2008. Received from Danisco, 2009.
Eastman, 2006. Eastman TXIB formulation additive for vinyl plastisols. Acessed August 2009 at http://www.eastman.com/Literature_Center/L/L231.pdf
Eastman, 2009c. Eastman Plasticizers, Selector Chart. Accessed 2009 at http://www.eastman.com/Literature_Center/L/L174.pdf
Eastman, 2009f. Eastman product data sheet for TXIB, accessed June 2009 at: http://ws.eastman.com/ProductCatalogApps/PageControllers/
ProdDatasheet_PC.aspx?Product=71066420&sCategoryName=Generic
Eastmann. 2001. Eastmann 425 plasticiser. For vinyl plastisols and vinyl compounds. Eastmann Chemical Company, Kingsport. Accessed at: http://www.eastman.com/NR/rdonlyres/FCB732B0-9A7A-483C-B76D-B8C481EC346B/0/L230.pdf
ECB. 2003. European Union Risk Assessment Report: 1,2-benzenedicarboxylic acid, di-C8-10- branched alkyl esters, C9-rich and di-“isononyl” phthalate (DINP). Institute for Health and Consumer Protection, European Chemicals Bureau, Ispra.
ECB. 2004. European Union Risk Assessment Report: dibutyl phthalate (DBP). Institute for Health and Consumer Protection, European Chemicals Bureau, Ispra.
ECB. 2007. European Union Risk Assessment Report: Benzyl butyl phthalate (BBP). Institute for Health and Consumer Protection, European Chemicals Bureau, Ispra
ECB. 2008. European Union Risk Assessment Report: bis(2-ethylhexyl)phthalate (DEHP). Institute for Health and Consumer Protection, European Chemicals Bureau, Ispra.
ECPI. 2009. Information from the website of the European Council for Plasticisers and Intermediates (ECPI), Brussels at: www.ecpi.org
Exxon Mobil. 2009. Jayflex plasticisers. http://www.exxonmobilchemical.com./Public_Products/Oxo/Plasticisers
/Worldwide/Jay_ProductFrontPage.asp
ExxonMobil (2009). Personal communication, 2009.
ExxonMobil (2009b). Jayflex DINA sales specifications, accessed June 2009 at: http://www.exxonmobilchemical.com/Public_Files/Oxo/Plasticizers/
NorthAmerica/Jayflex_DINA_datasheet.pdf:
FCPSA. 2008a. Weekmakers in speelgoed- en kinderverzorgingartikelen. VERVOLGONDERZOEK 2008. The Food and Consumer Product Safety Authority, the Dutch Ministry of Agriculture, Nature and Food Quality. (Deel)projectnummer: ND072201, 7 maart 2008 [In Dutch] Accessed at: http://www.vwa.nl/cdlpub/servlet/CDLServlet?p_file_id=31625
FCPSA. 2008b. Weekmakers in speelgoed- en kinderverzorgingartikelen. The Food and Consumer Product Safety Authority, the Dutch Ministry of Agriculture, Nature and Food Quality. October 2008 [In Dutch] Accessed at: http://www.vwa.nl/cdlpub/servlet/CDLServlet?p_file_id=31627
Genovique, 2009. Personal communication. 2009.
Genovique, 2009b. Product information on Benzoflex 2088 and 9-88 accessed August 2009 at http://www.genovique.com/cms.aspx?TabID=29
Hoffmann, L. 1996. Massestrømsanalyse for phthalater [Substance flow analyis of phthalates]. Environmental Project 320. Danish Environmental Protection Agency, Copenhagen.
Karbæk, K. 2003. Evaluation of plasticisers for PVC for medical devices. Environmental Projekt Nr. 744. Danish Environmental Protection Agency, Copenhagen
Krauskopf, L.G. and A. Goodwin. Plasticisers. In; C.E. Wilkes, C. A. Daniels and J.W. Summers. PVC Handbook. Hanser Gardener Publ. Cincinnati.
Laxness, 2009. Personal communication. 2009.
Laxness, 2009b. Product data sheet on Mesamoll from Lanxess.
Melitek, 2006. Environmental Product Declaration, Meliflex. Received from Melitek, 2009.
Melitek, 2009. Personal communication.
MPW, 2008. Phthalates controversy shows no sign of slowing. By John Clark. November 14th, 2008, accessed at http://mpw.plasticstoday.com/node/15774:
Nilsson, N.H., J. Lorenzen and O.C. Hansen. 2002. Substitution af phthalatblødgjort PVC-vandmadras hos Akva Waterbeds [Substitution of phthalate softened PVC water mattress by Akva Waterbeds]. Environmental Projekt Nr. 739 2002. Danish Environmental Protection Agency, Copenhagen. (In Danish)
Nordic Eco-labelling, 2007. About Swan labelling of Peritoneal Dialysis (PD) and Intravenous (IV) sets - Draft for review 14 March 2007 - Background to ecolabelling.
Pedersen, L.B., 1999. Plast og Miljø (Plastic and environment; in Danish), Teknisk Forlag, Copenhagen, 1999. As cited in (Nordic Eco-labelling, 2007).
Postle, M., C. Corden, M. van den Berg and T. Sanderson. 2000. The availability of substitutes for soft PVC containing phthalates in certain toys and childcare articles. RPA and Ritox for the European Commission.
SCENIHR. 2008. Opinion on the safety of medical devices containing DEHP-plasticized PVC or other plasticiser s on neonates and other groups possibly at risk. Scientific Committee on Emerging and Newly Identified Health Risks (SCENIHR). European Commission, Brussels.
Sears J.K. and Darby J.R., 1982. The technology of plasticizers. Wiley-Interscience, John Wiley & Sons, New York, 1982.
SPIN. 2008. Substances in Preparations in Nordic Countries. Database based on data from the Product Registries of Norway, Sweden, Denmark and Finland. At: http://195.215.251.229/DotNetNuke/default.aspx
Stripple, H. R. Westman and D. Holm.2008. Development and environmental improvements of plastics for hydrophilic catheters in medical care - an environmental evaluation. Journal of Cleaner Production 2008;16: 1764 - 1776
Stuer-Lauridsen, F., S. Mikkelsen, S. Havelund, M. Birkved and L.P. Hansen. Environmental and health assessment of alternatives to phthalates and to flexible PVC. Environmental Project No. 590. 2001. Danish Environmental Protection Agency, Copenhagen.
TURI. 2006. Five chemicals study. Toxics Use Reduction Institute (TURI), University of Massachusetts Lowell, for the Commonwealth of Massachusetts. Chapter on alternatives to DEHP available at: http://www.turi.org/library/turi_publications/five_chemicals_study/
final_report/chapter_7_dehp#7.3
Vertellus, 2009. Personal communication. 2009.
Vertellus, 2009b. Morflex Tech Bulletin 101, received from Vertellus, 2009)
Wilson, 1995. Plasticisers - Principles and practices. The Institute of Materials, London, 1995. Citing BP Chemicals.
Annex 1 Companies and organisations contacted
Organisations
Plastindustrien i Danmark, Copenhagen
Foreningen af Legetøjsfabrikanter i Danmark
Nordic Association of Toy Manufacturers
Companies
Baby Sam A/S
BASF
BRIO AB
Danisco
Dantoy A/S
Dracco, Allerød
Eastman Chemicals
Genovique
Hama
Hasbro
Indo-Nippon ChemicalsExxon Mobil
K.E. Mathiasen A/S
Lanxess (formerly Bayer)
Lego
Poul Willumsen A/S
Ripladan
TOP-TOY A/S
Vertellus (formerly Morflex)
Annex 2 Introduction to plasticiser families
This Annex provides some basic information on the different plasticiser substances families summarised in section 3.2.
Terephthalates
Terephthalic acid is identical to phthalic acid, except for the physical placement of one part of the molecule. It may be reacted with an appropriate alcohol to produce terephthalate esters which are used as plasticisers. In practice, terephthalates are more commonly used in the USA than elsewhere. An example of a commonly used terephthalate is DEHT, which in spite of its close chemical similarity with DEHP has distinctly different health and environment characteristics (TURA, 2006). According to Krauskopf and Godwin (2005), DEHT is commercially available at similar price as DEHP.
Benzoates
Benzoates are the esterification products of benzoic acid and selected glycols, usually diols. Preferred glycols are dipropylene glycol and butane diols. One commonly used benzoate is dipropylene glycol dibenzoate, DGD (commercially Benzoflex® 9-88). Its preferred use is in PVC flooring products, owing to its strong solvating strength, and it reportedly controls plasticiser bleeding into asphalt adhesives. In vinyl sheet flooring, the benzoate enhances processing, while the low molecular weight contributes a hardened, stain resistant surface, due to volatilization, similar to the effect of BBP in flooring. Benzoates are generally strong solvaters due to the high aromaticity, as are lower molecular weight phthalates such as dihexyl (DHP) and butyl, octyl (BOP), as well as butylbenzyl (BBP). Commercial practice includes the use of up to 10–20% of the plasticiser system as “strong solvating” type plasticisers, such as aryl-alkyl phthalates (e.g. BBP), benzoates, sulfonates, and so forth (Krauskopf and Godwin, 2005).
Citrates
Citrate plasticisers are tetraesters (Krauskopf and Godwin, 2005). Examples of citrate plasticiser esters include triethyl citrate, acetyl triethyl citrate, tributyl citrate, acetyl tributyl citrate and tri-(2-ethylhexyl)-citrate. They are produced from citric acid and are traditionally used to plasticise vinyl resins used in applications including medical equipment and packaging films. Of the approximately 230,000 tons of citric acid used annually in Western Europe, 9% are applied for industrial uses such as for plasticisers. Tributyl citrate is used in PVC, polyvinyl chloride/vinylidene chloride copolymers or polyvinyl chloride/vinyl acetate resins that are subsequently used for such items as food-wrapping film. Among the advantages of tributyl citrate is that it is heat-stable and does not discolour when processed in compounded resins. Acetyl tributyl citrate is used in electrical coatings and casings because of its solvating characteristics. It is also used in inks. Acetyl tributyl citrate and acetyl triethyl citrate are also used in hair sprays and aerosol bandages. Triethyl citrate has applications in the food industry as flavour and flavour emulsion (ECPI 2009).
Trimellitates
These materials are produced by the esterification of a range of alcohols with trimellitic anhydride (TMA), which is similar in structure to phthalic anhydride with the exception of a third carbonyl group on the aromatic ring. Consequently, esters are produced in the ratio of three moles of alcohol to one mole of anhydride. Common esters in this family are Tris-2-ethyhexyl trimellitate (Tri-octyl trimellitate - TOTM), L79TM (an ester of mixed semi-linear C7 and C9 alcohols, and L810TM, an ester of mixed C8 and C10 linear alcohols. The principle features of these esters, when processed with PVC, is their low volatility, and consequently large volumes of trimellitate esters are used in high specification electrical cable insulation and sheathing. The extraction and migration resistance of these materials are also significantly improved relative to the phthalates. The low volatile loss also results in usage in automotive interior applications where the issue of windscreen fogging is important. In this respect they often compete with the linear high molecular weight phthalates such as 911P (ECPI 2009).
Adipates
Alcohols of similar chain length to those used in phthalate manufacture can be esterified with adipic acid, rather than phthalic anhydride, to produce the family of adipate plasticisers which are a part of the "aliphatic dibasic esters" in the table above. For example, esterification of 2-ethylhexanol with adipic acid yields di-2-ethylhexyl adipate (DEHA), also known as di-octyl adipate (DOA), which is reported to be the most used adipate plasticiser (ECPI 2009; Krauskopf and Godwin 2005). The family of adipic acid esters used in PVC applications improves low temperature performance relative to phthalates and give significantly lower plastisol viscosities in plastisol applications, due to the lower inherent viscosities of the plasticisers themselves. Adipates used are typically based on alcohols in the C8 to C10 range (ECPI 2009). Typically, lower molecular weight alcohols are used with higher molecular weight acids, and vice versa, such that the total carbon content per molecule ranges between C18 and C26. This maintains the apolar/polar ratio required to provide PVC compatibility along with low temperature properties (Krauskopf and Godwin, 2005).
Relative to phthalates, adipates have higher volatilities and higher migration rates, and are generally higher priced. As a result, it is not uncommon for adipates to be used in blends with phthalates to produce a compromise of properties (ECPI, 2009). Diisononyl adipate (DINA) is used for low temperature applications requiring lower plasticiser volatility (Krauskopf and Godwin, 2005).
Sebacatesa and azelates
Esters produced from 2-ethylhexanol and higher alcohols with linear aliphatic acids are used in some demanding flexible PVC applications where superior low temperature performance is required. Di-2-ethylhexyl sebacate (DOS) and di-2-ethylhexyl azelate (DOZ) are the most common members of this group, but di-isodecyl Sebacate (DIDS) is also used. They give good low temperature performance in combination with adipates. Their usage has generally been limited to extremely demanding low temperature flexibility specifications (e.g. underground cable sheathing in arctic environments) (ECPI 2009).
Phosphates
Phosphate plasticisers may be considered as “inorganic esters”, where the phosphate plays the role otherwise played by carboxylic acids, and bonds with alcohols or phenols to form the desired plasticisers. An important feature of phosphate plasticisers is that they, in addition to plasticising the PVC, act as a flame retardant. While hard PVC is quite resistant to fire, the addition of plasticisers generally decreases the fire resistance, and additional fire resistance can be provided by adding a fire retarding plasticiser. Commercial phosphate plasticisers use combinations of aryl (aromatic) and C8 and C10 alkyl (carbon chain) groups to offer a balance of fire reduction, volatility, and efficiency. 2-ethyhexyl diphenyl phosphate has widespread use in flexible PVC applications due to its combination of properties of plasticising efficiency, low temperature performance, migration resistance and fire retardancy. Tris(2-ethylhexyl) phosphate, tricresyl phosphate (TCP) are other examples of phosphate plasticisers. Phosphate plasticisers may be combined with other plasticisers to reduce formulating costs (ECPI 2009; Krauskopf and Godwin 2005).
Sulfonates
Sulfonates exhibit strong solvency for PVC. One example is the phenyl cresyl esters of pentadecyl sulfonic acid. It is reportedly resistant to hydrolyses and diffusion controlled plasticiser losses (Krauskopf and Godwin, 2005). ASE (alkylsulfonic phenylester) is also an example of a sulfonate plasticiser.
Epoxides
Epoxy plasticisers enhance thermal and UV stability of PVC. They are the only class of plasticisers that undergo a chemical grafting (side-chain bonding) onto the PVC polymer. They are thus internal plasticiser, contrary to phthalates. This chemical family is composed of essentially two types of epoxidized natural products. Epoxidized oils are prepared from soybean oil (ESBO or ESO) and linseed oil (ELSO). These oils have molecular weights of approx. 1,000, causing them to perform as low volatility plasticisers. The primary performance attributes of epoxy plasticisers are their role in PVC stabilization, which is accomplished at low concentrations in the PVC.
Polymeric plasticisers
Polymeric plasticisers are typically polyesters, with a molecular weight range from 1,000 to 8,000. Polyester plasticisers often have the structure of combined (bonded) propylene glycol or butylene glycol with aliphatic dibasic acids such as adipates. The greater the plasticiser viscosity, or molecular weight, the greater its permanence (e.i. resistance to loss by diffusion and evaporation). Polymeric plasticisers composed of branched structures are more resistant to diffusivity losses than those based on linear isomeric structures; on the other hand they are more susceptible to oxidative attack (degradation). The polarity, or the oxygen-to-carbon ratio, also impacts extraction resistance of the polymerics. Lower polarity materials exhibit better extraction resistance towards polar extraction fluids such as soapy water (Krauskopf and Godwin, 2005).
Other plasticiser types
Other plasticisers exist, many of which are also (like phthalates, etc.) esters of alcohols and carboxylic acids.
Pentaerythritol esters are a type of “miscellaneous” plasticisers that impart both low volatility and diffusivity. Pentaerythritol is a tetra alcohol esterified with straight chain fatty acids to make plasticisers (Krauskopf and Godwin, 2005). Trimethyl pentanyl diisobutyrate (TXIB) is another example (Eastman, 2009).
Annex 3 Summary of technical performance parameters
A summary of some technical performance parameters for the assessed alternatives compared to DEHP, DBP, BBP and other selected phthalates is given below..
| Plasticiser in PVC, conc. 40% =67 phr in same PVC resin*1 | Shore A hardness*2 | Volatility,% lost, 1 day at 87 °C over activated carbon | Extracted in water,% | Extracted in kerosene (jet fuel, etc.),% |
|---|---|---|---|---|
| Phthalates: | ||||
| DEHP | 69 | 4.5 | 0.01 | 44 |
| DEHP (PVC2)*3 | 73 | 3.6 | 0.02 | 54.7 |
| DBP | 62 | 45.4 | 0.25 | 9.1 |
| BBP | 68 | 7.7 | 0.07 | 3.4 |
| DIDP | 71 | 1.8 | 0.03 | 74 |
| DINP | 73 | 2.1 | 0.07 | 76.7 |
| Non-phthalates: | ||||
| ASE | 72 | 5.3 | 0.03 | 4.8 |
| ATBC | 73 | 17.8 | 0.09 | |
| DEGD (as single substance) | 69 | 5.5 | 0.75 | 3.4 |
| DGD | 71 | 7.9 | 0.45 | 2.9 |
| DEHT (PVC2)*3 | 76 | 1.9 | 0.09 | 70.8 |
| DINA | 72 | 4.1 | 0.14 | 80.4 |
| TXIB | 76 | 23.7 | 2.83 | 5.2 |
| COMGHA*4 | 88.0 | NA | NA | NA |
| DEHP *4 | 90.0 | NA | NA | NA |
Notes: *1: phr = parts per hard resin, meaning parts per weight of hard PVC. *2: A measure for the plasticiser's efficiency in making PVC flexible; the lower the number, the softer the PVC and the more efficient plasticiser. *3 Measured performance in another PVC resin (the same for DEHP and EHT). *4 Data from Danisco; at concentration 40 phr; PVC resin type not specified.
Annex 4 Background data for the environmental and health assessment
Click here to see: Annex 4 Background data for the environmental and health assessment
Version 1.0 November 2010, © Danish Environmental Protection Agency